Scalable Visible-Light Synthesis of S-Aryl Thiosulfones for Advanced Pharmaceutical Intermediates
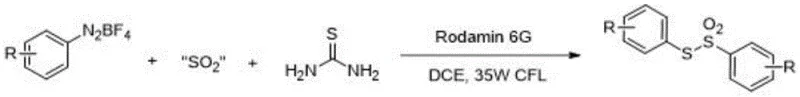
The landscape of organic synthesis for sulfur-containing scaffolds is undergoing a significant transformation driven by the demand for greener, more efficient methodologies. A pivotal advancement in this domain is detailed in patent CN109134326B, which discloses a novel synthetic route for S-aryl thiosulfone compounds. These compounds are increasingly recognized as versatile building blocks in medicinal chemistry, serving as precursors for both benzenesulfonyl and phenyl sulfide fragments. The disclosed method leverages visible light photoredox catalysis to achieve a three-component coupling reaction involving aryl diazonium tetrafluoroborates, thiourea, and solid sulfur dioxide substitutes. This approach represents a paradigm shift from traditional thermal methods, offering a pathway that operates under exceptionally mild conditions while maintaining high efficiency and product purity. For R&D directors and process chemists, this technology offers a robust alternative to legacy syntheses that often suffer from harsh reaction parameters and complex waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of S-aryl thiosulfone motifs has relied heavily on two primary strategies, both of which present distinct operational challenges for large-scale manufacturing. The first strategy involves the selective oxidation of disulfides, a process that frequently requires strong oxidizing agents which can lead to over-oxidation issues, generating sulfone byproducts that are difficult to separate from the desired thiosulfone. The second conventional route utilizes the coupling of existing sulfonyl reagents with thioether compounds, often necessitating the use of corrosive acids or unstable gaseous sulfur dioxide. These traditional methods not only impose severe safety constraints due to the handling of hazardous gases and strong acids but also limit the functional group tolerance of the substrate. Furthermore, the requirement for elevated temperatures or cryogenic conditions in some variations increases energy consumption and complicates the engineering controls required for safe operation, thereby inflating the overall cost of goods sold (COGS) for these critical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a radical-based assembly that circumvents the need for harsh oxidants or gaseous reagents. By utilizing thiourea as a sulfur source and solid metabisulfites (such as Na2S2O5 or K2S2O5) as sulfur dioxide surrogates, the process eliminates the logistical nightmares associated with gas handling. The reaction proceeds at room temperature (25°C) under the irradiation of a standard 35W fluorescent lamp, utilizing Rodamin 6G as an organic photocatalyst. This visible-light-driven approach ensures that the reaction energy is delivered precisely where needed, minimizing thermal degradation of sensitive functional groups. The simplicity of the reagent profile—combining readily available industrial chemicals with a common dye catalyst—drastically simplifies the supply chain logistics. Moreover, the workup procedure is streamlined to a direct concentration followed by column chromatography, avoiding the extensive aqueous quenching and extraction steps typical of acid-mediated processes, which significantly reduces solvent usage and waste generation.
Mechanistic Insights into Rodamin 6G-Catalyzed Radical Coupling
The elegance of this synthesis lies in its intricate yet efficient radical mechanism, which orchestrates the formation of two distinct radical species that converge to form the target bond. The process initiates with the nucleophilic addition of thiourea to the aryl diazonium tetrafluoroborate, forming a phenylisothiourea salt intermediate which subsequently eliminates urea to generate a thiophenol anion. Upon irradiation, the Rodamin 6G photocatalyst enters an excited state, possessing sufficient oxidative potential to convert the thiophenol anion into a thiophenol radical via a single electron transfer (SET) oxidation. Simultaneously, the reduced form of the photocatalyst engages in a reductive SET with another molecule of the aryl diazonium salt, cleaving the nitrogen bond to release nitrogen gas and generate a highly reactive aryl radical. This aryl radical rapidly traps the sulfur dioxide released from the solid surrogate to form a benzenesulfonyl radical. The final step involves the radical-radical coupling between the thiophenol radical and the benzenesulfonyl radical, forging the unique S-S(O)2 linkage characteristic of S-aryl thiosulfones.
This dual-radical generation strategy ensures high atom economy and minimizes side reactions commonly associated with ionic pathways. The use of solid sulfur dioxide substitutes like DABSO or sodium metabisulfite provides a controlled release of SO2, preventing the formation of polysulfones or other oligomeric impurities. From an impurity control perspective, the mild conditions prevent the decomposition of sensitive substituents such as esters, nitriles, or halogens, which might otherwise degrade under the strongly acidic or oxidative conditions of traditional methods. The result is a cleaner reaction profile that facilitates easier downstream purification, a critical factor for pharmaceutical intermediates where strict impurity thresholds must be met. The mechanistic understanding confirms that the reaction is driven by the redox properties of the photocatalyst rather than thermal activation, allowing for precise control over the reaction kinetics through light intensity modulation.
