Advanced Visible-Light Catalysis for Scalable Production of High-Purity Benzothiazole Derivatives in Pharmaceutical Manufacturing
Patent CN109053625B, granted on August 11, 2020, introduces a transformative methodology for synthesizing substituted benzothiazole C2 alkylated derivatives through visible-light photocatalysis that fundamentally redefines industry standards for pharmaceutical intermediate production. This innovation represents a critical advancement by completely eliminating the requirement for transition metal catalysts or strong bases that have historically constrained alkylation processes, thereby addressing persistent challenges in purity control and cost efficiency faced by global pharmaceutical manufacturers. The process leverages inexpensive organic dyes such as Eosin Y as photosensitizers coupled with potassium persulfate as an oxidant under ambient conditions, achieving high yields without thermal activation or specialized equipment. This breakthrough not only significantly enhances the environmental profile of the synthesis through reduced energy consumption but also establishes a new paradigm for economic viability in large-scale manufacturing operations. The methodology is particularly crucial for producing key intermediates in drug development pipelines where stringent purity requirements and cost pressures demand innovative solutions that maintain both scientific rigor and commercial practicality. By utilizing readily available LED lighting technology, this patent delivers a scalable platform that directly addresses the evolving needs of multinational pharmaceutical enterprises seeking sustainable production pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to benzothiazole C2 alkylation have been severely constrained by their reliance on transition metal catalysts or strong base systems that introduce multiple operational and economic challenges for pharmaceutical manufacturers. These methods typically require elevated temperatures exceeding 80°C or specialized reaction vessels to handle corrosive reagents, significantly increasing energy consumption and capital expenditure while creating safety hazards in large-scale production environments. The persistent issue of metal contamination necessitates complex and costly purification protocols to meet regulatory standards for pharmaceutical intermediates, often resulting in yield losses exceeding 15% during metal residue removal processes. Furthermore, the scarcity and price volatility of transition metals like palladium or iridium create supply chain vulnerabilities that directly impact production continuity and cost predictability for global pharmaceutical companies. These limitations are particularly acute when synthesizing complex derivatives required for high-value therapeutics where even trace impurities can compromise drug efficacy or safety profiles, forcing manufacturers to implement redundant quality control measures that further erode profit margins in competitive markets.
The Novel Approach
The patented methodology overcomes these critical limitations through an elegant visible-light catalyzed system that operates under ambient conditions using commercially accessible components, representing a paradigm shift in benzothiazole derivative synthesis. By employing organic dyes such as Eosin Y as photosensitizers at concentrations as low as 0.5 mol% alongside potassium persulfate as an oxidant, the process achieves high yields (76%-92%) without requiring transition metals or strong bases that have plagued conventional approaches. The reaction proceeds efficiently at room temperature using standard LED white light sources (15-20 W), eliminating energy-intensive heating/cooling cycles while maintaining excellent selectivity across diverse substrate combinations including various substituted benzothiazoles and N,N-dimethylamide compounds. This innovation directly addresses pharmaceutical manufacturers' dual imperatives of cost reduction and quality assurance by removing metal contamination risks entirely and simplifying purification workflows through inherently cleaner reaction profiles. The methodology's compatibility with standard laboratory equipment enables seamless scale-up from milligram to multi-kilogram quantities without reoptimization, providing an immediate pathway to commercial implementation that delivers both economic and operational advantages.
Mechanistic Insights into Visible-Light Catalyzed Benzothiazole Alkylation
The photoredox mechanism begins with visible-light excitation of Eosin Y (0.5-2.0 mol%), generating an excited state that facilitates single-electron transfer to potassium persulfate, producing sulfate radical anions that initiate radical formation from N,N-dimethylamide compounds. This cascade generates α-amino radicals that selectively attack the electron-deficient C2 position of substituted benzothiazoles through a radical addition pathway, avoiding the regioselectivity issues common in traditional alkylation methods. The catalytic cycle is completed through oxidation-reduction processes that regenerate the photosensitizer while maintaining high turnover numbers, with the entire sequence occurring under mild conditions that prevent decomposition of sensitive functional groups. This mechanism operates with exceptional precision due to the controlled radical generation enabled by visible-light activation, which minimizes competing side reactions that typically plague thermal or base-catalyzed approaches requiring harsher conditions.
Impurity control is significantly enhanced through the elimination of transition metals and strong bases that traditionally generate difficult-to-remove byproducts during benzothiazole alkylation processes. The ambient temperature operation prevents thermal degradation pathways that produce colored impurities or decomposition products requiring additional purification steps in conventional methods. The radical-based mechanism exhibits superior regioselectivity at the C2 position without competing N-alkylation or ring-opening side reactions that commonly occur in base-catalyzed systems, resulting in cleaner reaction profiles with fewer impurities to isolate during column chromatography purification. This inherent selectivity reduces the number of purification cycles needed to achieve pharmaceutical-grade purity standards while minimizing yield losses typically associated with complex impurity removal protocols in traditional manufacturing processes.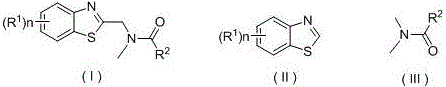
How to Synthesize Benzothiazole C2 Alkylated Derivatives Efficiently
This patented methodology provides a robust framework for synthesizing high-purity benzothiazole C2 alkylated derivatives through a visible-light catalyzed process that eliminates traditional limitations while maintaining excellent yield consistency across diverse substrate combinations. The innovation leverages commercially available components including organic photosensitizers and standard LED lighting systems to create a scalable manufacturing platform suitable for both laboratory development and commercial production environments. By operating under ambient conditions without requiring specialized equipment or hazardous reagents, this approach delivers significant operational advantages while ensuring consistent product quality essential for pharmaceutical applications. Detailed standardized synthesis procedures are provided below to facilitate immediate implementation in manufacturing settings.
