Advanced Nickel-Catalyzed Chain Walking Reduction for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective methods for synthesizing complex organic molecules, particularly those requiring specific isotopic labeling for metabolic studies. Patent CN115417782A introduces a groundbreaking "chain walking" type olefin transfer hydrogenation and hydrodeuteration reduction method that addresses these critical needs. This innovative technology utilizes a nickel-based catalytic system to achieve regioselective reduction and deuteration of terminal olefins in a single step. By employing water or heavy water as the proton or deuteron source, this method offers a significantly greener alternative to traditional protocols that rely on expensive and hazardous reagents. The process operates under mild conditions, typically ranging from 20°C to 50°C, which ensures compatibility with a wide array of sensitive functional groups commonly found in active pharmaceutical ingredients. This technical breakthrough not only simplifies the synthetic route but also enhances the overall economic viability of producing high-purity pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for olefin hydrogenation and deuteration have long been plagued by significant limitations regarding regioselectivity and reagent costs. In conventional transition metal-catalyzed reactions, the addition of hydrogen or deuterium typically occurs exclusively at the position of the carbon-carbon double bond. This lack of flexibility restricts the ability of chemists to introduce isotopic labels at specific remote positions within the molecular framework, which is often crucial for elucidating reaction mechanisms or studying biosynthetic pathways. Furthermore, existing protocols frequently require the use of alcohols or amines as proton sources, which can be costly, especially when deuterated variants are needed. These traditional approaches often necessitate harsh reaction conditions, including high temperatures and pressures, which can lead to the decomposition of thermally unstable substrates. The reliance on precious metal catalysts in some conventional methods also introduces challenges related to residual metal contamination, requiring extensive and expensive purification steps to meet stringent regulatory standards for pharmaceutical applications.
The Novel Approach
In stark contrast to these legacy techniques, the novel chain walking methodology described in the patent data revolutionizes the field by enabling the migration of the metal center along the alkyl chain. This unique mechanism allows for the precise installation of hydrogen or deuterium atoms at positions distant from the original double bond, providing access to structural motifs that were previously difficult or impossible to synthesize efficiently. The use of water or heavy water as the proton source represents a paradigm shift towards green chemistry, drastically reducing the environmental footprint and raw material costs associated with isotopic labeling. The reaction proceeds smoothly at near-room temperature, eliminating the need for energy-intensive heating or high-pressure equipment. This operational simplicity translates directly into enhanced safety profiles and reduced capital expenditure for manufacturing facilities. By overcoming the regioselectivity barriers of the past, this approach opens new avenues for the design and synthesis of next-generation deuterated drugs and specialized chemical intermediates.
Mechanistic Insights into Nickel-Catalyzed Chain Walking Reduction
The core of this transformative technology lies in the sophisticated interplay between the nickel catalyst, the ligand system, and the hydride source. The reaction initiates with the activation of the nickel precursor, such as nickel bromide or nickel chloride, by the bipyridine ligand to form an active catalytic species. Upon interaction with the reducing agent, typically pinacolborane or triethoxysilane, a nickel-hydride intermediate is generated. This active species undergoes insertion into the terminal olefin, followed by a series of beta-hydride elimination and re-insertion steps that effectively "walk" the metal center along the carbon chain. This chain-walking process continues until the thermodynamically favored position is reached, where the final protonation or deuteration occurs using water or heavy water. The choice of ligand, such as 2,2'-bipyridine or its derivatives, plays a critical role in modulating the electronic and steric environment of the nickel center, thereby controlling the extent of migration and ensuring high regioselectivity. Understanding these mechanistic nuances is essential for optimizing the reaction parameters to achieve maximum yield and purity for diverse substrate classes.
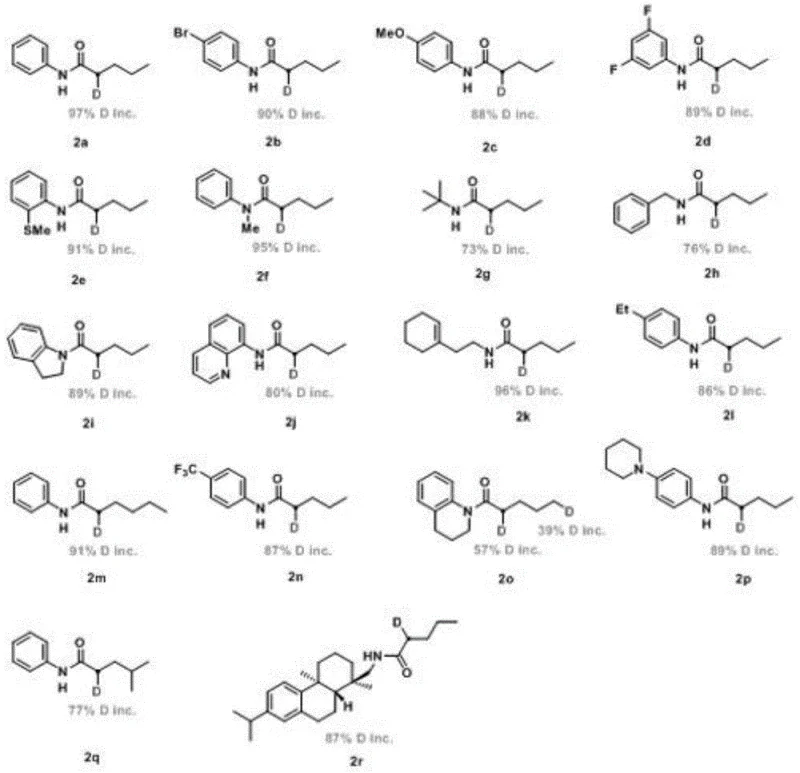
Controlling the impurity profile is paramount in the production of pharmaceutical intermediates, and this nickel-catalyzed system demonstrates exceptional selectivity. The mild reaction conditions inherently suppress side reactions such as polymerization or over-reduction, which are common pitfalls in aggressive hydrogenation processes. The use of water as a quencher and proton source simplifies the workup procedure, allowing for the easy removal of boron byproducts and catalyst residues through standard aqueous extraction. Column chromatography is subsequently employed to isolate the pure product, ensuring that the final material meets the rigorous quality specifications required for downstream applications. The robustness of the catalytic cycle against various functional groups, including halides, ethers, and heterocycles, further underscores the versatility of this method. For R&D teams, this means a reliable platform for generating libraries of deuterated analogs without the need for extensive protecting group strategies or multi-step synthetic sequences, thereby accelerating the drug discovery timeline.
