Optimizing Steroid Intermediate Production via Advanced Homogeneous Phase Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical steroid intermediates, particularly for the synthesis of Dehydroepiandrosterone Acetate, a pivotal precursor for various hormonal therapies. A significant technological advancement in this domain is detailed in patent CN102746356A, which introduces a novel homogeneous phase method that fundamentally alters the reaction kinetics and safety profile of the production process. This innovation addresses long-standing inefficiencies in the traditional synthesis route, specifically targeting the oximation and rearrangement stages which have historically been bottlenecks for yield and purity. By shifting from heterogeneous systems to a fully dissolved homogeneous state, the process ensures that reactants interact at a molecular level without the diffusion limitations imposed by solid precipitates. This technical report analyzes the mechanistic superiority of this approach and its profound implications for commercial scale-up, offering a compelling value proposition for R&D directors and supply chain managers alike who prioritize consistency and regulatory compliance in their API intermediate sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
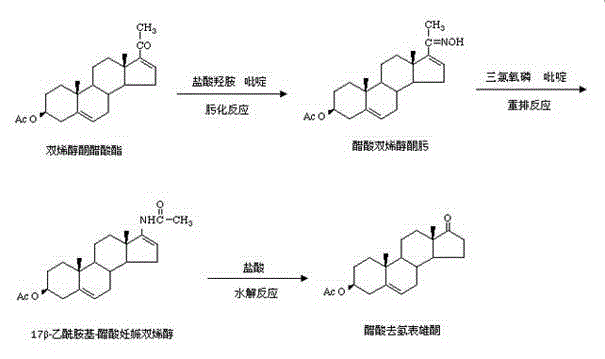
Historically, the industrial production of Dehydroepiandrosterone Acetate has relied on legacy protocols that utilize ethanol as the primary solvent for the oximation reaction. In these traditional setups, the solubility of both the starting material, Dehydropregnenolone acetate, and the resulting oxime intermediate is notoriously poor in ethanol. This physical limitation forces the reaction to proceed in a heterogeneous state, characterized by a thick, semi-solid slurry where solid particles of the substrate are often encapsulated by the product layer. Consequently, the hydroxylamine hydrochloride cannot effectively penetrate these aggregates to react with the unconverted ketone, leading to incomplete conversions even after extended reaction times of up to five hours. Furthermore, the subsequent rearrangement step in conventional methods frequently employs purified petroleum benzine, a solvent now recognized for its severe carcinogenicity and stringent residual limits, posing significant occupational health hazards and complicating waste disposal protocols for modern manufacturing facilities aiming for green chemistry standards.
The Novel Approach
The patented homogeneous phase method revolutionizes this workflow by introducing a binary solvent system comprising methanol and chloroform for the oximation stage. This specific solvent combination is engineered to fully dissolve the steroid substrate and the oxime product simultaneously, creating a clear, single-phase liquid reaction medium. This transition from a solid-liquid heterogeneous mixture to a true solution eliminates mass transfer resistance, allowing the oximation to reach completion rapidly within just three hours while achieving product contents exceeding 99%. In the rearrangement phase, the process replaces hazardous petroleum benzine with a safer mixture of toluene and chloroform. This substitution not only mitigates toxicological risks but also leverages the azeotropic properties of the new solvent system to facilitate easier removal and recovery. The result is a streamlined process that boosts the overall yield to approximately 85% and ensures the final refined product meets rigorous purity specifications of 99.5% or higher, setting a new benchmark for quality in steroid intermediate manufacturing.
Mechanistic Insights into Homogeneous Phase Oximation and Rearrangement
The core chemical advantage of this technology lies in the manipulation of solubility parameters to drive reaction equilibrium. In the oximation step, the use of a methanol-chloroform mixture (in a weight ratio ranging from 2:1 to 1.3) creates an environment where the polarity is balanced to solvate both the organic steroid backbone and the inorganic hydroxylamine salt. Unlike the ethanol system where the product precipitates prematurely and coats the reactant, the homogeneous solution allows for continuous collision between the carbonyl group of the ketone and the nucleophilic nitrogen of the hydroxylamine. This ensures that the formation of the oxime proceeds to near-total conversion, evidenced by the melting point elevation from 198°C in crude traditional products to 202°C in the new process, indicating a much tighter impurity profile. The absence of solid barriers means that reaction kinetics are governed purely by chemical potential rather than diffusion rates, drastically reducing the energy input required for agitation and heating.
Furthermore, the rearrangement mechanism utilizing phosphorus oxychloride (POCl3) is critically dependent on thermal management to prevent degradation. The patent highlights a sophisticated temperature control strategy where the dropping temperature is maintained lower than the holding temperature. Specifically, during the addition of the POCl3-toluene mixture, the temperature is kept between 4°C and 8°C to manage the intense exotherm generated by the reaction of POCl3 with trace moisture. However, once the addition is complete, the temperature is allowed to rise to a holding range of 9°C to 12°C. This deliberate gradient is essential because the thermometer reading represents the bulk liquid temperature, which lags behind the localized hot spots at the滴加点 (dropping point). By keeping the bulk cooler during addition and warmer during incubation, the process avoids 'burning' the sensitive steroid skeleton while providing sufficient thermal energy to drive the Beckmann rearrangement and subsequent hydrolysis to completion, thereby minimizing the formation of high-boiling impurities that are difficult to remove in later purification stages.
