Transforming Ketone Synthesis With Palladium-Catalyzed Beta-C-H Activation Technology
The landscape of organic synthesis is continually evolving, driven by the need for more efficient and selective methods to construct complex molecular architectures. Patent CN111212824B introduces a groundbreaking approach to the beta-C(sp3)-H functionalization of ketones, a transformation that has long been considered a significant challenge in synthetic chemistry. This technology leverages a palladium(II) catalytic system in conjunction with a commercially available aminooxyacetic acid promoter to achieve directed beta-iodination and arylation. Unlike previous methods that relied on stoichiometric oxime-directed palladation or transient directing groups with limited scope, this invention establishes a robust L,X-type directing group strategy. The practical implications of this patent are profound for the manufacturing of high-value pharmaceutical intermediates and fine chemicals, offering a pathway to access sterically hindered quaternary centers with superior tolerance for various functional groups. By overcoming the limitations of transient directing group approaches, this method provides a reliable solution for the direct functionalization of inert beta-C-H bonds, thereby expanding the synthetic utility of ubiquitous ketone starting materials in industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct functionalization of beta-C(sp3)-H bonds in ketones has been hindered by significant chemical and practical obstacles. Conventional strategies often relied on stoichiometric amounts of palladium or required the use of transient directing groups that were incompatible with many synthetically desirable conversion methods. Free transient directing groups, such as amino acids, frequently interfered with coupling agents and catalysts, leading to poor reaction efficiency and limited substrate scope. Furthermore, classical enolization alkylation processes are kinetically favored at the alpha-position, making the selective formation of beta-substituted products, especially those with quaternary centers, extremely difficult. The need for harsh conditions or specialized reagents in older methods often resulted in low yields and complicated purification processes, creating bottlenecks in the supply chain for complex intermediates. These limitations restricted the ability of chemical manufacturers to efficiently produce diverse beta-functionalized ketones, which are critical building blocks for numerous active pharmaceutical ingredients and agrochemicals.
The Novel Approach
The novel approach detailed in the patent data utilizes a stable oxime linkage formed from aminooxyacetic acid to create an L,X-type directing group that fundamentally changes the reactivity profile of the system. This covalent directing group overcomes the instability issues associated with imine bonds in transient strategies, allowing for the formation of highly active palladium precursors that facilitate C-H insertion. The method demonstrates exceptional compatibility with a wide range of substrates, including those containing alpha-functional groups, double bonds, and triple bonds, which were previously problematic. By employing a palladium(II) catalyst with iodine or aryl iodides, the process achieves high yields of beta-iodinated or beta-arylated products under relatively mild conditions. The ability to install the auxiliary without chromatography and subsequently remove it under standard acidic hydrolysis conditions streamlines the overall synthesis, reducing both time and material costs. This strategic shift from transient to stable covalent directing groups represents a significant technological leap, enabling the commercial scale-up of complex ketone derivatives that were previously inaccessible or economically unviable.
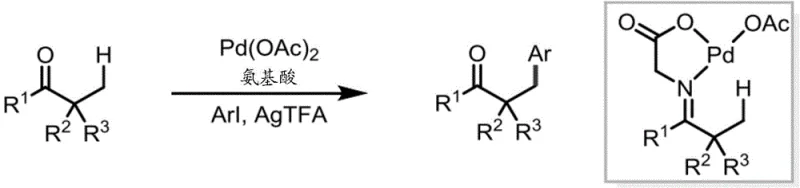
Mechanistic Insights into Pd(II)-Catalyzed Beta-C-H Functionalization
The core of this technological advancement lies in the unique coordination chemistry of the L,X-type directing group, which involves a neutral nitrogen donor and an anionic oxygen donor from the carboxylate. This bidentate coordination模式 creates a thermodynamically favorable environment for the palladium center, avoiding the formation of unstable cationic Pd(II) species that plague L,L-type directing groups. In the catalytic cycle, the carboxylate group assists in the C-H bond cleavage step, likely through a concerted metalation-deprotonation mechanism, which is more efficient than the ligand displacement required in other systems. The stability of the oxime bond ensures that the directing group remains intact throughout the catalytic turnover, preventing premature dissociation that could lead to side reactions or catalyst deactivation. This mechanistic robustness allows the reaction to proceed with high selectivity for the beta-position, even in the presence of acidic alpha-hydrogens that would typically compete in enolate chemistry. The isolation and characterization of the palladacycle intermediate provide direct evidence for this coordination mode, confirming the rational design of the directing group and validating the proposed reaction pathway for industrial replication.
