Advanced Synthesis of 3-N-Acetyl Etimicin for High-Purity Antibiotic Manufacturing
Advanced Synthesis of 3-N-Acetyl Etimicin for High-Purity Antibiotic Manufacturing
The pharmaceutical industry's relentless pursuit of safer, more effective antibiotics has placed aminoglycosides like etimicin sulfate under intense scrutiny regarding their impurity profiles. Patent CN112608350B introduces a groundbreaking synthetic methodology for producing 3-N-acetyl etimicin, a critical reference standard and process-related impurity. This technical disclosure addresses a significant gap in the market, where previously no standardized synthesis or commercial availability existed for this specific compound. For R&D directors and quality control managers, the ability to synthesize this molecule with high fidelity is paramount for validating the safety and efficacy of the final drug substance. The patent outlines a robust, three-step chemical sequence that transforms etimicin into the target acetyl derivative with exceptional purity, reaching up to 99% in optimized examples. This development not only facilitates rigorous impurity tracking but also underscores the importance of precise structural modification in mitigating the ototoxicity associated with long-term aminoglycoside therapy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific aminoglycoside impurities like 3-N-acetyl etimicin has been plagued by significant technical hurdles and supply chain fragility. Traditional approaches often relied on non-selective acylation strategies that resulted in complex mixtures of regioisomers, necessitating laborious and low-yield purification steps. Furthermore, the absence of a dedicated, scalable route meant that reference standards were frequently sourced from limited, non-GMP compliant channels, introducing variability into quality control assays. The chemical complexity of the etimicin scaffold, with its multiple reactive amine and hydroxyl groups, makes selective functionalization at the 3-N position exceptionally challenging without sophisticated protecting group strategies. Consequently, manufacturers faced prolonged lead times and inflated costs when attempting to secure high-purity samples for regulatory submissions. The lack of a defined synthetic pathway also hindered the ability to perform comprehensive toxicological assessments, as the exact nature and quantity of impurities in the bulk drug could not be accurately benchmarked against a verified standard.
The Novel Approach
The methodology disclosed in patent CN112608350B represents a paradigm shift in how complex aminoglycoside derivatives are accessed. By employing a strategic sequence of selective protection, thermal acylation, and controlled deprotection, the inventors have achieved a highly efficient route to the target molecule. The process begins with the selective masking of reactive sites using di-tert-butyl dicarbonate, which sets the stage for regioselective modification. This is followed by a unique thermal treatment with formic acid that facilitates the introduction of the acetyl group under controlled conditions. Finally, a mild basic hydrolysis removes the protecting groups to reveal the desired 3-N-acetyl etimicin. This approach drastically simplifies the workflow, eliminating the need for cryogenic conditions or expensive transition metal catalysts often seen in alternative methodologies. The result is a streamlined process that is not only chemically elegant but also practically viable for commercial-scale production of reference standards.
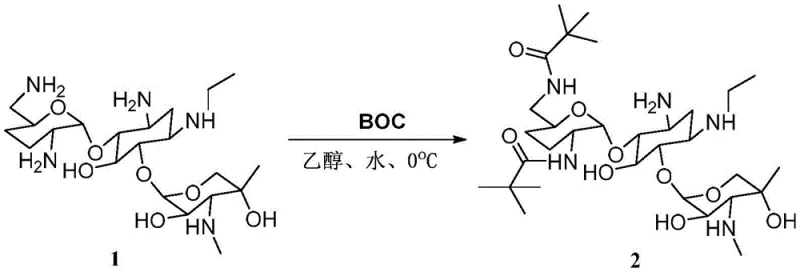
Mechanistic Insights into Selective N-Protection and Thermal Acylation
The core innovation of this synthesis lies in the precise manipulation of the etimicin scaffold's reactivity. The initial step involves the reaction of etimicin with di-tert-butyl dicarbonate (BOC anhydride) in a mixed solvent system of ethanol and water. This step is critical for differentiating between the various nucleophilic centers on the molecule. The steric bulk of the tert-butyl carbamate groups effectively shields specific amine functionalities, preventing unwanted side reactions during the subsequent acylation phase. The choice of solvent and temperature (0°C to 5°C) is meticulously optimized to favor the formation of the 3',5'-bis-protected intermediate, ensuring that the 3-N position remains accessible or is selectively targeted in the next step. This level of control is essential for minimizing the formation of regioisomeric byproducts that would otherwise complicate downstream purification and compromise the purity of the final reference standard.
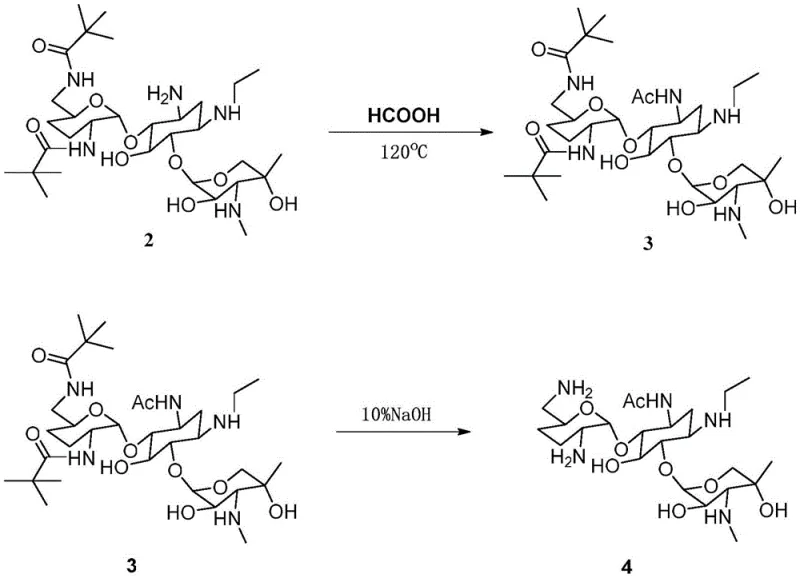
Following protection, the intermediate undergoes a thermal reaction with formic acid at elevated temperatures (120°C). While formic acid is typically associated with formylation, the specific conditions described in the patent facilitate the formation of the acetyl derivative, likely through a complex mechanistic pathway involving in-situ activation or specific catalytic effects of the solvent matrix. This step demonstrates the robustness of the protected intermediate, which withstands harsh acidic conditions without degradation of the glycosidic linkages. The final deprotection using 10% sodium hydroxide at 125°C cleaves the carbamate protecting groups, regenerating the free amines while retaining the newly installed acetyl moiety. The stability of the 3-N-acetyl group under these basic conditions highlights the kinetic selectivity of the acylation, ensuring that the target structure is preserved throughout the workup. This mechanistic understanding allows for precise tuning of reaction parameters to maximize yield and minimize impurity generation.
