Industrial Synthesis of 2-Deoxy-Beta-Adenosine: A Breakthrough in Scalable Nucleoside Manufacturing
Introduction to Advanced Nucleoside Synthesis Technology
The pharmaceutical and biotechnology sectors are constantly seeking robust, scalable pathways for producing critical DNA building blocks, and the synthesis method detailed in patent CN100537589C represents a significant leap forward in this domain. This technology specifically addresses the production of 2-deoxy-beta-adenosine, a fundamental unit of DNA that is indispensable for antiviral research and genetic engineering applications. Historically, the industrial manufacture of this nucleoside has been plagued by low stereoselectivity and cumbersome purification requirements, which severely limit supply chain reliability. By leveraging a novel protective group strategy and optimized coupling conditions, this patent outlines a route that bypasses the traditional bottlenecks of isomer separation. For R&D directors and procurement specialists alike, understanding this methodology is crucial, as it offers a viable path toward a reliable pharmaceutical intermediate supplier status by ensuring consistent quality and reduced operational complexity. The transition from laboratory curiosity to industrial reality hinges on such process innovations that prioritize both chemical efficiency and economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the landscape of 2-deoxy-beta-adenosine synthesis was dominated by methods that were fundamentally unsuited for large-scale commercial operations. One prominent historical approach involved the direct condensation of adenine with 2-deoxy-D-ribose, which notoriously resulted in an equimolar mixture of alpha and beta isomers. This lack of stereocontrol necessitated extensive chromatographic separation, a technique that is prohibitively expensive and time-consuming when scaling from grams to tons. Another reported method utilized 1,3-dichloro-1,1,3,3-tetraisopropyldisiloxane (TPDS-Cl2) to protect hydroxyl groups, followed by deoxygenation using toxic tin compounds. The reliance on hazardous reagents not only escalated safety risks but also introduced severe environmental compliance challenges regarding waste disposal. Furthermore, other literature methods involving trimethylsilyl protection still suffered from poor selectivity ratios, often yielding beta-to-alpha ratios of only 9:1 at best, with overall yields before deprotection hovering around 43% to 46%. These inefficiencies created a significant barrier to entry for cost reduction in pharmaceutical intermediates manufacturing, as the cumulative loss of material and solvent rendered the processes economically unviable for mass production.
The Novel Approach
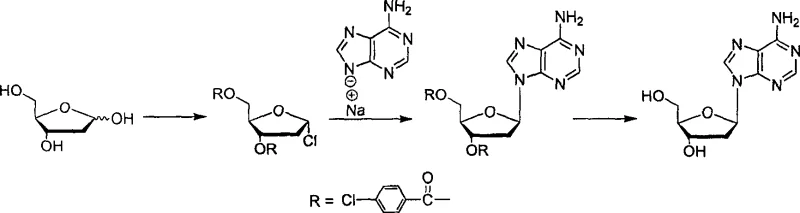
In stark contrast to these legacy techniques, the method disclosed in CN100537589C introduces a streamlined, high-efficiency pathway that eliminates the need for chromatographic purification entirely. The core innovation lies in the strategic use of p-chlorobenzoyl groups to protect the sugar moiety, which not only stabilizes the intermediate but also sterically directs the subsequent glycosylation towards the desired beta-configuration. By converting adenine into its sodium salt prior to coupling, the nucleophilicity is enhanced, allowing the reaction to proceed under milder conditions in a mixed solvent system of butanone and ethyl acetate. This approach ensures that the crude product obtained after the coupling step is already enriched in the target isomer, allowing for direct progression to the deprotection stage without intermediate isolation. The final deprotection using a methanol-ammonia solution is clean and efficient, yielding a white crystalline product with a sharp melting point of 183.5°C. This holistic redesign of the synthetic route transforms the production of high-purity 2-deoxy-beta-adenosine from a complex, multi-step purification challenge into a straightforward, linear process suitable for commercial scale-up of complex nucleoside analogs.