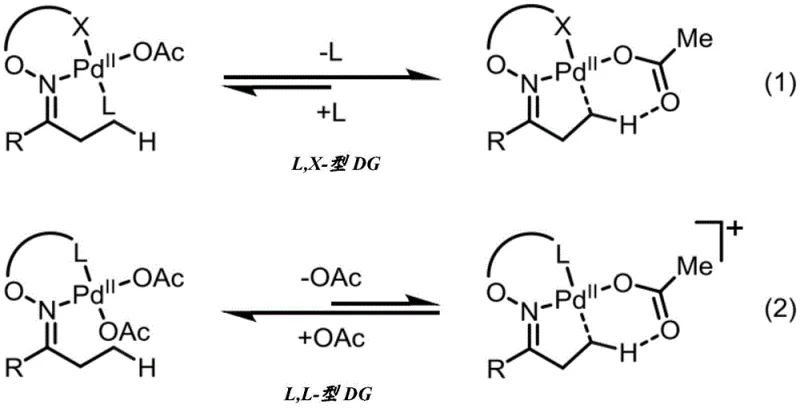
Impurity control is a critical aspect of this process, particularly given the potential for over-iodination or reaction at competing sites. The steric and electronic properties of the aminooxyacetic acid auxiliary play a pivotal role in suppressing the formation of di- or tri-iodinated byproducts, although reaction conditions can be tuned to favor mono-substitution. The use of specific solvents like 1,4-dioxane or hexafluoroisopropanol (HFIP) further enhances selectivity by modulating the solubility and reactivity of the intermediates. The method exhibits superior tolerance for various functional groups, meaning that protecting group strategies can often be minimized, reducing the number of synthetic steps and associated waste. By understanding the precise interaction between the palladium catalyst and the L,X-type ligand, manufacturers can optimize reaction parameters to maximize the purity of the final beta-substituted ketone. This level of mechanistic control is essential for meeting the stringent quality standards required in the pharmaceutical industry, where impurity profiles must be tightly managed to ensure patient safety and regulatory compliance.
How to Synthesize Beta-Substituted Ketones Efficiently
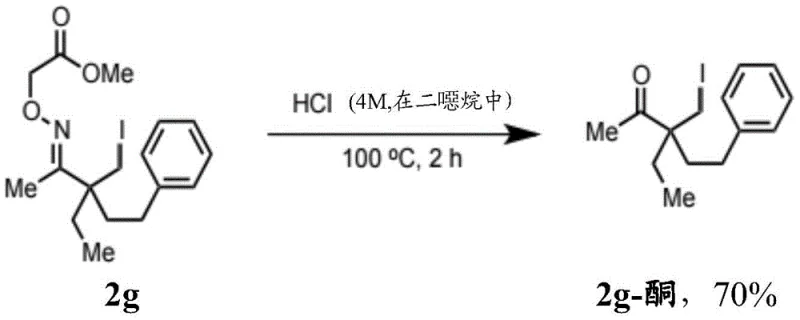
The synthesis of these high-value intermediates follows a logical three-step sequence that is amenable to standard chemical manufacturing equipment. The process begins with the condensation of the ketone substrate with aminooxyacetic acid, followed by the palladium-catalyzed functionalization step, and concludes with the hydrolytic removal of the auxiliary. Detailed operational parameters, including specific molar ratios, temperature profiles, and workup procedures, are critical for achieving the reported yields and selectivity. The following guide outlines the standardized synthesis steps derived from the patent data to ensure reproducibility and safety in a production environment.
