Advanced One-Pot Synthesis of Pyraflufen-ethyl: Technical Breakthroughs and Commercial Scalability
Advanced One-Pot Synthesis of Pyraflufen-ethyl: Technical Breakthroughs and Commercial Scalability
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates, with Pyraflufen-ethyl standing out as a critical molecule for modern weed control. A recent technical disclosure, specifically patent CN113831333A, introduces a groundbreaking synthesis method that addresses long-standing inefficiencies in the production of this sulfuryl pyraflufen derivative. This novel approach leverages a streamlined one-pot reaction strategy that integrates hydroxymethylation, etherification, and condensation steps, fundamentally altering the economic and technical landscape for manufacturers. By minimizing intermediate isolation and optimizing reaction conditions, this method offers a robust pathway for producing high-purity herbicide intermediates with enhanced process safety. For industry stakeholders, understanding the nuances of this patent is essential for evaluating future supply chain strategies and technological partnerships. The structural integrity and functional efficacy of the final product are paramount, as illustrated by the core chemical architecture below.
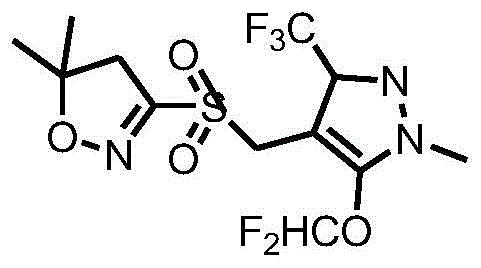
As a reliable agrochemical intermediate supplier, it is crucial to recognize that the adoption of such advanced synthetic routes can significantly impact the availability and cost structure of downstream herbicide formulations. The technical breakthroughs detailed in this patent not only improve chemical yield but also align with broader industry goals of sustainability and operational efficiency. This report provides a deep dive into the mechanistic advantages, commercial implications, and scalability potential of this new synthesis method, offering actionable insights for R&D directors, procurement managers, and supply chain heads alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
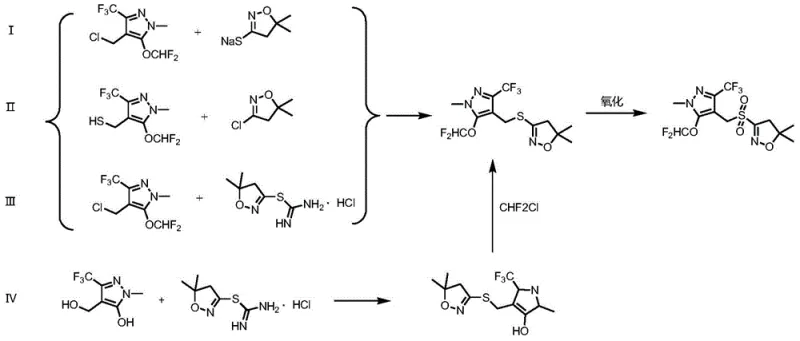
Historically, the synthesis of Pyraflufen-ethyl has been plagued by complex multi-step processes that incur substantial operational costs and material losses. Existing literature and prior art, such as routes described in patents WO2004013106 and WO2011063842, typically involve the separate preparation of key intermediates like sodium sulfide or mercaptan derivatives, which necessitates additional halogenation and substitution steps. These conventional pathways often suffer from low overall yields, with some reported total yields hovering around 64 percent due to cumulative losses at each isolation stage. Furthermore, the reliance on multiple reactor setups and extensive purification procedures increases the consumption of solvents and energy, thereby inflating the environmental footprint of the manufacturing process. The need to handle hazardous intermediates separately also introduces significant safety risks and logistical challenges in a commercial plant setting. Consequently, these inefficiencies create bottlenecks that hinder the ability to meet growing market demand competitively.
The Novel Approach
In stark contrast to the fragmented nature of traditional methods, the novel approach disclosed in CN113831333A consolidates the initial synthetic stages into a cohesive one-pot operation. This method initiates with the hydroxymethylation of 1-methyl-3-trifluoromethyl-5-hydroxy-1H-pyrazole, followed immediately by etherification with difluorochloromethane within the same reaction vessel. By eliminating the need to isolate the hydroxypyrazole methanol intermediate, the process drastically reduces solvent usage and handling time, leading to a more streamlined workflow. The subsequent condensation with isoxazole thiourea salt is performed in situ, leveraging phase transfer catalysis to enhance reaction kinetics without the need for harsh conditions. This integration not only simplifies the equipment requirements but also minimizes the exposure of operators to potentially hazardous chemicals. The result is a synthesis route that is inherently more suitable for industrial production, offering a clear pathway for cost reduction in agrochemical manufacturing through process intensification.
Mechanistic Insights into One-Pot Hydroxymethylation and Oxidation
The core of this technological advancement lies in the precise control of reaction parameters during the hydroxymethylation and etherification phases. The process begins by reacting the pyrazole starting material with formaldehyde in the presence of an alkaline solution, such as sodium hydroxide or potassium carbonate, within an organic solvent like ethanol or acetonitrile. Maintaining the temperature between 0-30°C is critical to prevent side reactions and ensure the selective formation of the hydroxymethyl intermediate. Once this step is complete, difluorochloromethane gas is introduced directly into the system, facilitating an efficient etherification that converts the hydroxyl group into a difluoromethoxy moiety. The seamless transition between these steps without workup preserves the energy of the system and maintains the concentration of reactive species, which is key to achieving high conversion rates. This mechanistic efficiency is further supported by the use of specific phase transfer catalysts, such as tetrabutylammonium bromide, which facilitate the interaction between organic and aqueous phases during the subsequent condensation.
