Advanced Manufacturing of 5-Methoxy-2-Methyltryptamine for Global Pharmaceutical Supply Chains
Advanced Manufacturing of 5-Methoxy-2-Methyltryptamine for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing bioactive scaffolds, particularly those targeting the central nervous system. Patent CN110746340A introduces a highly efficient synthetic methodology for 5-methoxy-2-methyltryptamine, a critical intermediate with significant potential in drug discovery. This compound serves as a versatile building block for developing novel therapeutics targeting 5-HT receptors, which are pivotal in treating mental health disorders. The structural versatility of this scaffold allows for further derivatization into complex bioactive molecules, including glucocorticoid receptor modulators and other heterocyclic systems. By leveraging this patented technology, manufacturers can secure a reliable supply of high-purity intermediates essential for advancing clinical candidates from early research to commercial viability.
The strategic value of this synthesis lies in its ability to produce a key precursor that links to a broader family of pharmacologically active compounds. As illustrated in the structural analysis, the core indole motif found in 5-methoxy-2-methyltryptamine is a foundational element for generating diverse chemical entities. These derivatives exhibit strong binding affinities to specific biological receptors, making them indispensable for the development of next-generation medications for conditions such as rheumatoid arthritis and bronchial asthma. Consequently, establishing a stable manufacturing process for this intermediate is not merely a chemical exercise but a strategic supply chain imperative for pharmaceutical companies aiming to innovate in the CNS and anti-inflammatory therapeutic spaces.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional routes for synthesizing substituted tryptamines often suffer from significant drawbacks that hinder industrial adoption. Many classical methods rely on harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents that complicate safety protocols and waste management. Furthermore, conventional alkylation strategies frequently result in poor regioselectivity, leading to difficult-to-separate isomeric impurities that compromise the purity profile required for pharmaceutical applications. The reliance on expensive catalysts or multi-step protection-deprotection sequences also inflates the cost of goods sold (COGS), making these routes economically unviable for large-scale production. Additionally, low overall yields in older methodologies necessitate larger reactor volumes and increased solvent consumption, thereby exacerbating environmental burdens and reducing process efficiency.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN110746340A offers a streamlined two-step process that addresses these historical inefficiencies. The novel approach utilizes a Henry reaction (nitroaldol condensation) followed by a direct reduction, eliminating the need for complex protecting groups. This route operates under relatively mild thermal conditions, specifically heating to 60°C for the condensation and 70°C for the reduction, which significantly lowers energy consumption compared to high-temperature alternatives. The use of common solvents like n-hexane and tetrahydrofuran simplifies the supply chain logistics, as these are standard inventory items in most chemical manufacturing facilities. Moreover, the process achieves exceptional yields, with the intermediate formation exceeding 95% and the final reduction step surpassing 90%, demonstrating a level of efficiency that drastically reduces raw material waste.
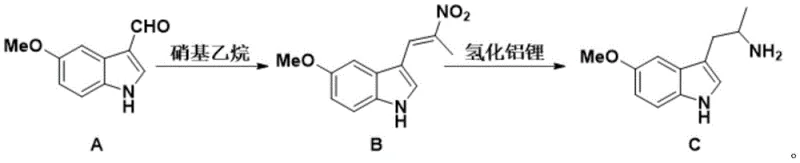
Mechanistic Insights into Nitroaldol Condensation and Hydride Reduction
The first stage of this synthesis involves the condensation of 5-methoxyindole-3-formaldehyde with nitroethane in the presence of ammonium acetate. This reaction proceeds through a nitroaldol mechanism where the acidic alpha-protons of nitroethane are deprotonated to form a nucleophilic nitronate species. This nucleophile attacks the electrophilic carbonyl carbon of the indole aldehyde, followed by dehydration to yield the conjugated nitroalkene intermediate. The choice of ammonium acetate as a mild base catalyst is crucial, as it facilitates the reaction without promoting side reactions such as polymerization of the aldehyde or degradation of the sensitive indole ring. The resulting orange solid intermediate is obtained with high purity, indicating that the reaction kinetics favor the formation of the desired E-isomer, which is essential for the subsequent reduction step.
