Advanced Air-Catalytic Oxidation for Commercial Scale Production of 3,4-Dihydroxybenzaldehyde
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with economic viability, particularly for critical building blocks like 3,4-Dihydroxybenzaldehyde. Patent CN101676253B introduces a transformative approach to synthesizing this valuable intermediate through an air catalytic oxidation method, marking a significant departure from traditional stoichiometric oxidation processes. This technology leverages a multi-component composite metal oxide catalyst to facilitate the conversion of 3,4-dihydroxymandelic acid into the target aldehyde using atmospheric oxygen. For R&D Directors and Procurement Managers alike, this represents a pivotal shift towards greener chemistry that does not compromise on yield or scalability. By replacing expensive and wasteful oxidants with air, the process inherently lowers the cost of goods sold while simplifying the impurity profile, making it an ideal candidate for reliable pharmaceutical intermediates supplier partnerships aiming for long-term sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-Dihydroxybenzaldehyde has been plagued by inefficient methodologies that impose heavy burdens on both production costs and environmental compliance. Conventional routes often involve the bromination of p-hydroxybenzaldehyde followed by hydrolysis, a sequence that necessitates the handling of hazardous halogens and generates substantial acidic waste streams requiring neutralization. Alternatively, earlier oxidative decarboxylation methods relied heavily on stoichiometric amounts of compound copper salts. While effective in the laboratory, these copper-based systems present severe challenges during commercial scale-up of complex pharmaceutical intermediates due to the difficulty in recovering the metal salts. The accumulation of copper residues in the final product necessitates rigorous and costly purification steps to meet stringent purity specifications required by downstream drug manufacturers. Furthermore, the consumption of stoichiometric oxidants results in poor atom economy, directly inflating the raw material expenditure and creating a supply chain vulnerability dependent on fluctuating metal prices.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a heterogeneous multi-component metal oxide catalyst system that operates efficiently with air as the terminal oxidant. This method fundamentally alters the economic equation by eliminating the need for stoichiometric chemical oxidants, thereby achieving significant cost reduction in pharmaceutical intermediates manufacturing. The catalyst, composed of elements such as Aluminum, Copper, Molybdenum, Manganese, Iron, Zinc, and Sodium, is designed for stability and reusability, allowing it to be recovered and recycled multiple times without significant loss of activity. This shift not only reduces the direct material costs but also drastically simplifies the work-up procedure, as there is no massive influx of reduced metal byproducts to separate. The process operates under mild alkaline conditions at temperatures between 85-95°C, ensuring safety and ease of operation in standard stainless steel reactors, which enhances the overall reliability of the supply chain for high-purity pharmaceutical intermediates.
Mechanistic Insights into Air-Catalytic Oxidative Decarboxylation
The core of this technological advancement lies in the sophisticated interaction between the multi-component metal oxide surface and the 3,4-dihydroxymandelic acid substrate. The catalytic cycle initiates with the adsorption of the substrate onto the active sites of the metal oxide lattice, where the electron transfer properties of the transition metals (such as Copper and Manganese) facilitate the activation of molecular oxygen from the air stream. This activated oxygen species then promotes the oxidative decarboxylation of the mandelic acid moiety, effectively cleaving the carbon-carbon bond to release carbon dioxide and form the aldehyde functionality. The presence of multiple metal components creates a synergistic effect that stabilizes the transition states and prevents the over-oxidation of the sensitive catechol ring, a common side reaction that leads to tar formation and yield loss. By maintaining the reaction pH in a weakly alkaline range, the solubility of the intermediate is optimized while ensuring the catalyst surface remains active, resulting in a clean conversion pathway that maximizes the formation of the desired 3,4-Dihydroxybenzaldehyde.
Impurity control is another critical aspect where this mechanism excels, addressing the primary concerns of R&D Directors regarding product quality. Traditional methods often struggle with residual heavy metals and halogenated byproducts that are difficult to purge to ppm levels. In this air oxidation system, the primary byproduct is water and carbon dioxide, alongside the unreacted starting material which can be easily recycled. The heterogeneous nature of the catalyst means that metal leaching is minimized, ensuring that the final product meets rigorous QC standards without requiring extensive chelation treatments. The selectivity of the catalyst ensures that the hydroxyl groups on the benzene ring remain intact, preserving the chemical integrity required for subsequent coupling reactions in antibiotic or vasoactive drug synthesis. This high level of selectivity contributes to a narrower impurity spectrum, facilitating faster regulatory approval and reducing the risk of batch rejection.