Mechanistic Insights into Stereoselective Glycosylation
The success of this synthesis relies heavily on the precise mechanistic interplay between the protected sugar donor and the adenine acceptor. The initial protection of 2-deoxy-D-ribose with p-chlorobenzoyl chloride in pyridine creates a 1-chloro-2-deoxy-3,5-O-bis-(chlorobenzoyl)-ribose intermediate. The electron-withdrawing nature of the chlorobenzoyl groups plays a dual role: it stabilizes the anomeric center against unwanted hydrolysis and exerts a steric influence that disfavors the formation of the alpha-anomer during the coupling phase. When the adenine sodium salt, generated by treating adenine with sodium hydroxide in anhydrous methanol, is introduced into the reaction mixture, the N9 nitrogen acts as a potent nucleophile. The reaction proceeds via an SN2-like displacement mechanism where the chloride leaving group is displaced by the adenine anion. The choice of solvent is critical here; the mixture of butanone and ethyl acetate provides the optimal polarity to solubilize both the organic sugar derivative and the ionic adenine salt, facilitating intimate molecular contact without promoting side reactions. This careful balancing of electronic and steric factors ensures that the glycosidic bond forms with high fidelity, locking the nucleobase into the biologically active beta-orientation.
Furthermore, the impurity profile of this process is inherently managed by the chemistry itself, rather than by downstream purification. In traditional methods, the presence of alpha-isomers and unreacted starting materials often requires repeated recrystallizations or column chromatography, which drastically reduces overall yield. In this novel route, the byproducts formed during the coupling step, such as unreacted adenine salts or hydrolyzed sugar derivatives, are effectively removed during the aqueous workup and washing stages. The use of dilute sulfuric acid and sodium bicarbonate washes helps to neutralize any residual acidic or basic species, while the final ammonolysis step cleaves the ester protecting groups cleanly. The resulting crude solid can be recrystallized directly from water to achieve pharmaceutical-grade purity. This mechanism of 'built-in' purification is a key advantage for reducing lead time for high-purity nucleosides, as it minimizes the number of unit operations required between the reactor and the final packaging line, thereby enhancing throughput and consistency.
How to Synthesize 2-Deoxy-Beta-Adenosine Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters outlined in the patent to ensure maximum yield and stereoselectivity. The process begins with the preparation of the chloro-sugar intermediate, where temperature control during the addition of p-chlorobenzoyl chloride is vital to prevent degradation. Following this, the generation of the adenine sodium salt must be performed under anhydrous conditions to avoid premature hydrolysis of the sensitive chloro-sugar. The coupling reaction is notably robust, tolerating a range of stirring times, but optimal results are achieved by allowing the mixture to stir at room temperature for an extended period to ensure complete conversion. Finally, the deprotection step utilizes ammonia gas saturated in methanol, a reagent that is both effective and easily removable, leaving no toxic residues. For a detailed breakdown of the specific masses, volumes, and timing for each stage, please refer to the standardized protocol below.
- Protect 2-deoxy-D-ribose with p-chlorobenzoyl chloride in pyridine to form 1-chloro-2-deoxy-3,5-O-bis-(chlorobenzoyl)-ribose.
- Generate adenine sodium salt by reacting adenine with sodium hydroxide in anhydrous methanol under reflux conditions.
- Couple the adenine sodium salt with the protected chloro-sugar in a butanone and ethyl acetate solvent system at room temperature.
