Scalable Electrochemical Synthesis of 3-Alkylthio-4-Anilinocoumarin Derivatives for Global Pharma Supply Chains
Scalable Electrochemical Synthesis of 3-Alkylthio-4-Anilinocoumarin Derivatives for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize complex heterocyclic scaffolds. Patent CN112501643A introduces a groundbreaking electrochemical synthesis method for producing 3-alkylthio-4-anilinocoumarin compounds, a class of molecules with significant biological activity and potential in drug development. This technology replaces hazardous chemical oxidants with electricity, utilizing a simple undivided cell setup with iron and nickel foam electrodes. For R&D directors and procurement managers, this represents a paradigm shift towards sustainable manufacturing, offering a route that eliminates the need for stoichiometric oxidants while maintaining high selectivity and yield under mild conditions. The method's reliance on abundant raw materials and its compatibility with various functional groups make it a robust candidate for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-alkylthio-4-anilinocoumarin derivatives has relied heavily on chemical oxidation strategies that suffer from significant inefficiencies and environmental drawbacks. A prominent prior art method involves the reaction of 4-anilinocoumarin with Bunte salts catalyzed by iodine salts, as illustrated in the reaction scheme below. This conventional approach necessitates the use of 1.5 equivalents of Bunte salts, which are not only expensive and limited in source but also result in poor atom economy. The generation of stoichiometric byproducts complicates the downstream purification process, often requiring chromatographic separation which is impractical for large-scale production. Furthermore, the extended reaction times and harsh conditions associated with these chemical oxidants increase energy consumption and operational risks, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
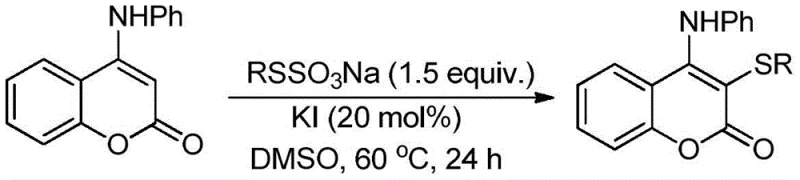
The Novel Approach
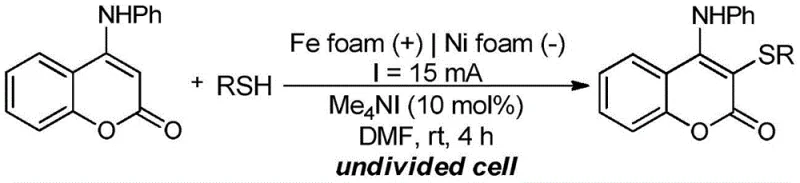
In stark contrast, the novel electrochemical methodology described in the patent utilizes electrons as a traceless oxidant, fundamentally altering the reaction landscape. As depicted in the general reaction scheme, the process couples 4-anilinocoumarin directly with readily available thiol compounds in an N,N-dimethylformamide solution containing an organic quaternary ammonium iodide catalyst. By applying a direct current across a foamed iron anode and a foamed nickel cathode, the system generates reactive iodine species in situ without the need for external chemical oxidants. This approach not only simplifies the reaction setup by operating in an undivided cell at room temperature but also drastically improves the atomic efficiency of the transformation. The elimination of expensive sulfur sources like Bunte salts and the ability to use simple alkyl thiols significantly reduces raw material costs, making this a highly attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Electrochemical C-H Thioetherification
Understanding the catalytic cycle is crucial for R&D teams aiming to optimize this process for specific substrates. The mechanism relies on the anodic oxidation of iodide ions derived from the organic quaternary ammonium salt catalyst. At the surface of the foamed iron anode, iodide anions lose electrons to form reactive iodine cations or electrophilic iodine species. These active species then interact with the 4-anilinocoumarin substrate to generate a key 3-iodo-4-anilinocoumarin intermediate. This intermediate is highly reactive towards nucleophilic attack by the thiol compound, leading to the formation of the desired carbon-sulfur bond and the release of molecular iodine. The elegance of this system lies in its closed-loop nature, where the molecular iodine is subsequently reduced back to iodide at the cathode, completing the catalytic cycle and ensuring the continuous regeneration of the active catalyst species throughout the reaction duration.
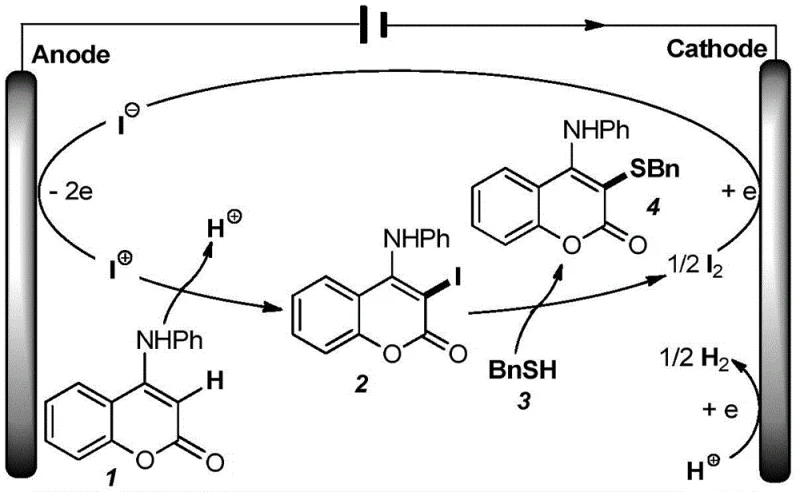
Beyond the primary catalytic cycle, the choice of electrode materials plays a pivotal role in controlling impurity profiles and maximizing yield. The patent data highlights that the foamed structure of the iron anode and nickel cathode provides a significantly larger surface area compared to planar metal sheets, enhancing current efficiency and mass transfer. This physical configuration minimizes side reactions such as over-oxidation of the thiol or decomposition of the coumarin scaffold, which are common pitfalls in electrochemical organic synthesis. Furthermore, the cathodic reduction of protons to hydrogen gas serves as a benign half-reaction that balances the electron flow without generating toxic waste. This precise control over the redox environment ensures that the final product exhibits a clean impurity profile, reducing the burden on quality control laboratories and facilitating easier regulatory approval for pharmaceutical applications.
