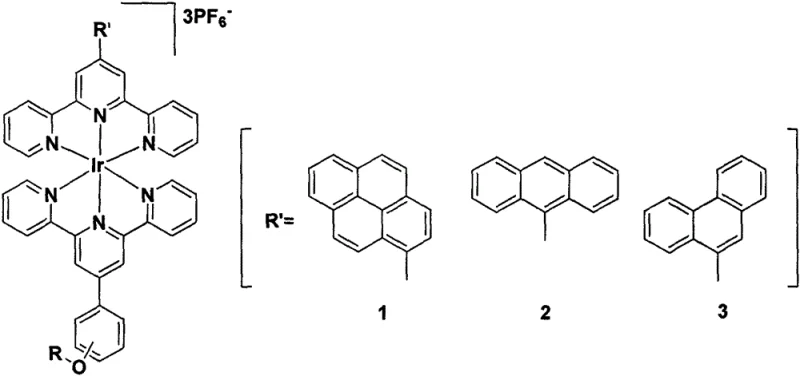
Advanced Bis-Terpyridyl Iridium (III) Complexes for Scalable Photodynamic Therapy Applications
Advanced Bis-Terpyridyl Iridium (III) Complexes for Scalable Photodynamic Therapy Applications
Introduction to Next-Generation Photosensitizer Technology
The landscape of photodynamic therapy (PDT) is undergoing a significant transformation driven by the need for more stable and efficient photosensitizing agents. As detailed in patent CN111662333A, a novel class of bis-terpyridyl iridium (III) complexes has emerged as a promising candidate for overcoming the limitations of traditional porphyrin-based drugs. These complexes are engineered to possess exceptional photophysical properties, including high quantum yields for singlet oxygen generation, which is the cytotoxic species responsible for destroying tumor cells during PDT treatment. The structural versatility of these iridium compounds allows for precise tuning of their emission wavelengths and solubility profiles, making them highly adaptable for various medical applications ranging from oncology to dermatology. By leveraging the heavy atom effect of the iridium center, these molecules facilitate efficient intersystem crossing to the triplet state, thereby maximizing the therapeutic output per unit of light energy administered. This technological breakthrough represents a critical step forward for pharmaceutical developers seeking reliable high-purity pharmaceutical intermediates that can be consistently manufactured at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the first and second generations of photosensitizers, such as hematoporphyrin derivatives and metal phthalocyanines, have been plagued by significant drawbacks that hinder their widespread clinical adoption. A primary concern with these conventional agents is their complex mixture composition, which often leads to inconsistent therapeutic outcomes and difficulties in regulatory approval due to batch-to-batch variability. Furthermore, many traditional photosensitizers suffer from poor water solubility, necessitating the use of toxic solubilizing agents that can introduce additional side effects for patients undergoing treatment. The long clearance times associated with first-generation drugs also result in prolonged skin photosensitivity, forcing patients to avoid sunlight for weeks after administration, which severely impacts their quality of life. Additionally, the synthetic routes for these older compounds often involve harsh conditions and difficult purification steps, leading to higher production costs and supply chain bottlenecks for reliable photosensitizer supplier networks. These cumulative issues create a pressing demand for chemically defined, single-component alternatives that offer improved safety profiles and manufacturing efficiency.
The Novel Approach
The novel synthetic methodology described in the patent data offers a robust solution by utilizing well-defined terpyridine ligands coordinated to a central iridium atom. This approach ensures that the final product is a pure, single chemical entity, eliminating the variability inherent in natural product extracts or complex mixtures. The use of cyclometalation reactions allows for the formation of extremely stable carbon-metal bonds, which significantly enhances the chemical and thermal stability of the complex compared to nitrogen-coordinated analogues. Moreover, the modular nature of the terpyridine ligand synthesis enables chemists to introduce specific functional groups, such as ether chains or polyaromatic systems, to fine-tune the lipophilicity and targeting capabilities of the drug. This level of molecular precision not only improves the pharmacokinetic properties but also simplifies the quality control processes required for commercial scale-up of complex pharmaceutical intermediates. By shifting towards these well-characterized organometallic systems, manufacturers can achieve cost reduction in photodynamic therapy manufacturing through streamlined synthesis and higher overall yields.