- Perform deprotection using a saturated ammonia methanol solution at low temperature followed by recrystallization to obtain the pure beta-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity of supply. The most immediate impact is seen in the drastic simplification of the manufacturing workflow. By eliminating the need for column chromatography, the process removes one of the most resource-intensive steps in fine chemical production. Chromatography typically consumes vast quantities of silica gel and organic solvents, creating a significant burden on waste management systems and increasing the cost of goods sold. Removing this step not only lowers direct material costs but also accelerates the production cycle, allowing for faster turnover of batches and improved responsiveness to market demand. Additionally, the avoidance of toxic tin compounds means that the facility does not need to invest in specialized heavy metal removal equipment or deal with the regulatory complexities associated with hazardous waste disposal. This translates to substantial cost savings and a reduced environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the high atom economy and the reduction in purification overhead. Since the intermediate products do not require isolation and purification before the next step, the overall processing time is significantly shortened. The reagents used, such as p-chlorobenzoyl chloride and common solvents like butanone and ethyl acetate, are commodity chemicals that are readily available at competitive prices. This accessibility ensures that the raw material costs remain stable and predictable, shielding the production budget from the volatility often seen with exotic catalysts or specialized reagents. Furthermore, the high yield of the final deprotection step, reported to be around 88% to 94%, ensures that the value added in the earlier stages is not lost at the finish line, maximizing the return on investment for every kilogram of starting material processed.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reaction conditions. The process operates at moderate temperatures, mostly between 25°C and 40°C, which reduces the energy load on the manufacturing plant and minimizes the risk of thermal runaway incidents. The starting materials, including 2-deoxy-D-ribose and adenine, are well-established commodities with multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base allows procurement teams to negotiate better terms and secure long-term contracts with confidence. Moreover, the stability of the intermediates, particularly when stored under appropriate conditions, allows for flexible scheduling of production runs, enabling the manufacturer to build up inventory buffers during periods of low demand to meet sudden spikes in orders without delay.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to full commercial production is straightforward due to the absence of complex physical separation techniques. The workup procedures involve standard liquid-liquid extractions and filtrations, which are easily replicated in large-scale reactors equipped with standard agitation and separation capabilities. The environmental profile of the process is favorable, as the primary waste streams consist of aqueous salts and recoverable organic solvents, which can be treated using conventional wastewater treatment facilities. This ease of compliance with environmental regulations reduces the administrative burden on the EHS (Environment, Health, and Safety) team and minimizes the risk of production stoppages due to regulatory non-compliance. The ability to produce high volumes of 2-deoxy-beta-adenosine with a consistent quality profile makes this method an ideal candidate for meeting the growing global demand for nucleoside-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of the method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing lines or for procurement officers assessing the quality standards of potential suppliers. The focus is on resolving uncertainties regarding purity, safety, and operational efficiency to facilitate informed decision-making.
Q: Why is this synthesis method superior to traditional direct condensation?
A: Traditional direct condensation of adenine and deoxyribose typically yields a 1:1 mixture of alpha and beta isomers, requiring difficult and costly chromatographic separation. This patented method utilizes specific p-chlorobenzoyl protecting groups and adenine sodium salts to achieve high stereoselectivity for the beta-isomer, completely eliminating the need for column chromatography.
Q: Does this process involve toxic heavy metal catalysts?
A: No, unlike some prior art methods that rely on toxic tin compounds for deoxygenation or coupling, this process utilizes standard organic reagents such as p-chlorobenzoyl chloride and sodium hydroxide. This significantly reduces environmental hazards and simplifies waste treatment protocols for large-scale manufacturing.
Q: What is the expected purity and yield of the final product?
A: According to the patent data, the final deprotection and recrystallization steps yield pure 2-deoxy-beta-adenosine with a melting point of 183.5°C. The process avoids intermediate purification steps, streamlining the workflow while maintaining high stereochemical integrity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Deoxy-Beta-Adenosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality nucleoside intermediates play in the development of next-generation antiviral and oncology therapies. Our technical team has extensively analyzed the synthetic route described in CN100537589C and possesses the expertise to implement this chemistry with precision and scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the stereochemical integrity of every batch. We understand that in the pharmaceutical industry, there is no margin for error, which is why our quality assurance protocols are designed to exceed international standards, providing you with a partner you can trust for long-term collaboration.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis method to your specific volume and purity requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies further opportunities to optimize your supply chain economics. We encourage you to request specific COA data and route feasibility assessments to validate our capabilities against your project milestones. Whether you are in the early stages of drug discovery or preparing for commercial launch, our commitment to technical excellence and customer support ensures that your project remains on track and within budget.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