- Condense the target ketone with commercially available aminooxyacetic acid in pyridine solvent to form the oxime directing group intermediate without chromatography.
- Perform palladium(II)-catalyzed beta-C(sp3)-H iodination or arylation using iodine or aryl iodides in aprotic solvents like dioxane or HFIP.
- Cleave the oxime auxiliary group under acidic conditions, such as concentrated hydrochloric acid in dioxane, to yield the final beta-substituted ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial benefits that directly address the pain points of procurement and supply chain management in the fine chemical sector. The elimination of chromatography for the installation of the directing group significantly reduces solvent consumption and processing time, leading to a leaner manufacturing process. The use of commercially available aminooxyacetic acid as a promoter ensures a stable and reliable supply of key reagents, mitigating the risk of raw material shortages that can disrupt production schedules. Furthermore, the ability to access sterically hindered quaternary centers opens up new avenues for the synthesis of complex drug candidates, allowing companies to diversify their product portfolios without investing in entirely new infrastructure. The robustness of the reaction conditions implies a lower risk of batch failure, enhancing overall supply chain reliability and consistency. These factors combine to create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves cost optimization by eliminating the need for expensive transition metal removal steps often associated with other catalytic methods, as the auxiliary can be removed via simple acid hydrolysis. The high atom economy and reduced solvent usage due to the lack of chromatographic purification contribute to a lower overall cost of goods sold. By streamlining the synthesis into fewer steps with higher yields, manufacturers can realize substantial savings in labor and utility costs. The use of readily available starting materials further drives down the input costs, making the final beta-substituted ketones more competitive in the global market.
- Enhanced Supply Chain Reliability: Reliance on commercially available reagents like aminooxyacetic acid and standard palladium salts ensures that the supply chain is not dependent on custom-synthesized or scarce materials. The robustness of the reaction against various functional groups reduces the need for multiple protected intermediates, simplifying inventory management and reducing lead times. This stability allows for more accurate forecasting and planning, ensuring that downstream customers receive their orders on time. The scalability of the method from gram to kilogram scales without significant loss in efficiency supports a continuous and reliable supply of critical intermediates for large-scale drug production.
- Scalability and Environmental Compliance: The method is designed with scalability in mind, utilizing solvents and conditions that are manageable in large-scale reactors. The reduction in waste generation, particularly from chromatography media and excessive solvent use, aligns with increasingly stringent environmental regulations and sustainability goals. The ability to perform the reaction in air for certain steps simplifies the engineering requirements for the production facility, lowering capital expenditure. Efficient waste streams and the potential for solvent recovery further enhance the environmental profile of the process, making it an attractive option for manufacturers committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-C-H functionalization technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this process into their existing workflows. The information provided here serves as a foundational reference for further technical discussions and feasibility assessments.
Q: What is the primary advantage of the aminooxyacetic acid directing group over transient directing groups?
A: The aminooxyacetic acid forms a stable oxime bond that acts as an L,X-type directing group, overcoming the incompatibility of free transient amino acids with oxidizing or coupling agents and enabling a broader substrate range.
Q: Can this method access sterically hindered quaternary centers?
A: Yes, the protocol allows for rapid access to various sterically hindered quaternary centers at the beta-position, which are difficult to achieve using classical enolization alkylation processes.
Q: Is chromatography required for the installation of the directing group?
A: No, a key practical advantage of this method is the facile installation of the aminooxyacetic acid additive without the need for chromatography, significantly simplifying the workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Iodoketones Supplier
The technological potential of this palladium-catalyzed beta-C-H functionalization route is immense, offering a strategic advantage for companies looking to innovate their synthetic pathways. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of beta-iodoketones or beta-aryl ketones meets the highest industry standards. We understand the complexities of handling palladium catalysts and optimizing directing group strategies, and our team is ready to translate this patent data into a robust, GMP-compliant manufacturing process for your specific needs.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel synthetic route. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us help you overcome engineering bottlenecks and secure a reliable supply of high-quality intermediates for your next generation of pharmaceutical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
