Advanced Lewis Acid Catalyzed Cyclization for High-Purity Dextromethorphan Impurity D Standard
Advanced Lewis Acid Catalyzed Cyclization for High-Purity Dextromethorphan Impurity D Standard
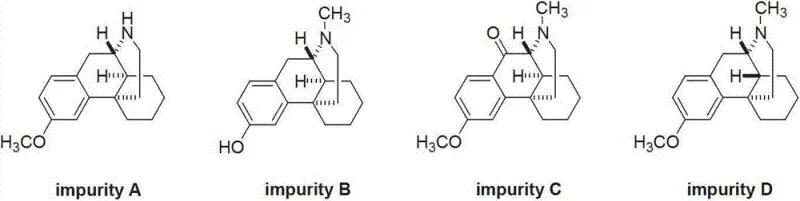
In the rigorous landscape of pharmaceutical quality control, the availability of precise impurity reference standards is paramount for ensuring patient safety and regulatory compliance. Patent CN103265489A introduces a robust and highly selective preparation method for ent-(14S)-3-methoxy-17-methylmorphinan, a critical diastereomeric impurity associated with Dextromethorphan Hydrobromide. This specific compound, often referred to as Impurity D in pharmacopeial standards, presents unique stereochemical challenges that traditional bulk synthesis routes fail to address efficiently. By leveraging a specialized Lewis acid-catalyzed cyclization strategy, this technology enables the direct construction of the 14S stereocenter, bypassing the arduous separation processes typically required to isolate trace diastereomers. For R&D directors and procurement specialists alike, mastering this synthesis pathway represents a significant leap forward in securing reliable supply chains for high-purity analytical standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of specific stereoisomeric impurities like ent-(14S)-3-methoxy-17-methylmorphinan has been fraught with inefficiency and unpredictability. In standard industrial syntheses of Dextromethorphan, the reaction conditions are optimized to favor the thermodynamically stable 14R configuration, rendering the 14S isomer merely a minor byproduct. Consequently, obtaining sufficient quantities of this impurity for validation studies usually relies on the laborious isolation from mother liquors or degradation products, a process that yields negligible amounts and often results in co-eluting contaminants. Furthermore, the structural similarity between the target impurity and the main API makes chromatographic separation extremely resource-intensive, requiring preparative HPLC columns that drive up costs and extend lead times significantly. This dependency on waste stream isolation creates a bottleneck, making the supply of certified reference materials volatile and inconsistent.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this paradigm by shifting from isolation to direct, stereoselective synthesis. Instead of fighting against the thermodynamic preference of the bulk drug synthesis, this approach utilizes a specifically designed precursor, (+)-N-methyl-1-(4-methoxyl) benzyl-1,2,3,4,5,6,7,8-octahydro isoquinoline, which is primed for cyclization into the desired 14S configuration. By employing a Lewis acid catalyst under controlled thermal conditions, the reaction pathway is directed to close the ring with the specific spatial orientation required for Impurity D. This "designer synthesis" approach ensures that the target molecule is the primary product of the reaction rather than a trace contaminant. As a result, manufacturers can achieve high purity levels (>98%) in a single synthetic sequence, drastically simplifying the downstream purification workflow and ensuring a consistent, scalable source of this critical quality control standard.
Mechanistic Insights into Lewis Acid-Catalyzed Intramolecular Cyclization
The core of this synthetic breakthrough lies in the precise manipulation of electronic and steric factors during the ring-closing step. The reaction proceeds via a Lewis acid-mediated intramolecular Friedel-Crafts alkylation mechanism. When a strong Lewis acid, such as aluminum chloride (AlCl3), is introduced to the reaction mixture, it coordinates with the electron-rich centers of the precursor molecule, effectively activating the benzylic position for nucleophilic attack. This activation lowers the energy barrier for the formation of the new carbon-carbon bond that closes the morphinan skeleton. Crucially, the stereochemical outcome is dictated by the conformational constraints of the octahydroisoquinoline ring system in the transition state. The bulky N-methyl group and the existing chiral centers guide the approaching aromatic ring to attack from the face that results in the 14S configuration, demonstrating excellent diastereocontrol inherent to the substrate's geometry.
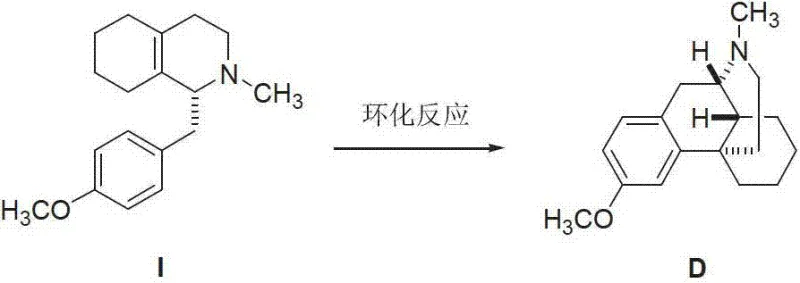
Furthermore, the choice of solvent and temperature plays a pivotal role in maintaining the integrity of the stereocenter while maximizing conversion. The patent specifies the use of chlorinated solvents like methylene dichloride, which provide a polar environment suitable for stabilizing the cationic intermediates generated by the Lewis acid without participating in side reactions. Operating at a moderate temperature of 40°C strikes a delicate balance; it provides sufficient thermal energy to overcome the activation barrier for cyclization while preventing the epimerization of the sensitive C14 center that might occur at higher temperatures. This mechanistic understanding allows process chemists to fine-tune the reaction parameters, ensuring that the formation of other potential impurities (such as the 14R isomer or over-alkylated byproducts) is minimized, thereby delivering a product profile that matches the stringent requirements of pharmacopeial reference standards.