How to Synthesize Deuterated Amides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and atmospheric control to maximize efficiency. The patent outlines a straightforward protocol where the olefin derivative, nickel source, ligand, and reducing agent are combined in a solvent like dichloromethane or tetrahydrofuran. The addition of heavy water initiates the transfer deuteration, and the mixture is heated gently to facilitate the chain walking process. Reaction progress is conveniently monitored using thin-layer chromatography, allowing for precise determination of the endpoint. Once the reaction is complete, a simple aqueous workup followed by column chromatography yields the high-purity deuterated product. The detailed standardized synthesis steps for this process are provided in the guide below to assist technical teams in replicating these results.
- Prepare the reaction mixture by combining the terminal olefin derivative, nickel salt catalyst, bipyridine ligand, and pinacolborane reducing agent in an inert solvent like dichloromethane or THF.
- Introduce heavy water (D2O) or regular water as the proton/deuteron source into the reactor under a nitrogen atmosphere to ensure anhydrous conditions for the catalyst prior to initiation.
- Heat the mixture to mild temperatures between 20°C and 50°C for 10 to 24 hours, monitoring progress via TLC, followed by aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed chain walking technology offers substantial strategic benefits that extend beyond mere technical performance. The shift from precious metal catalysts to abundant nickel salts significantly reduces the raw material cost base, mitigating the financial risks associated with volatile metal markets. Furthermore, the replacement of expensive deuterated organic solvents with commercially available heavy water represents a drastic reduction in input costs for isotope-labeled compounds. The mild operating conditions eliminate the need for specialized high-pressure reactors, allowing for production in standard glass-lined or stainless steel vessels, which enhances asset utilization and flexibility. These factors combine to create a more resilient and cost-efficient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of high-cost reagents with commodity chemicals. By utilizing nickel salts instead of palladium or platinum catalysts, manufacturers can avoid the substantial expenses linked to precious metal recovery and refining. Additionally, the use of water as the proton source removes the dependency on costly deuterated alcohols or amines, which traditionally account for a significant portion of the bill of materials in deuteration projects. The simplified purification workflow, which avoids complex distillation or crystallization steps often required to remove amine byproducts, further lowers processing costs. These cumulative savings contribute to a more competitive pricing structure for the final active pharmaceutical ingredients, enhancing market positioning.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the reliance on widely available and stable raw materials. Nickel salts and bipyridine ligands are commodity chemicals with robust global supply networks, reducing the risk of shortages that can plague specialized reagents. The operational simplicity of the reaction, which does not require high-pressure hydrogen gas, eliminates safety hazards and logistical complexities associated with gas storage and handling. This allows for manufacturing in a broader range of facilities, increasing geographic diversification of supply sources. Consequently, lead times for high-purity pharmaceutical intermediates can be reduced, ensuring consistent availability for downstream drug formulation and clinical trial material production.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles and regulatory expectations. The absence of toxic amine waste streams and the use of water as a benign byproduct simplify wastewater treatment and reduce the environmental burden of the manufacturing process. The mild temperature profile ensures that the reaction can be scaled up from gram to tonnage quantities without encountering significant heat transfer limitations or safety runaway risks. This ease of scale-up facilitates rapid technology transfer from R&D to commercial production, enabling faster time-to-market for new drug candidates. Compliance with increasingly stringent environmental regulations is thus achieved without compromising on yield or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chain walking reduction technology. These insights are derived directly from the patent specifications and are intended to clarify the operational capabilities and limitations of the process for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: What distinguishes this chain walking method from traditional olefin hydrogenation?
A: Unlike traditional methods that only reduce the double bond directly, this nickel-catalyzed chain walking mechanism migrates the metal center along the carbon chain, allowing for remote functionalization and specific isotopic labeling at non-double bond positions.
Q: Are the reaction conditions suitable for sensitive pharmaceutical intermediates?
A: Yes, the process operates under exceptionally mild conditions, typically between 20°C and 50°C, which prevents thermal degradation of sensitive functional groups often found in complex drug molecules.
Q: What represents the primary cost advantage of this synthesis route?
A: The method utilizes commercially available heavy water or distilled water as the proton source instead of expensive deuterated alcohols or amines, significantly lowering raw material costs while simplifying the post-reaction purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olefin Transfer Hydrogenation Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed chain walking technology for the future of pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of nickel catalysis, including rigorous QC labs that enforce stringent purity specifications for every batch. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry, leveraging our deep technical expertise to optimize yield and minimize impurities.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this technology can reduce your overall manufacturing expenses. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving efficiency and innovation in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