Mechanistic Insights into Cyclometalation and Ligand Coordination
The core of this technology lies in the efficient cyclometalation of the iridium center, which is achieved through a carefully controlled reaction with terpyridine derivatives in high-boiling solvents like ethylene glycol. The process typically begins with the condensation of substituted aldehydes and acetylpyridine under basic conditions to form the terpyridine ligand, a step that is critical for establishing the electronic properties of the final complex. Once the ligand is formed, it reacts with iridium trichloride trihydrate at elevated temperatures, typically around 160°C, to form a chloro-bridged dimer intermediate. This intermediate is crucial as it activates the metal center for subsequent ligand exchange reactions, allowing for the introduction of the second terpyridine ligand to complete the octahedral coordination sphere. The heavy atom effect of the iridium promotes strong spin-orbit coupling, which facilitates the population of the triplet excited state necessary for efficient energy transfer to molecular oxygen. Understanding these mechanistic details is vital for R&D teams aiming to optimize the reaction conditions for maximum purity and minimal impurity formation.
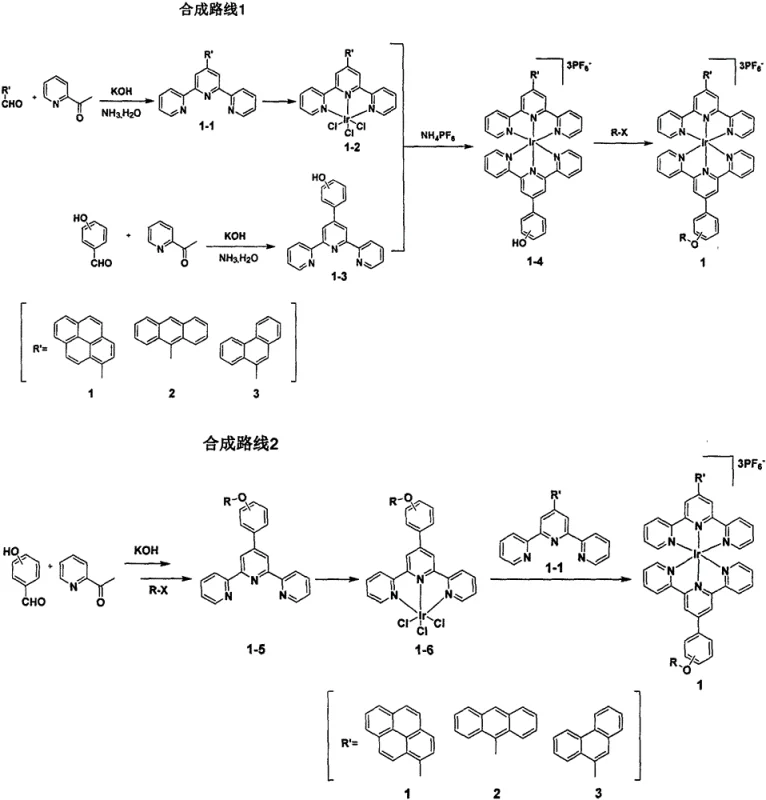
Impurity control is managed through the selection of high-purity starting materials and the implementation of rigorous purification protocols such as column chromatography using neutral aluminum oxide. The patent specifies the use of ammonium hexafluorophosphate to precipitate the final cationic complex, a step that effectively separates the product from unreacted ligands and inorganic salts. The stability of the iridium-carbon bond ensures that the complex remains intact under physiological conditions, preventing the release of toxic free metal ions which is a common concern with other metallodrugs. Furthermore, the ability to modify the R groups on the ligand allows for the steric hindrance of potential degradation pathways, thereby extending the shelf-life of the active pharmaceutical ingredient. This robust chemical architecture ensures that the photosensitizer maintains its efficacy throughout the supply chain, from synthesis to final administration, providing supply chain heads with confidence in the product's integrity.
How to Synthesize Bis-Terpyridyl Iridium (III) Complex Efficiently
The synthesis of these high-value complexes involves a multi-step sequence that begins with the preparation of functionalized terpyridine ligands followed by metal coordination. The process is designed to be scalable, utilizing common laboratory reagents and equipment that can be easily adapted for industrial production environments. Detailed standard operating procedures for each reaction step, including specific temperature profiles and workup techniques, are essential for ensuring reproducibility and high yield. The following guide outlines the critical stages of the synthesis, emphasizing the key parameters that influence the quality of the final photosensitizer. For a complete breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Prepare terpyridine ligands via Kröhnke condensation using aldehydes and acetylpyridine under basic conditions.