How to Synthesize Dehydroepiandrosterone Acetate Efficiently
The synthesis of this high-value steroid intermediate requires precise adherence to the homogeneous phase protocol to replicate the reported yields and purity levels. The process begins with the dissolution of Dehydropregnenolone acetate in the methanol-chloroform solvent system, followed by the controlled addition of pyridine and hydroxylamine hydrochloride under reflux conditions. Following the isolation of the oxime intermediate, the material is subjected to the rearrangement reaction in a toluene-chloroform medium, where strict temperature zoning is applied during the addition of phosphorus oxychloride and subsequent hydrolysis with hydrochloric acid. The detailed operational parameters, including specific flow rates and quenching procedures, are critical for maintaining the integrity of the steroid nucleus. For a comprehensive, step-by-step technical guide on executing this synthesis in a pilot or production environment, please refer to the standardized protocol below.
- Perform oximation reaction using a methanol and chloroform mixed solvent system to ensure complete dissolution and homogeneous reaction conditions.
- Execute the Beckmann rearrangement using phosphorus oxychloride in a toluene and chloroform solvent mixture with precise temperature gradient control.
- Conduct hydrolysis and subsequent purification steps including steam distillation and recrystallization to obtain refined product with content exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this homogeneous phase technology translates directly into tangible operational efficiencies and risk mitigation. The shift away from carcinogenic solvents like petroleum benzine significantly reduces the regulatory burden associated with solvent residue testing and environmental compliance, streamlining the audit process for pharmaceutical customers. Moreover, the improved solubility characteristics of the new solvent system allow for a substantial increase in reactor loading capacity; whereas traditional methods might be limited by the viscosity of the slurry, the liquid nature of the homogeneous reaction enables manufacturers to process larger batches in the same vessel volume. This enhancement in volumetric efficiency means that production throughput can be scaled up without the immediate need for capital expenditure on new reactor infrastructure, offering a faster route to meeting surging market demand for steroid precursors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in solvent consumption and the increase in batch yield. By optimizing the solvent-to-substrate ratio from 5:1 in traditional methods to 3:1 in the new process, the volume of solvent required for dissolution and washing is drastically cut. This reduction lowers the direct cost of raw materials and significantly decreases the energy load required for solvent recovery and distillation. Additionally, the yield improvement from roughly 79% to 85% means that less starting material is wasted per kilogram of final product, effectively lowering the cost of goods sold (COGS) and enhancing the margin profile for large-scale production runs without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of purification steps required to remove stubborn impurities generated in inefficient reactions. The high selectivity of the homogeneous phase method results in a crude product with significantly fewer side products, simplifying the downstream purification workflow. This robustness reduces the likelihood of batch failures or the need for re-processing, which are common causes of delivery delays in fine chemical manufacturing. By stabilizing the reaction outcome through better thermal control and solubility management, suppliers can offer more predictable lead times and consistent inventory availability, which is crucial for downstream API manufacturers planning their own production schedules.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the replacement of Class 1 solvents with Class 2 solvents represents a major strategic advantage. Facilities utilizing this method face lower costs related to hazardous waste disposal and worker protection equipment, as the toxicity profile of toluene and chloroform is far more manageable than that of petroleum benzine. This alignment with modern green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner. The simplified solvent recovery system, aided by the azeotropic behavior of the toluene-chloroform mixture, further supports sustainable operations by maximizing solvent recycling rates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on how this method outperforms legacy technologies in terms of both chemical efficiency and operational safety. Understanding these nuances is essential for technical teams evaluating potential process transfers or vendor qualifications.
Q: How does the homogeneous phase method improve yield compared to traditional ethanol-based processes?
A: The homogeneous phase method utilizes a methanol-chloroform solvent system that fully dissolves both the substrate and the oxime product, eliminating the heterogeneous solid-liquid barriers found in ethanol processes. This ensures complete contact between reactants, increasing the overall yield from approximately 75-79% to 85% and improving product content to over 99.5%.
Q: What are the safety advantages of replacing petroleum benzine with toluene and chloroform?
A: Traditional methods often rely on purified petroleum benzine, which is classified as a carcinogenic solvent with strict residual limits. The novel process substitutes this with toluene and chloroform, which are Class 2 solvents with significantly lower toxicity profiles, thereby reducing regulatory compliance risks and improving workplace safety standards.
Q: Why is the temperature gradient critical during the phosphorus oxychloride addition step?
A: The reaction between phosphorus oxychloride and moisture is highly exothermic. The patented process controls the dropping temperature at 4-8°C but maintains a higher holding temperature of 9-12°C. This gradient prevents local overheating that could cause side reactions while ensuring sufficient thermal energy for the rearrangement to proceed efficiently after the initial addition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroepiandrosterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like the homogeneous phase method requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Dehydroepiandrosterone Acetate meets the exacting standards required for hormone synthesis. Our commitment to quality assurance ensures that the high purity and yield reported in the patent are consistently delivered to our global clientele.
We invite pharmaceutical companies and chemical distributors to explore how this optimized process can enhance their supply chain resilience. By leveraging our technical proficiency, you can secure a stable source of high-quality intermediates that aligns with modern safety and efficiency benchmarks. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