How to Synthesize 3-Alkylthio-4-Anilinocoumarin Efficiently
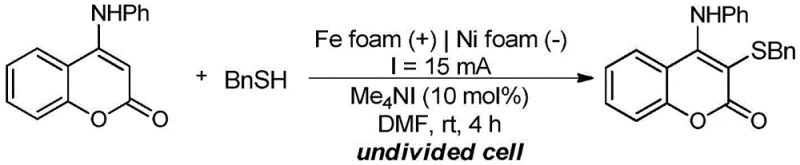
Implementing this electrochemical protocol requires careful attention to reaction parameters to achieve the reported high yields. The standard procedure involves dissolving the 4-anilinocoumarin substrate and the thiol reagent in DMF along with a catalytic amount of tetramethylammonium iodide. The specific reaction setup for a model substrate like benzyl mercaptan is shown below, demonstrating the simplicity of the reagent mixture. The reaction is conducted under a constant current of 15 mA at room temperature for approximately 4 hours. Monitoring the reaction progress via thin-layer chromatography ensures that the conversion is complete before workup. The detailed standardized synthesis steps, including specific molar ratios and electrode dimensions, are provided in the guide below to assist technical teams in replicating this high-efficiency process.
- Prepare the electrolyte solution by dissolving 4-anilinocoumarin, thiol compound, and tetramethylammonium iodide catalyst in N,N-dimethylformamide (DMF).
- Insert a foamed iron anode and a foamed nickel cathode into the undivided cell and apply a constant direct current of 15 mA at room temperature.
- After 4 hours, precipitate the product by adding water, filter the crystals, and recycle the filtrate containing the catalyst and solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical method offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio; replacing expensive, specialized sulfurating agents with commodity thiols and electricity significantly lowers the bill of materials. Additionally, the ability to recycle the solvent and catalyst system multiple times without significant loss of activity means that waste disposal costs are minimized, and the overall environmental footprint of the manufacturing process is substantially reduced. This aligns perfectly with modern corporate sustainability goals and regulatory pressures to minimize chemical waste in fine chemical production facilities.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and expensive Bunte salts directly translates to lower raw material expenditures. Since the catalyst and solvent can be recovered and reused effectively, the recurring cost per kilogram of product decreases significantly over time. Furthermore, the mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, resulting in substantial utility cost savings. The simplified workup procedure, which involves simple water precipitation rather than complex chromatography, reduces labor hours and consumable usage, driving down the overall cost of goods sold for these high-value intermediates.
- Enhanced Supply Chain Reliability: Relying on electricity as a reagent removes the supply chain volatility associated with specialized chemical oxidants that may face sourcing shortages or price fluctuations. The use of robust, non-precious metal electrodes like iron and nickel foam ensures that equipment maintenance and replacement costs remain low and predictable. The room temperature operation also reduces safety risks associated with thermal runaways, ensuring consistent production schedules and minimizing unplanned downtime. This stability is critical for maintaining continuous supply lines to downstream pharmaceutical customers who demand rigorous adherence to delivery timelines.
- Scalability and Environmental Compliance: The undivided cell design is inherently easier to scale than complex multi-compartment electrochemical reactors, facilitating a smoother transition from laboratory benchtop to pilot plant and commercial production. The absence of heavy metal catalysts and toxic oxidants simplifies wastewater treatment protocols, ensuring compliance with increasingly stringent environmental regulations. The high atom efficiency of the process means less waste is generated per unit of product, reducing the logistical burden of hazardous waste transport and disposal. This makes the technology particularly suitable for regions with strict environmental oversight, allowing for flexible global manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for technical teams evaluating the integration of this method into existing production workflows.
Q: Why is the electrochemical method superior to traditional Bunte salt methods?
A: Traditional methods require 1.5 equivalents of expensive Bunte salts and generate significant waste. The electrochemical approach uses electrons as traceless oxidants, operates at room temperature, and achieves higher atom efficiency without external chemical oxidants.
Q: What are the optimal electrode materials for this synthesis?
A: Experimental data indicates that a combination of a foamed iron anode and a foamed nickel cathode provides the highest current efficiency and catalytic effect, significantly outperforming platinum, copper, or graphite electrodes.
Q: Can the catalyst and solvent be recycled in this process?
A: Yes, the process allows for the direct recycling of the DMF solvent and the organic quaternary ammonium iodide catalyst after simple water precipitation of the product, reducing overall operational costs and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkylthio-4-Anilinocoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-alkylthio-4-anilinocoumarin meets the exacting standards required by global drug manufacturers. Our commitment to green chemistry aligns with this patent's methodology, allowing us to offer sustainable solutions without compromising on quality or consistency.
We invite you to collaborate with us to explore how this advanced electrochemical route can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can secure your supply of high-purity intermediates while driving down long-term manufacturing costs.