How to Synthesize Ent-(14S)-3-Methoxy-17-Methylmorphinan Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized protocol to ensure reproducibility and safety. The process begins with the preparation of an anhydrous reaction environment, as the Lewis acid catalyst is highly moisture-sensitive. The precursor is dissolved in the selected solvent, and the catalyst is added in a controlled manner to manage the exotherm. Following the reaction period, the workup involves careful quenching and extraction to remove aluminum salts, followed by concentration to yield the crude oil. While the patent outlines a specific embodiment, scaling this process requires attention to mixing efficiency and heat transfer to maintain the selectivity observed on the gram scale. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Dissolve the precursor (+)-N-methyl-1-(4-methoxyl) benzyl-1,2,3,4,5,6,7,8-octahydro isoquinoline in an anhydrous chlorinated solvent such as methylene dichloride under inert atmosphere.
- Add the Lewis acid catalyst, preferably aluminum chloride, in batches at low temperature (0°C) to control the exotherm and initiate the cyclization reaction.
- Warm the reaction mixture to 40°C and stir vigorously for approximately 5 hours, monitoring progress via TLC until the starting material is fully consumed.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this direct synthesis route offers profound strategic advantages over traditional isolation methods. The primary benefit is the decoupling of impurity standard production from the main API manufacturing schedule. Previously, the availability of Impurity D was tethered to the production volume of Dextromethorphan, creating supply risks during periods of low API demand. By establishing an independent synthetic line, companies can guarantee continuous availability of this critical QC material regardless of market fluctuations in the parent drug. Additionally, the simplified purification process reduces the reliance on expensive preparative chromatography resins and solvents, leading to a leaner cost structure. This efficiency translates into more competitive pricing for reference standards and reduces the overall cost of quality control operations for pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The elimination of complex separation technologies significantly lowers the operational expenditure associated with producing high-purity standards. Traditional methods often require multiple rounds of recrystallization or preparative HPLC to achieve the necessary purity, consuming vast amounts of solvents and stationary phases. In contrast, this Lewis acid-catalyzed route yields the target compound with high intrinsic purity, minimizing downstream processing steps. The use of commodity chemicals like aluminum chloride and common solvents further drives down raw material costs, making the production of this niche intermediate economically viable on a commercial scale without the need for exotic reagents.
- Enhanced Supply Chain Reliability: Establishing a dedicated synthesis pathway mitigates the risk of supply interruptions caused by variations in bulk drug production. Since the precursor can be synthesized or sourced independently, the production of the impurity standard becomes a predictable, standalone operation. This reliability is crucial for pharmaceutical companies that must maintain validated analytical methods; running out of a reference standard can halt quality release testing for the entire API batch. By securing a robust supply of ent-(14S)-3-methoxy-17-methylmorphinan through this method, supply chain leaders can ensure uninterrupted QA/QC workflows and avoid costly production delays.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor equipment and straightforward workup procedures that translate well from benchtop to multi-kilogram production. The reaction conditions are mild, avoiding extreme pressures or temperatures that would require specialized containment. Moreover, the streamlined nature of the synthesis reduces the overall solvent footprint and waste generation compared to multi-step isolation protocols. This aligns with modern green chemistry initiatives, allowing manufacturers to meet increasingly stringent environmental regulations while maintaining high throughput. The simplicity of the filtration and washing steps also facilitates easier handling of waste streams, further enhancing the sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific morphinan derivative. Understanding these details is essential for integrating this material into your quality control framework effectively. The answers are derived directly from the technical specifications and performance data outlined in the patent literature, ensuring accuracy and relevance for industry professionals.
Q: Why is synthesizing Ent-(14S)-3-Methoxy-17-Methylmorphinan challenging?
A: This compound is a diastereomer of Dextromethorphan with opposite configuration at the C14 position. Conventional synthesis favors the 14R configuration, making the 14S isomer a trace impurity that is difficult to isolate in high purity without a dedicated stereoselective route.
Q: What catalyst is preferred for this cyclization?
A: While various Lewis acids like FeCl3 or ZnCl2 can be used, Aluminum Chloride (AlCl3) is preferred in this specific protocol due to its ability to drive the intramolecular Friedel-Crafts alkylation efficiently at moderate temperatures, yielding high purity product.
Q: How does this method improve supply chain reliability for QC standards?
A: By synthesizing the impurity directly from a specific precursor rather than isolating it from bulk drug waste streams, manufacturers can guarantee consistent availability and purity of the reference standard, decoupling supply from the fluctuations of main API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ent-(14S)-3-Methoxy-17-Methylmorphinan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final pharmaceutical product depends on the precision of your analytical standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demands whether you require gram-scale samples for method development or kilogram quantities for routine QC. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in complex organic synthesis allows us to navigate the stereochemical nuances of morphinan derivatives, guaranteeing a product that is fully consistent with international pharmacopeial requirements.
We invite you to collaborate with us to optimize your supply chain for critical reference standards. By leveraging our advanced synthetic capabilities, you can secure a stable source of high-quality intermediates that support your regulatory filings and product releases. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can add value to your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