- Combine substituted benzothiazole with N,N-dimethylamide solvent under ambient air conditions while maintaining precise stoichiometric ratios between reactants.
- Introduce organic photosensitizer such as Eosin Y and potassium persulfate oxidant before initiating visible-light irradiation using LED white light sources at controlled power settings.
- Monitor reaction progression via thin-layer chromatography until completion, then perform column chromatography purification using petroleum ether/ethyl acetate solvent systems.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by delivering substantial operational improvements that enhance both cost efficiency and supply chain resilience for global manufacturers. The elimination of transition metal catalysts removes a major source of supply chain vulnerability while creating significant cost reduction opportunities through simplified processing requirements and reduced quality control burdens. These advantages are particularly valuable in today's competitive pharmaceutical landscape where procurement teams face increasing pressure to optimize costs without compromising on quality or delivery reliability. The process delivers tangible benefits across multiple dimensions of commercial operations while maintaining full compliance with stringent regulatory requirements for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The complete elimination of expensive transition metal catalysts removes both raw material costs and associated expenses from complex metal residue removal protocols required in traditional processes, significantly reducing overall production costs while avoiding specialized equipment investments typically needed for high-temperature reactions or metal recovery systems. This streamlined approach minimizes waste generation through inherently cleaner reaction profiles, reducing disposal costs and environmental compliance expenses while maintaining high yield consistency across diverse substrate combinations.
- Enhanced Supply Chain Reliability: By utilizing readily available organic dyes and standard chemical reagents instead of scarce transition metals, this methodology ensures consistent raw material availability regardless of geopolitical supply chain disruptions or market fluctuations affecting precious metals. The ambient temperature operation eliminates energy-intensive processing requirements that create production bottlenecks during peak demand periods, enabling more predictable scheduling and delivery timelines while reducing dependency on specialized equipment suppliers that can cause manufacturing delays.
- Scalability and Environmental Compliance: The room-temperature process demonstrates exceptional scalability from laboratory to commercial production without reoptimization requirements, enabling seamless transition from development to manufacturing phases while maintaining consistent product quality attributes essential for regulatory approval. The elimination of hazardous reagents and reduced energy consumption creates significant environmental compliance advantages by minimizing waste streams and lowering carbon footprint compared to conventional high-energy processes, aligning with global sustainability initiatives while reducing regulatory reporting burdens.
Frequently Asked Questions (FAQ)
The following questions address specific technical and commercial considerations based on patent CN109053625B's implementation details, providing clarity on how this innovation solves persistent challenges in pharmaceutical intermediate manufacturing while delivering tangible business value.
Q: How does this visible-light method eliminate transition metal catalysts while maintaining high yields?
A: The process utilizes organic dyes like Eosin Y as photosensitizers that generate reactive radical species through visible-light excitation, replacing transition metals in the catalytic cycle while achieving comparable yields of 76-92% under mild conditions.
Q: What scalability advantages does room-temperature synthesis offer for pharmaceutical intermediates?
A: Ambient temperature operation eliminates energy-intensive heating/cooling requirements and enables direct scale-up from laboratory to commercial production without reoptimization, significantly reducing capital expenditure and operational complexity.
Q: How does the elimination of transition metals impact supply chain reliability?
A: By removing dependence on scarce transition metals, the process ensures consistent raw material availability while avoiding complex purification steps required for metal residue removal, thereby enhancing production continuity and quality assurance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiazole C2 Alkylated Derivatives Supplier
Our patented visible-light catalysis platform represents a significant advancement in sustainable pharmaceutical intermediate manufacturing that directly addresses the evolving needs of global drug developers seeking reliable partners for complex molecule production. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art facilities equipped with rigorous QC labs capable of meeting the most demanding regulatory requirements. Our technical expertise ensures seamless technology transfer from laboratory to manufacturing scale while optimizing processes for maximum cost efficiency without compromising on quality or delivery reliability essential for pharmaceutical supply chains.
We invite procurement teams to request our Customized Cost-Saving Analysis which demonstrates specific economic benefits tailored to your manufacturing requirements through detailed route feasibility assessments and comparative cost modeling against conventional methods. Contact our technical procurement team today to obtain specific COA data and discuss how our innovative synthesis platform can enhance your supply chain resilience while delivering substantial economic advantages in high-purity benzothiazole derivative production.
