Revolutionizing Polymerizable Liquid Crystal Intermediate Production via Advanced Acid Halide Synthesis
Revolutionizing Polymerizable Liquid Crystal Intermediate Production via Advanced Acid Halide Synthesis
The landscape of electronic chemical manufacturing is constantly evolving, driven by the demand for higher purity and more efficient production routes for display materials. A significant technological breakthrough in this domain is detailed in patent CN108026017B, which introduces a novel method for producing acid halide solutions. This innovation specifically targets the synthesis of dicarboxylic acid chlorides, which serve as critical precursors for polymerizable liquid crystal compounds. Unlike traditional methodologies that necessitate the isolation and purification of unstable acid chloride intermediates, this patented approach utilizes a tetraalkylammonium salt-catalyzed system in non-aqueous miscible organic solvents. By enabling the direct use of concentrated acid halide solutions in subsequent esterification reactions, this technology offers a paradigm shift for reliable polymerizable liquid crystal intermediate suppliers seeking to optimize their manufacturing workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of monoester compounds from dicarboxylic acids has been plagued by inefficiencies inherent in the isolation of the acid chloride intermediate. In standard acyl halide methods, the reaction between a dicarboxylic acid and a chlorinating agent like thionyl chloride generates significant amounts of acidic byproducts, including sulfur dioxide, hydrogen chloride, and excess thionyl chloride. To prevent these acidic components from severely depressing the yield of the subsequent esterification reaction, conventional processes mandate a rigorous purification step. This typically involves removing solvents and low-boiling substances followed by recrystallization of the solid acid chloride. While feasible on a laboratory scale, this isolation protocol is industrially disadvantageous due to its complexity, high energy consumption, and the inevitable loss of material during solid handling and filtration. Furthermore, the instability of acid chlorides makes long-term storage and transportation risky, creating bottlenecks in the supply chain for high-purity electronic chemical intermediates.
The Novel Approach
The methodology disclosed in CN108026017B circumvents these logistical and chemical hurdles by redefining the state of the intermediate. Instead of isolating the dicarboxylic acid chloride as a solid, the process maintains it in a non-aqueous miscible organic solvent solution. The key innovation lies in the use of specific tetraalkylammonium salts as catalysts during the halogenation step. These catalysts allow for rapid conversion at moderate temperatures and, critically, remain compatible with the downstream esterification process. The reaction mixture is simply concentrated to remove volatile acidic byproducts, resulting in a stable mixed solution containing the acid halide, the catalyst, and a controlled, minimal amount of residual acid. This solution can then be directly fed into the esterification reactor. 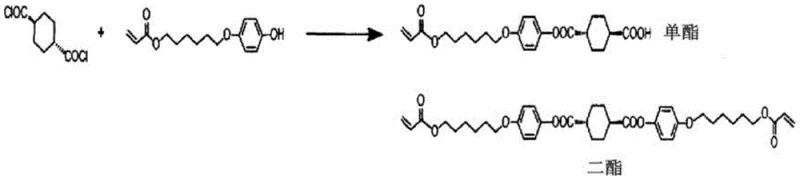
Mechanistic Insights into Tetraalkylammonium-Catalyzed Halogenation
The efficacy of this process is rooted in the unique physicochemical properties of the tetraalkylammonium salt catalysts. Unlike traditional catalysts such as N,N-dimethylformamide (DMF), which can form reactive Vilsmeier-Haack complexes that complicate downstream purification and potentially degrade product quality, tetraalkylammonium salts function primarily through phase transfer or nucleophilic activation mechanisms that are more benign in this specific context. The general reaction involves the interaction of the dicarboxylic acid with the halogenating agent in the presence of the quaternary ammonium salt, as illustrated by the general formulas for the reactants and catalysts. 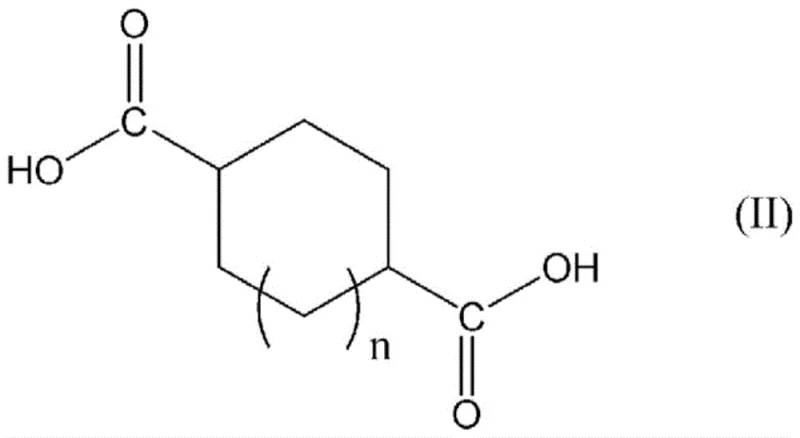
Furthermore, the choice of solvent plays a pivotal role in the success of this one-pot strategy. The patent specifies the use of non-aqueous miscible organic solvents with a Hildebrand solubility parameter ranging from 14.0 MPa^1/2 to 22.0 MPa^1/2. Solvents such as cyclopentyl methyl ether (CPME), chloroform, and toluene fall within this range. These solvents are selected because they effectively dissolve both the starting dicarboxylic acid and the resulting acid halide, while also being immiscible with water. This immiscibility is crucial for the final purification step, where the reaction mixture is washed with weakly acidic buffer solutions to remove residual catalyst and unreacted acid without hydrolyzing the sensitive ester bonds. The mechanistic pathway for the subsequent esterification involves the nucleophilic attack of the hydroxy compound on the acid chloride carbonyl carbon, facilitated by an organic base, leading to the formation of the monoester while the remaining acid chloride moiety is hydrolyzed to a carboxyl group during workup. 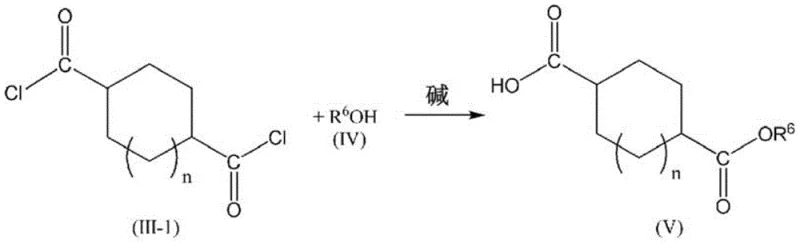
How to Synthesize Monoester Compounds Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters to maximize the yield of the monoester while minimizing diester formation. The process begins with the preparation of the acid halide solution, followed immediately by the addition of the hydroxy component and a base under controlled temperature conditions. The elimination of the isolation step significantly streamlines the workflow, reducing the total processing time and equipment footprint required for production. For R&D teams looking to adopt this methodology, understanding the stoichiometry and the specific washing protocols is essential to ensure the final product meets the stringent purity specifications required for display applications.
