Advanced Steroid Synthesis: Scalable Manufacturing of High-Purity Gestodene Intermediates
Advanced Steroid Synthesis: Scalable Manufacturing of High-Purity Gestodene Intermediates
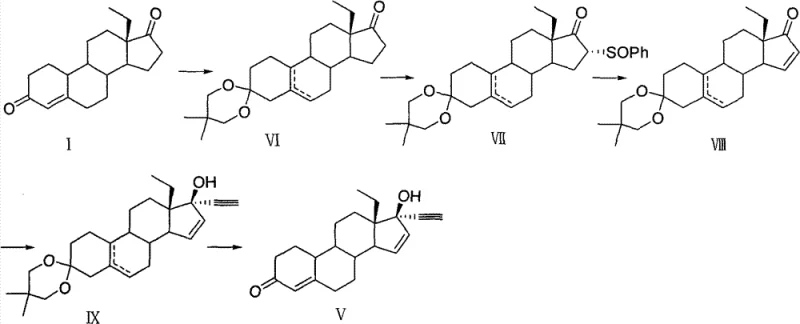
The pharmaceutical landscape for contraceptive agents continues to evolve, driven by the demand for higher purity intermediates and more sustainable manufacturing processes. Patent CN103319558A introduces a significant breakthrough in the synthesis of gestodene, a third-generation progestogen widely utilized in oral contraceptives due to its potent activity and lack of androgenic side effects. This technical disclosure outlines a novel pathway that fundamentally alters the handling of critical steroid intermediates, specifically targeting the C16 position modification. By shifting away from traditional ketal protection strategies that result in intractable oily mixtures, this new methodology enables the isolation of stable, crystalline solids through simple filtration. For R&D directors and process chemists, this represents a pivotal shift from laboratory-scale curiosity to robust industrial feasibility, ensuring that the supply chain for this high-value API remains uninterrupted and cost-effective.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gestodene has relied heavily on routes disclosed in earlier patents such as EP1586579, which utilize ethyl diketone compounds as starting materials. A critical bottleneck in these conventional pathways involves the protection of the C3 carbonyl group as a ketal, followed by substitution at the C16 position. The resulting 16-benzenesulfinyl betamethasone ketal structures are notoriously difficult to manage, often manifesting as oily mixtures that resist crystallization. This physical property necessitates the use of column chromatography for purification, a technique that is economically prohibitive and operationally cumbersome on a multi-kilogram or tonne scale. Furthermore, the removal of the ketal protecting group under acidic conditions in the final stages often leads to diminished yields and the formation of complex impurity profiles, creating significant challenges for quality control teams aiming to meet stringent pharmacopeial standards.
The Novel Approach
In stark contrast to the legacy methods, the innovative process described in CN103319558A employs a 3-ethoxy protection strategy rather than a ketal, fundamentally changing the physicochemical properties of the intermediates. The core of this advancement lies in the generation of Compound III, a 16-phenylsulfinyl derivative that precipitates as a distinct yellow solid upon quenching. This phase change from oil to solid is transformative for process engineering, as it allows for separation via simple elutriation or filtration, completely bypassing the need for silica gel chromatography. The subsequent elimination step to form the 15(16)-double bond is equally optimized, utilizing mild basic conditions in refluxing toluene to achieve high conversion rates. This streamlined approach not only simplifies the operational workflow but also enhances the overall mass balance of the synthesis, making it an ideal candidate for commercial scale-up in GMP environments.
Mechanistic Insights into Phenylsulfinyl Introduction and Elimination
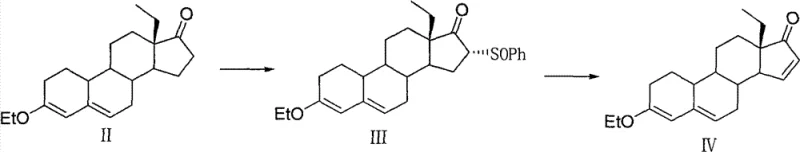
The chemical elegance of this new route is anchored in the selective functionalization of the steroid backbone. The process begins with the etherification of the starting ethyl diketone to form Compound II, establishing a stable 3-ethoxy group that withstands the subsequent reaction conditions better than acid-labile ketals. The introduction of the phenylsulfinyl group at the C16 position is achieved through a nucleophilic substitution mechanism facilitated by strong bases such as potassium tert-butoxide. The choice of solvent, preferably tetrahydrofuran (THF), plays a crucial role in solubilizing the reactants while maintaining the temperature window between 15°C and 30°C, which is critical for controlling regioselectivity and preventing side reactions at other sensitive sites on the steroid nucleus.