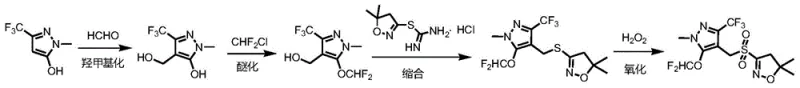
Following the formation of the thioether intermediate, the final oxidation step utilizes hydrogen peroxide in the presence of a catalyst like sodium tungstate or ammonium molybdate. This oxidation is carefully managed at temperatures ranging from 20-80°C to convert the sulfide linkage into the desired sulfone group without over-oxidation or degradation of the sensitive isoxazole ring. The use of hydrogen peroxide is particularly advantageous from an environmental standpoint, as it produces water as the only byproduct, aligning with green chemistry principles. Rigorous quenching with sodium thiosulfate or sodium sulfite ensures that any residual oxidant is neutralized, guaranteeing the stability and purity of the final crystalline product. This detailed mechanistic understanding allows for precise scaling and optimization, ensuring that the high-purity herbicide intermediates produced meet the stringent specifications required by global regulatory bodies.
How to Synthesize Pyraflufen-ethyl Efficiently
Implementing this synthesis route requires a thorough understanding of the sequential addition of reagents and the maintenance of specific thermal conditions to maximize yield and purity. The process is designed to be robust, allowing for flexibility in solvent choice and base selection while maintaining high performance across different batch sizes. Operators must monitor the reaction progress via liquid chromatography to ensure complete consumption of starting materials before proceeding to the next stage, particularly during the gas introduction phase. Detailed standard operating procedures are essential to replicate the success of the patent examples, which demonstrate yields exceeding 95 percent in the final oxidation step. For a comprehensive guide on the exact molar ratios, stirring speeds, and workup procedures, please refer to the standardized protocol outlined below.
- Perform hydroxymethylation of 1-methyl-3-trifluoromethyl-5-hydroxy-1H-pyrazole with formaldehyde in alkaline organic solvent at 0-30°C.
- Conduct etherification by introducing difluorochloromethane gas into the reaction mixture without isolation.
- Add isoxazole thiourea salt and phase transfer catalyst for condensation, followed by oxidation with hydrogen peroxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method presents compelling advantages for procurement managers and supply chain leaders seeking to optimize their sourcing strategies. The consolidation of multiple reaction steps into a single vessel significantly reduces the capital expenditure required for reactor infrastructure, as fewer units are needed to achieve the same production output. This simplification also translates to lower operational costs, as there is a marked decrease in the consumption of solvents, energy, and labor hours associated with intermediate transfers and purifications. By eliminating the need for expensive and hazardous intermediate isolations, the process inherently reduces waste generation and disposal costs, contributing to a more sustainable and economically viable manufacturing model. These efficiencies collectively enhance the competitiveness of the supply chain, allowing for more aggressive pricing strategies without compromising on quality or margin.
- Cost Reduction in Manufacturing: The one-pot nature of the synthesis eliminates the need for multiple isolation and purification steps, which are traditionally the most cost-intensive parts of fine chemical production. By reducing the number of unit operations, the process minimizes solvent loss and energy consumption, leading to substantial cost savings in utility and raw material usage. Furthermore, the higher overall yield means that less starting material is required to produce the same amount of final product, directly lowering the cost of goods sold. This economic efficiency is critical for maintaining profitability in a market where price pressure from downstream formulators is constantly increasing.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the complexity of the supply chain by decreasing the number of distinct raw materials and intermediates that need to be sourced and managed. With fewer steps comes a lower risk of bottlenecks or delays caused by equipment failure or quality issues at intermediate stages. The use of readily available reagents like formaldehyde and hydrogen peroxide ensures that the supply of key inputs remains stable and resilient against market fluctuations. This reliability is essential for meeting just-in-time delivery schedules and maintaining strong relationships with downstream herbicide manufacturers who depend on consistent supply.
- Scalability and Environmental Compliance: The robustness of the reaction conditions, particularly the moderate temperature ranges and the use of aqueous workups, makes this process highly scalable from pilot plant to commercial production volumes. The reduced generation of hazardous waste and the use of environmentally benign oxidants facilitate easier compliance with increasingly strict environmental regulations. This scalability ensures that production can be ramped up quickly to meet surges in demand without the need for significant process re-engineering. Additionally, the cleaner process profile reduces the burden on waste treatment facilities, further lowering the environmental compliance costs associated with large-scale manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this new synthesis technology. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for making informed decisions about technology adoption and partnership opportunities. The insights provided here aim to clarify the practical implications of the chemical innovations discussed in the previous sections.
Q: How does the new one-pot method improve yield compared to traditional routes?
A: The novel route described in CN113831333A eliminates intermediate isolation steps, reducing material loss and significantly improving overall yield compared to the 64% yield of previous methods.
Q: What are the key safety considerations for the oxidation step?
A: The oxidation using hydrogen peroxide requires precise temperature control between 20-80°C and the use of quenching agents like sodium thiosulfate to ensure safe handling of exothermic reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the simplification of reaction steps and the ability to perform multiple transformations in a single reactor make this method highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyraflufen-ethyl Supplier
The technical potential of this one-pot synthesis route underscores the importance of partnering with a CDMO expert capable of translating complex laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of this specific chemistry are managed with precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Pyraflufen-ethyl meets the highest international standards. We understand the critical nature of agrochemical intermediates in the global food supply chain and are committed to delivering consistent quality and reliability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this more efficient method. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the compatibility of this technology with your existing manufacturing infrastructure. Together, we can drive efficiency and innovation in the agrochemical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