- React dicarboxylic acid with a halogenating agent in a non-aqueous miscible organic solvent using a tetraalkylammonium salt catalyst.
- Concentrate the resulting reaction mixture to remove acidic byproducts while retaining the acid halide in solution.
- Directly react the concentrated acid halide solution with a hydroxy compound and base to form the target monoester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this tetraalkylammonium-catalyzed process translates into tangible operational improvements that go beyond simple yield metrics. By fundamentally altering the production workflow, manufacturers can achieve substantial cost savings and enhanced supply reliability. The elimination of the recrystallization and drying steps for the acid chloride intermediate removes a major bottleneck in the production schedule. This reduction in unit operations not only lowers energy consumption but also decreases the demand for specialized isolation equipment, thereby reducing capital expenditure and maintenance costs associated with complex solid-handling machinery.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the workflow. By avoiding the isolation of the acid chloride, manufacturers eliminate the material losses typically associated with filtration, washing, and drying of solid intermediates. Additionally, the ability to use the reaction solution directly means that solvent recovery systems can be optimized for a continuous or semi-continuous flow rather than batch-wise solid processing. This efficiency leads to a significant reduction in the overall cost of goods sold (COGS) for the final monoester intermediate, making it a more competitive option in the global market for electronic chemicals.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by the instability of key intermediates. Acid chlorides are notoriously moisture-sensitive and difficult to store for extended periods. By keeping the intermediate in a stabilized solution form, the risk of degradation during storage is mitigated, allowing for more flexible inventory management. Furthermore, the shorter production cycle time means that manufacturers can respond more rapidly to fluctuations in demand from downstream panel makers. This agility ensures a more consistent supply of high-purity intermediates, reducing the risk of production stoppages for customers relying on just-in-time delivery models.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages. The reduction in solvent usage and the elimination of solid waste streams associated with recrystallization mother liquors contribute to a greener manufacturing profile. The process utilizes solvents that are amenable to efficient recovery and reuse, aligning with increasingly strict environmental regulations in the chemical sector. Moreover, the simplicity of the liquid-phase processing facilitates easier scale-up from pilot plants to commercial-scale reactors, as heat and mass transfer issues common in slurry processing are minimized. This scalability ensures that the technology can meet the growing volume requirements of the display industry without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acid halide solution technology. These insights are derived directly from the experimental data and embodiments provided in the patent literature, offering a clear understanding of the process capabilities and limitations for potential partners and technical evaluators.
Q: Why are tetraalkylammonium salts preferred over DMF for acid halide synthesis?
A: Tetraalkylammonium salts facilitate rapid reaction at lower temperatures and, crucially, do not adversely affect subsequent esterification yields unlike DMF residues which can promote side reactions.
Q: How does this method improve supply chain reliability for liquid crystal materials?
A: By eliminating the isolation and recrystallization steps of the acid chloride intermediate, the process reduces production lead time and minimizes material loss, ensuring more consistent availability.
Q: What is the role of the Hildebrand solubility parameter in solvent selection?
A: Solvents with a Hildebrand parameter between 14.0 and 22.0 MPa^1/2, such as CPME or chloroform, optimize the solubility of both reactants and products while facilitating efficient washing and purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acid Halide Solution Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in maintaining a competitive edge in the electronic materials sector. Our technical team has extensively analyzed the implications of patent CN108026017B and possesses the expertise to implement this tetraalkylammonium-catalyzed route effectively. We understand that transitioning to a new process requires confidence in the partner's ability to handle complex chemistry. Our facility is equipped with state-of-the-art reactors and purification systems capable of managing the specific solvent and catalyst requirements of this technology. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every batch for impurities, ensuring that the final monoester intermediates meet the exacting standards of the display industry.
We invite you to explore how this innovative manufacturing route can benefit your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of this advanced acid halide solution technology can drive efficiency and reliability in your production of polymerizable liquid crystal compounds.