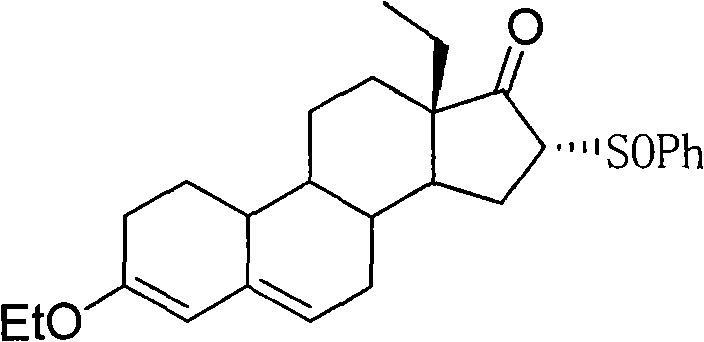
Following the successful installation of the sulfinyl moiety, the pathway proceeds to an elimination reaction designed to construct the critical 15(16)-double bond found in gestodene. This transformation is executed using organic bases like triethylamine in aromatic solvents such as toluene. The mechanism likely involves an E2-type elimination where the base abstracts a proton, leading to the expulsion of the phenylsulfinyl leaving group. The robustness of this step is evidenced by the high molar yields reported, often exceeding 80%, and the ability to drive the reaction to completion by incremental addition of the base. This mechanistic reliability ensures that the final intermediate, Compound IV, is produced with high stereochemical integrity, minimizing the burden on downstream purification processes and ensuring a clean substrate for the final ethynylation step.
How to Synthesize 16-Phenylsulfinyl Steroid Intermediate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the new solid-state intermediate. Operators must adhere to strict temperature protocols during the base-mediated substitution to ensure the formation of the desired yellow solid without degradation. The workup procedure is notably simplified, relying on aqueous quenching and filtration rather than complex extraction sequences. For detailed standard operating procedures regarding reagent grades, stirring rates, and specific drying protocols, please refer to the standardized synthesis guide below which outlines the step-by-step technical requirements for reproducible manufacturing.
- React 3-ethoxy-13β-ethyl-pregna-3,5-dien-17-one with methyl benzenesulfinate and a strong base like potassium tert-butoxide in THF at 15-30°C to obtain the solid 16-phenylsulfinyl intermediate.
- Isolate the resulting yellow solid intermediate via simple elutriation or filtration, avoiding complex chromatographic separation techniques.
- Perform elimination reaction using triethylamine in refluxing toluene to generate the 15(16)-double bond compound, ready for subsequent ethynylation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel synthetic route offers profound advantages for supply chain resilience and cost management. The primary value driver is the elimination of column chromatography, a unit operation that is both capital-intensive and a major bottleneck in batch processing. By replacing this with simple filtration, manufacturers can significantly reduce cycle times and solvent consumption, leading to a leaner production footprint. This efficiency translates directly into improved throughput capabilities, allowing suppliers to respond more agilely to fluctuations in market demand for gestodene and its derivatives without the need for massive infrastructure investments.
- Cost Reduction in Manufacturing: The shift from oily intermediates requiring chromatographic purification to crystalline solids that can be filtered represents a substantial reduction in processing costs. Chromatography consumes vast quantities of silica gel and organic solvents, both of which carry high procurement and disposal costs. By removing this step, the process inherently lowers the variable cost per kilogram of the intermediate. Furthermore, the higher yields observed in the deprotection and elimination steps mean that less starting material is wasted, optimizing the raw material utilization rate and enhancing the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: The robustness of the solid intermediate improves supply continuity by reducing the risk of batch failures associated with difficult separations. Oily mixtures are prone to emulsions and losses during workup, which can lead to unpredictable delivery schedules. In contrast, the predictable crystallization behavior of Compound III ensures consistent batch-to-batch quality and quantity. This reliability is critical for long-term supply agreements with pharmaceutical companies, where consistency is paramount. Additionally, the use of common, commercially available reagents like triethylamine and toluene mitigates the risk of raw material shortages.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste generation, but this route inherently minimizes environmental impact by reducing solvent load. The absence of silica gel waste simplifies waste stream management and lowers the burden on wastewater treatment facilities. The ability to run the elimination reaction at reflux in toluene is a well-understood unit operation in chemical engineering, facilitating seamless technology transfer from pilot plants to large-scale reactors. This ease of scale-up ensures that the supply of high-purity intermediates can grow in tandem with the commercial success of the final drug product, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this steroid synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the operational advantages and chemical specifics of the new method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the new 16-phenylsulfinyl intermediate superior to previous ketal-protected versions?
A: Unlike previous ketal-protected intermediates which formed difficult-to-separate oily mixtures requiring column chromatography, the new 16-phenylsulfinyl etherate exists as a distinct yellow solid. This physical state allows for simple separation via filtration or elutriation, drastically improving suitability for industrial-scale production.
Q: What are the specific reaction conditions for the phenylsulfinyl introduction step?
A: The reaction is conducted under mild conditions, typically between 15°C and 30°C. It utilizes organic solvents such as tetrahydrofuran (THF) or dichloromethane, with potassium tert-butoxide serving as the preferred base to facilitate the substitution at the C16 position efficiently.
Q: How does this process impact the overall yield of gestodene precursors?
A: The process demonstrates high molar yields, with the substitution step achieving approximately 65-70% and the subsequent elimination step reaching up to 88%. The elimination of difficult purification steps like column chromatography further preserves material throughput compared to traditional acidic deprotection methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gestodene Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process validation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel steroid synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence ensures that the gestodene intermediates we supply meet the exacting standards required by global regulatory bodies, providing our partners with peace of mind and a competitive edge in the marketplace.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By leveraging our optimized manufacturing protocols, we can help you achieve significant efficiencies in your supply chain. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities align with your strategic sourcing objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