- React the ligand with iridium trichloride trihydrate in ethylene glycol at 160°C to form the chloro-bridged dimer intermediate.
- Complete the coordination sphere by reacting the intermediate with a second ligand and precipitating with ammonium hexafluorophosphate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-based synthesis route offers substantial benefits for procurement and supply chain management teams looking to optimize their sourcing strategies. The use of readily available starting materials, such as substituted benzaldehydes and acetylpyridine, reduces the risk of raw material shortages and price volatility that often plague the supply of exotic chemical intermediates. The synthetic pathway avoids the use of expensive transition metal catalysts that require complex removal steps, thereby significantly simplifying the downstream processing and reducing the overall cost of goods sold. Additionally, the high stability of the final product minimizes losses during storage and transportation, ensuring that the delivered material meets stringent purity specifications without the need for special handling conditions. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined synthesis process eliminates the need for costly purification steps associated with traditional porphyrin extraction, leading to significant operational savings. By avoiding the use of expensive noble metal catalysts in the ligand formation step and utilizing efficient precipitation methods for isolation, the overall production cost is drastically reduced. This economic efficiency allows for more competitive pricing structures without compromising on the quality or purity of the final active ingredient. Furthermore, the high yield of the cyclometalation reaction ensures that raw material utilization is maximized, further driving down the cost per kilogram of the produced photosensitizer.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the synthesis of the terpyridine ligands ensures a stable and continuous supply of raw materials, mitigating the risk of production delays. The robustness of the iridium complex also means that it can withstand standard shipping conditions without degradation, reducing the need for expensive cold chain logistics. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of large pharmaceutical partners. By partnering with a reliable photosensitizer supplier who utilizes this technology, companies can secure a long-term source of high-quality materials for their drug development pipelines.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, using solvents like ethanol and ethylene glycol which are easier to recover and recycle compared to chlorinated solvents. This aligns with modern environmental regulations and reduces the burden of waste disposal, making the process more sustainable and compliant with green chemistry principles. The ability to scale from gram to kilogram quantities without significant changes to the reaction parameters ensures that the technology can meet the growing demand for photodynamic therapy agents. This scalability supports the commercial scale-up of complex pharmaceutical intermediates, enabling rapid transition from clinical trials to market availability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bis-terpyridyl iridium complexes. The answers are derived from the specific technical advantages and synthesis details outlined in the patent documentation, providing clarity for potential partners. Understanding these aspects is key to evaluating the feasibility of integrating this technology into existing manufacturing workflows. For more detailed technical data, our team is available to provide specific route feasibility assessments.
Q: What are the stability advantages of these iridium complexes compared to traditional porphyrins?
A: These octahedral iridium (III) complexes exhibit superior stability in biological media and air compared to first-generation porphyrin derivatives, reducing degradation during storage and handling.
Q: How does the ligand modification affect the solubility for pharmaceutical formulation?
A: The introduction of ether chains or specific aromatic groups on the terpyridine ligand significantly enhances water solubility and bioavailability, facilitating easier formulation into injectable dosage forms.
Q: Is the synthesis process scalable for commercial production of photosensitizers?
A: Yes, the synthetic route utilizes common solvents like ethylene glycol and ethanol, and standard purification techniques like column chromatography, making it highly adaptable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Terpyridyl Iridium (III) Complex Supplier
The development of bis-terpyridyl iridium (III) complexes represents a significant advancement in the field of photodynamic therapy, offering a blend of high efficacy and manufacturing practicality. NINGBO INNO PHARMCHEM, as a leading CDMO expert, possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be translated into a reliable commercial supply. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of photosensitizer meets the highest international standards for pharmaceutical use. We understand the critical nature of supply continuity for drug development and are committed to providing a stable source of these advanced materials.
We invite you to discuss how our manufacturing capabilities can support your specific project needs and help you achieve a Customized Cost-Saving Analysis for your supply chain. Our technical procurement team is ready to assist you in evaluating the potential of this technology for your applications. Please contact us to request specific COA data and route feasibility assessments to begin your partnership with a trusted leader in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