The second stage employs lithium aluminum hydride (LiAlH4) to effect the simultaneous reduction of both the nitro group and the carbon-carbon double bond. This powerful reducing agent transfers hydride ions to the electron-deficient beta-carbon of the nitroalkene and the nitrogen atom of the nitro group. The mechanism likely involves the initial formation of an aluminate complex, followed by hydrolytic workup to release the free amine. Conducting this reaction in anhydrous tetrahydrofuran ensures the stability of the hydride reagent, while the controlled temperature ramp from 0°C to 70°C manages the exothermic nature of the reduction. The quenching procedure using saturated sodium hydroxide and sodium sulfate is specifically designed to safely decompose excess hydride and facilitate the separation of aluminum salts, resulting in a clean organic phase that requires minimal purification to achieve the final 99% purity specification.
How to Synthesize 5-Methoxy-2-Methyltryptamine Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to ensure reproducibility and safety. The process begins with the preparation of the nitroalkene intermediate under an inert nitrogen atmosphere to prevent oxidation of the sensitive indole moiety. Following the isolation of the intermediate, the reduction step must be performed with careful attention to moisture exclusion until the quenching phase. The detailed operational protocol involves precise stoichiometric ratios and temperature controls that have been optimized to maximize throughput while maintaining safety standards. For a comprehensive understanding of the specific equipment requirements and safety handling procedures for lithium aluminum hydride, operators should refer to the standardized guidelines provided below.
- Condense 5-methoxyindole-3-formaldehyde with nitroethane using ammonium acetate at 60°C to form the nitroalkene intermediate.
- Reduce the intermediate using lithium aluminum hydride in anhydrous THF, heating to 70°C to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages driven by the accessibility and cost-effectiveness of the starting materials. The primary raw material, 5-methoxyindole-3-formaldehyde, is a widely available commodity chemical, reducing the risk of supply bottlenecks that often plague specialized intermediate manufacturing. Furthermore, the auxiliary reagents such as nitroethane and ammonium acetate are inexpensive bulk chemicals, contributing to a significantly lower raw material cost base compared to routes requiring precious metal catalysts or exotic reagents. The high yields reported in the patent translate directly into improved material efficiency, meaning less feedstock is required to produce a kilogram of the final product, which inherently drives down the variable cost of production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex purification steps results in a leaner manufacturing process. By avoiding the need for chromatographic purification and relying instead on simple crystallization and extraction techniques, the operational expenditure associated with solvent recovery and waste disposal is drastically minimized. The high atom economy of the Henry reaction ensures that the majority of the input mass is incorporated into the product, reducing the cost per unit of output. Additionally, the mild reaction conditions reduce energy costs associated with heating and cooling, further enhancing the economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: The reliance on standard solvents like n-hexane and ethyl acetate ensures that the supply chain is resilient to disruptions, as these materials are stocked by numerous global suppliers. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring specialized high-pressure or cryogenic setups, allows for flexible manufacturing across different geographic locations. This flexibility mitigates the risk of single-source dependency and enables pharmaceutical companies to diversify their supplier base, ensuring continuous availability of this critical intermediate for their drug development pipelines.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of the reaction on a kilogram scale in the patent examples. The straightforward workup procedures, involving filtration and liquid-liquid extraction, are easily adaptable to large reactor vessels without significant engineering challenges. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with residual metal limits in pharmaceutical products. The high purity of the final product also minimizes the generation of hazardous waste streams associated with extensive purification efforts, aligning with modern green chemistry principles and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-methoxy-2-methyltryptamine. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing synthesis workflows. The clarity provided here aims to facilitate informed decision-making regarding procurement and process development strategies.
Q: What is the overall yield of the patented synthesis route?
A: The patent reports a yield of ≥95% for the intermediate formation and ≥90% for the final reduction step, ensuring high material efficiency.
Q: Are the raw materials commercially available for large-scale production?
A: Yes, the process utilizes 5-methoxyindole-3-formaldehyde and nitroethane, which are inexpensive and readily accessible commodity chemicals.
Q: How does this method improve upon traditional tryptamine synthesis?
A: It avoids complex multi-step protections and utilizes mild conditions (60-70°C) with simple workup procedures like filtration and extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methoxy-2-Methyltryptamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical innovation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material and commercial manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our capability to replicate and optimize the patented synthesis of 5-methoxy-2-methyltryptamine allows us to offer a consistent and reliable supply to our global partners.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our solutions are perfectly aligned with your quality and timeline expectations for bringing new therapies to market.