How to Synthesize 3-N-Acetyl Etimicin Efficiently
The synthesis of 3-N-acetyl etimicin as described in the patent data offers a clear, reproducible pathway for generating this critical pharmaceutical intermediate. The process is designed to be operationally simple, utilizing standard laboratory equipment and readily available reagents, which lowers the barrier to entry for manufacturing this high-value compound. By adhering to the specific molar ratios and temperature profiles outlined in the examples, chemists can consistently achieve high purity levels suitable for analytical reference standards. The following guide summarizes the critical operational phases required to execute this synthesis effectively, ensuring that the delicate balance between protection and functionalization is maintained throughout the reaction sequence.
- Protect etimicin using di-tert-butyl dicarbonate in ethanol/water at 0°C to form the bis-protected intermediate.
- React the protected intermediate with formic acid at 120°C for 4 hours to introduce the acetyl functionality.
- Perform base-mediated deprotection using 10% sodium hydroxide at 125°C, followed by silica gel column chromatography for isolation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the substantial reduction of supply chain risk associated with sourcing rare aminoglycoside impurities. By enabling in-house production or securing a reliable contract manufacturing partner for 3-N-acetyl etimicin, companies can decouple their quality control operations from volatile external markets. The simplicity of the reaction conditions—avoiding exotic catalysts or extreme cryogenic requirements—translates directly into lower capital expenditure and reduced operational overhead. Furthermore, the high yields reported in the patent examples suggest a material-efficient process that minimizes waste generation, aligning with modern sustainability goals and reducing the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of complex multi-step purification sequences and the use of commodity chemicals like formic acid and sodium hydroxide drive down the cost of goods sold significantly. By avoiding the need for specialized chiral catalysts or expensive protecting group reagents that require difficult removal steps, the overall process economics are improved. This cost efficiency allows for the production of larger batches of reference standards at a fraction of the historical cost, freeing up budget for other critical R&D initiatives. Additionally, the high purity achieved reduces the need for repetitive recrystallization or chromatography, further lowering solvent consumption and labor costs associated with downstream processing.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is vital for maintaining regulatory compliance. Unlike natural extraction methods or semi-synthetic routes prone to variability, this chemical synthesis provides a predictable output that can be scaled according to demand. This reliability mitigates the risk of production stoppages due to the unavailability of critical reference materials. Moreover, the use of stable, shelf-stable reagents means that raw material inventory can be managed more effectively, reducing the likelihood of supply disruptions caused by the short shelf-life of sensitive intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram-scale commercial production without significant re-engineering. The reaction conditions are compatible with standard stainless steel reactors, facilitating easy technology transfer to manufacturing sites. From an environmental perspective, the aqueous workup and the use of relatively benign reagents simplify waste treatment protocols. The reduction in hazardous waste streams and solvent usage contributes to a greener manufacturing profile, helping companies meet increasingly stringent environmental regulations and corporate social responsibility targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 3-N-acetyl etimicin. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind this novel method. Understanding these details is crucial for stakeholders evaluating the integration of this intermediate into their quality control workflows.
Q: Why is 3-N-acetyl etimicin critical for etimicin sulfate production?
A: 3-N-acetyl etimicin is a key process-related impurity in the synthesis of etimicin sulfate. Accurate quantification and control of this impurity are essential for meeting stringent regulatory safety standards and ensuring the ototoxicity profile of the final antibiotic remains within safe limits.
Q: What are the advantages of the disclosed synthesis route over traditional methods?
A: The patented method offers a significantly simplified workflow compared to complex semi-synthetic routes. By utilizing readily available reagents like di-tert-butyl dicarbonate and formic acid, the process eliminates the need for exotic catalysts, thereby reducing operational complexity and improving overall yield consistency.
Q: How does this method impact supply chain stability for aminoglycoside manufacturers?
A: By establishing a reliable in-house or contract manufacturing route for this specific reference standard, manufacturers can reduce dependency on scarce external suppliers. The robust nature of the reaction conditions ensures consistent batch-to-batch reproducibility, securing the supply chain for critical quality control materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-N-Acetyl Etimicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development and safety validation of next-generation antibiotics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering 3-N-acetyl etimicin that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the advanced synthetic techniques described in patent CN112608350B, we can provide a consistent, high-quality supply of this essential reference standard to support your regulatory filings and quality assurance programs.
We invite you to collaborate with us to optimize your supply chain for aminoglycoside manufacturing. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact us today to request specific COA data and route feasibility assessments, and discover how our expertise can enhance the efficiency and safety of your pharmaceutical production processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
