Advanced Synthesis of S-phenyl-4-toluenesulfonate: Technical Upgrades for Commercial Manufacturing
The chemical landscape for producing critical sulfiding reagents is undergoing a significant transformation, driven by the urgent need for safer and more efficient manufacturing protocols. As detailed in patent CN102531983A, a groundbreaking methodology has been established for the synthesis of S-phenyl-4-toluenesulfonate, a vital intermediate widely utilized in pharmaceutical and agrochemical applications. This innovative approach fundamentally shifts the paradigm from hazardous legacy processes to a robust, solid-state reagent system that prioritizes operator safety and environmental stewardship without compromising on yield or purity. By leveraging the unique reactivity of sodium p-toluenesulfinate and N-phenylmercaptosuccinimide, this technology offers a compelling solution for reliable pharmaceutical intermediate supplier networks seeking to modernize their production capabilities. The strategic implementation of this patent not only addresses the technical challenges of scalability but also aligns perfectly with the growing global demand for green chemistry solutions in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of S-phenyl-4-toluenesulfonate relied heavily on the condensation of p-toluenesulfonyl chloride with thiophenol, typically conducted in dichloromethane with pyridine as a base. This traditional pathway is fraught with severe operational hazards, primarily due to the intrinsic properties of thiophenol, which is notorious for its extreme toxicity, high volatility, and repulsive odor that necessitates specialized containment infrastructure. Furthermore, p-toluenesulfonyl chloride is chemically unstable and prone to hydrolysis, leading to inconsistent batch quality and the generation of acidic byproducts that corrode reactor vessels. The reliance on chlorinated solvents like dichloromethane exacerbates environmental compliance issues, creating a complex waste stream that is costly to treat and dispose of safely. These cumulative factors result in elevated production costs, extended downtime for maintenance, and significant regulatory scrutiny, making the conventional route increasingly untenable for modern cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel synthetic route disclosed in the patent utilizes sodium p-toluenesulfinate and N-phenylmercaptosuccinimide as the core building blocks, reacting them in benign organic solvents such as toluene or n-hexane. This method operates under mild conditions, typically between 10°C and 50°C, which drastically reduces energy consumption and thermal stress on the equipment. The substitution of liquid thiophenol with solid, non-volatile N-phenylmercaptosuccinimide eliminates the risk of inhalation exposure and odor complaints, thereby simplifying facility requirements and enhancing workplace safety. Moreover, the stability of the sodium sulfinate salt ensures consistent stoichiometry and reaction kinetics, leading to reproducible high yields often exceeding 85%. This technological leap facilitates the commercial scale-up of complex pharmaceutical intermediates by providing a cleaner, safer, and more economically viable pathway that minimizes the formation of hazardous byproducts and streamlines the downstream purification process.
Mechanistic Insights into Nucleophilic Substitution and S-S Bond Formation
The core of this synthesis lies in a sophisticated nucleophilic substitution mechanism where the sulfur atom of the sodium p-toluenesulfinate acts as a potent nucleophile attacking the electrophilic sulfur center of the N-phenylmercaptosuccinimide. This interaction facilitates the cleavage of the N-S bond in the succinimide derivative and the concurrent formation of the desired S-S bond, resulting in the target S-phenyl-4-toluenesulfonate structure. The reaction proceeds efficiently in non-polar or moderately polar aprotic solvents, which help to stabilize the transition state without interfering with the ionic character of the sulfinate salt. The choice of solvent plays a critical role in solvating the ionic species while maintaining the solubility of the organic succinimide derivative, ensuring a homogeneous reaction environment that maximizes collision frequency between reactants. Understanding this mechanistic nuance is essential for reducing lead time for high-purity pharmaceutical intermediates, as it allows process chemists to fine-tune parameters such as addition rates and temperature profiles to suppress side reactions.
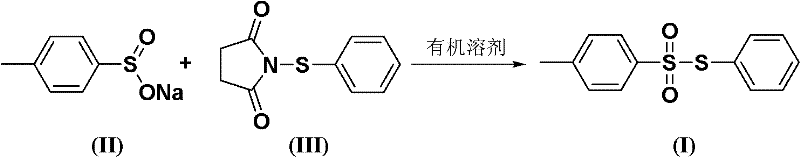
Impurity control is another critical aspect governed by the reaction mechanism, particularly regarding the prevention of over-oxidation or hydrolysis of the sensitive sulfinate species. The mild temperature range of 30°C to 50°C specified in the patent is strategically chosen to provide sufficient activation energy for the substitution while avoiding thermal degradation pathways that could generate sulfone or disulfide impurities. Additionally, the use of a slight excess of sodium p-toluenesulfinate, with molar ratios ranging from 1:1.0 to 1:2.0, ensures complete consumption of the more valuable N-phenylmercaptosuccinimide, driving the equilibrium towards the product. The subsequent workup involving saturated brine washing effectively removes inorganic salts and residual succinimide byproducts, yielding a crude product with purity levels consistently above 98.5%. This inherent selectivity reduces the burden on final purification steps, such as recrystallization or column chromatography, thereby preserving overall yield and minimizing solvent usage in the final stages of production.