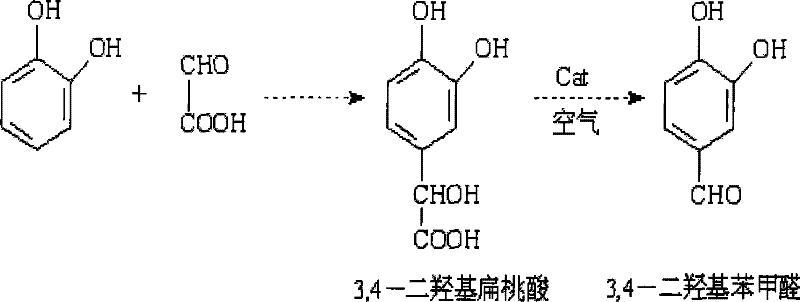
How to Synthesize 3,4-Dihydroxybenzaldehyde Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters established in the patent to ensure reproducibility and maximum yield. The process begins with the preparation of the precursor, 3,4-dihydroxymandelic acid, via the condensation of catechol and glyoxylic acid, which must be purified and dried before entering the oxidation stage. Once dissolved in water and adjusted to the correct pH, the addition of the specific multi-component catalyst initiates the reaction upon heating and aeration. The detailed standardized synthesis steps, including specific mixing rates and cooling protocols, are critical for maintaining the catalyst's structural integrity and ensuring consistent batch-to-batch performance. For technical teams looking to adopt this methodology, following the precise operational guidelines is essential to replicate the high yields reported in the intellectual property documentation.
- Condense catechol and glyoxylic acid under alkaline conditions to form 3,4-dihydroxymandelic acid, then separate and dry the pure product.
- Dissolve the dried acid in water, adjust pH to weakly alkaline, and add the multi-component composite metal oxide catalyst.
- Introduce air at 80-110 L/h and heat to 85-95°C with stirring to complete the oxidative decarboxylation reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the adoption of this air-catalytic oxidation technology translates into tangible strategic advantages that extend beyond simple unit price negotiations. The elimination of stoichiometric oxidants and the ability to recycle the catalyst significantly reduce the variable costs associated with production, allowing for more competitive pricing structures in long-term contracts. Moreover, the reliance on air as a reagent removes the logistical complexities and safety hazards associated with transporting and storing hazardous chemical oxidants, thereby enhancing supply chain resilience. This process stability ensures reducing lead time for high-purity pharmaceutical intermediates, as production schedules are less likely to be disrupted by raw material shortages or regulatory hurdles related to hazardous waste disposal. The scalability of the reaction allows manufacturers to respond rapidly to market demand surges without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive chemical oxidants with free atmospheric air and the implementation of a reusable catalyst system. By avoiding the purchase of stoichiometric amounts of copper salts or other heavy metal oxidants, the direct material cost per kilogram of product is drastically lowered. Additionally, the simplified downstream processing reduces the consumption of solvents, energy, and labor hours required for purification and waste treatment. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, offering a more attractive total cost of ownership for buyers seeking a reliable pharmaceutical intermediates supplier.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of raw material markets, particularly for specialized reagents. This synthesis route mitigates such risks by utilizing commodity chemicals like catechol and glyoxylic acid, which are widely available from multiple global sources. The robustness of the catalyst further ensures that production capacity is not limited by the availability of niche reagents. Consequently, manufacturers can maintain consistent inventory levels and honor delivery commitments even during periods of market instability, providing buyers with the confidence needed for just-in-time manufacturing strategies and long-term project planning.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the ability to scale production without generating excessive hazardous waste is a critical competitive advantage. This air oxidation method produces minimal toxic waste, primarily consisting of aqueous streams that are easier to treat compared to the heavy metal sludge generated by traditional methods. This environmental compatibility facilitates smoother permitting processes for plant expansions and reduces the liability associated with waste disposal. The process is inherently safe and scalable from pilot plants to multi-ton commercial reactors, ensuring that the commercial scale-up of complex pharmaceutical intermediates can proceed without encountering unforeseen engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this air-catalytic oxidation technology. These insights are derived directly from the patent data and practical considerations for industrial application, providing clarity for stakeholders evaluating this synthesis route. Understanding these details is crucial for making informed decisions about integrating this method into existing production portfolios or sourcing strategies.
Q: What are the primary advantages of using air oxidation over traditional copper salt methods?
A: Traditional methods often rely on stoichiometric amounts of compound copper salts which are difficult to recover and generate significant heavy metal waste. The air catalytic oxidation method described in CN101676253B utilizes atmospheric oxygen as the oxidant and a reusable multi-component metal oxide catalyst, drastically reducing raw material costs and environmental disposal burdens while simplifying the post-reaction purification process.
Q: What yields can be expected from this catalytic process?
A: According to the experimental data within the patent, the process consistently achieves yields ranging from 72.3% to 90.7%. The yield optimization depends on precise control of the catalyst-to-substrate molar ratio (0.02-0.05), temperature maintenance between 85-95°C, and regulated air flow rates, ensuring high efficiency suitable for industrial application.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the methodology is explicitly designed for scalability. The use of air as a cheap and abundant oxidant, combined with a catalyst that is easy to recover and reuse, addresses key economic bottlenecks. Furthermore, the reaction conditions avoid extreme pressures or hazardous reagents, making the commercial scale-up of complex pharmaceutical intermediates safer and more economically viable for supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydroxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. Our technical team has extensively analyzed the potential of the air-catalytic oxidation method described in CN101676253B and possesses the expertise to implement this technology at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to bulk supply is seamless and robust. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3,4-Dihydroxybenzaldehyde meets the exacting standards required for API synthesis and fine chemical applications.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through advanced process chemistry. By leveraging our capabilities, you can secure a stable source of high-quality intermediates while benefiting from the economic advantages of this green oxidation technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our production capabilities align with your project goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