How to Synthesize S-phenyl-4-toluenesulfonate Efficiently
Implementing this synthesis requires precise control over reagent addition and thermal management to replicate the high efficiencies observed in the patent examples. The process begins with the dissolution of sodium p-toluenesulfinate in a preferred solvent like toluene, followed by the controlled dropwise addition of the N-phenylmercaptosuccinimide solution to manage the exotherm and ensure uniform mixing. Maintaining the reaction temperature within the optimal window of 30°C to 50°C for a duration of 5 to 8 hours is crucial for achieving maximum conversion without compromising product integrity. The detailed standardized synthesis steps, including specific solvent volumes and workup procedures, are outlined below to guide process engineers in establishing a robust manufacturing protocol.
- Dissolve sodium p-toluenesulfinate in an organic solvent such as toluene or n-hexane under stirring at a controlled temperature between 10°C and 50°C.
- Slowly add a solution of N-phenylmercaptosuccinimide dissolved in the same organic solvent to the reaction mixture, maintaining the molar ratio between 1: 1.0 and 1:2.0.
- After the reaction is complete, remove the solvent, add saturated brine, separate the organic layer, and evaporate to dryness to obtain the final product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this novel synthesis route offers transformative advantages that directly impact the bottom line and supply chain resilience. By replacing hazardous and volatile raw materials with stable, solid alternatives, companies can significantly reduce the costs associated with specialized storage, handling, and personal protective equipment. The elimination of foul-smelling thiophenol not only improves community relations and regulatory compliance but also removes the need for expensive scrubbing systems and odor control technologies. Furthermore, the use of common, recyclable solvents like toluene simplifies solvent recovery operations, leading to substantial reductions in raw material expenditure and waste disposal fees. These operational efficiencies translate into a more competitive cost structure, enabling suppliers to offer better pricing while maintaining healthy margins in a volatile market environment.
- Cost Reduction in Manufacturing: The shift to stable solid reagents eliminates the premium pricing and logistical complexities associated with handling hazardous liquids like thiophenol and unstable sulfonyl chlorides. This change reduces the need for specialized containment infrastructure and lowers insurance premiums related to chemical storage risks. Additionally, the high reaction yields and simplified workup procedure minimize raw material waste and solvent consumption, driving down the variable cost per kilogram of the final product. The overall process efficiency allows for a leaner manufacturing operation with reduced utility consumption and lower labor intensity for safety monitoring.
- Enhanced Supply Chain Reliability: Sourcing stable solid intermediates like sodium p-toluenesulfinate and N-phenylmercaptosuccinimide is inherently more reliable than procuring volatile and odorous liquids that face strict transportation regulations. The robustness of these materials ensures longer shelf life and reduced risk of degradation during transit, mitigating the potential for supply disruptions caused by spoiled batches. This stability allows for larger inventory buffers and more flexible production scheduling, ensuring consistent delivery timelines to downstream pharmaceutical and agrochemical clients. Consequently, the supply chain becomes more resilient to external shocks and regulatory changes affecting the transport of hazardous goods.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly toxic byproducts make this process exceptionally scalable from pilot plant to multi-ton commercial production without significant engineering hurdles. The reduced generation of hazardous waste simplifies effluent treatment processes, ensuring compliance with increasingly stringent environmental protection laws globally. This environmental compatibility future-proofs the manufacturing asset against tightening regulations, avoiding costly retrofits or shutdowns. The ability to scale efficiently while maintaining a low environmental footprint positions the manufacturer as a preferred partner for sustainability-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are designed to clarify the operational benefits and feasibility of adopting this method for large-scale production. Understanding these details is crucial for stakeholders evaluating the technical readiness and economic viability of the new process.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: The novel method eliminates the use of thiophenol, a highly toxic and foul-smelling liquid, and p-toluenesulfonyl chloride, which is unstable. Instead, it utilizes stable solid reagents like sodium p-toluenesulfinate and N-phenylmercaptosuccinimide, significantly reducing occupational health risks and equipment corrosion requirements.
Q: Which solvents provide the highest yield for S-phenyl-4-toluenesulfonate production?
A: According to patent data, aromatic hydrocarbons like toluene and aliphatic hydrocarbons like n-hexane are preferred. Toluene specifically demonstrated yields up to 88% with purity exceeding 98.8%, outperforming polar solvents like ethanol or THF which resulted in significantly lower conversion rates.
Q: How does this process impact environmental compliance and waste treatment?
A: By avoiding volatile chlorinated solvents like dichloromethane as the primary medium and eliminating malodorous sulfur byproducts, the process generates less hazardous waste. The use of recoverable solvents like toluene further simplifies the three-waste treatment protocol, aligning with stricter environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-phenyl-4-toluenesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By integrating the safe and efficient synthesis route described in patent CN102531983A, we offer a supply solution that balances technical excellence with commercial pragmatism.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing process can benefit your specific supply chain requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this superior production method. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments tailored to your project needs, ensuring that you have all the necessary information to make informed sourcing decisions.