How to Synthesize S-Aryl Thiosulfones Efficiently
Implementing this photoredox protocol requires careful attention to the stoichiometry and atmospheric conditions to maximize yield and reproducibility. The patent specifies a molar ratio where the aryl diazonium salt serves as the limiting reagent (1.0 equivalent), while the sulfur dioxide substitute is used in excess (2.0 equivalents) to ensure complete trapping of the aryl radical. Thiourea is employed at 1.5 equivalents to drive the initial formation of the thiophenol precursor. The reaction must be conducted under an inert atmosphere, typically high-purity nitrogen or argon, to prevent the quenching of radical intermediates by oxygen, which would otherwise lead to oxidation byproducts and reduced yields. The choice of solvent is also critical, with 1,2-dichloroethane (DCE) or acetonitrile (MeCN) identified as optimal media for solubilizing the ionic diazonium salts while maintaining compatibility with the photocatalytic cycle.
- Prepare the reaction mixture by adding aryl diazonium tetrafluoroborate, sulfur dioxide solid substitute (e.g., Na2S2O5), thiourea, and Rodamin 6G catalyst into a reaction vessel under inert atmosphere.
- Add organic solvent such as 1,2-dichloroethane (DCE) or acetonitrile and stir the mixture under 35W fluorescent light irradiation at room temperature (25°C) for approximately 12 hours.
- Monitor reaction progress via TLC, then concentrate the solution under reduced pressure and purify the crude product using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this photoredox-mediated synthesis offers tangible strategic benefits that extend beyond mere technical feasibility. The substitution of hazardous gaseous reagents with stable, shelf-stable solids like sodium metabisulfite and thiourea fundamentally alters the risk profile of the manufacturing process. This shift not only enhances workplace safety but also simplifies regulatory compliance and storage requirements, as these solids do not require specialized high-pressure containment or cryogenic infrastructure. The reliance on visible light as the energy source, rather than high-temperature heating or cryogenic cooling, translates directly into reduced utility costs and a lower carbon footprint for the production facility. Furthermore, the use of Rodamin 6G, an inexpensive and commercially abundant organic dye, eliminates the dependency on scarce and costly transition metal catalysts, mitigating supply chain volatility associated with precious metals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous reagents. By replacing strong oxidants and gaseous sulfur dioxide with cheap, bulk-available solids, the raw material costs are significantly lowered. Additionally, the mild reaction conditions reduce the energy load on the manufacturing plant, as there is no need for extensive heating or cooling systems. The simplified workup procedure, which avoids complex aqueous washes required to neutralize strong acids, further reduces solvent consumption and waste disposal costs, leading to a more lean and cost-effective production model for high-value fine chemicals.
- Enhanced Supply Chain Reliability: The robustness of the reagent supply chain is a critical advantage. Thiourea and metabisulfites are commodity chemicals produced on a massive scale for various industries, ensuring a stable and continuous supply with minimal risk of shortage. Unlike specialized sulfonyl chlorides or unstable diazo compounds that may have long lead times, these starting materials are readily accessible from multiple global suppliers. This diversity in sourcing options empowers procurement teams to negotiate better terms and ensures that production schedules are not disrupted by single-source bottlenecks, thereby enhancing the overall resilience of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but the use of simple fluorescent lamps and the absence of exothermic hazards make this process highly scalable. The reaction generates nitrogen gas as the only gaseous byproduct, which is environmentally benign and easy to vent, avoiding the need for complex scrubbing systems required for acidic or toxic gases. The high selectivity of the radical coupling minimizes the formation of toxic byproducts, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile makes the process attractive for long-term sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their own pipelines.
Q: What are the key advantages of this photoredox method over traditional oxidation strategies?
A: Unlike traditional methods that require harsh oxidants or strong acidic conditions to oxidize disulfides, this patented process operates under mild visible light conditions at room temperature. It utilizes inexpensive, stable solid sources for sulfur dioxide and thiourea, eliminating the need for hazardous gaseous SO2 and reducing safety risks associated with strong oxidizers.
Q: Can this synthesis be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It uses commercially available raw materials like thiourea and sodium metabisulfite, operates at ambient temperature without cryogenic cooling, and employs a simple workup procedure involving concentration and column chromatography, making it suitable for large-scale manufacturing of complex fine chemicals.
Q: What is the role of Rodamin 6G in this reaction mechanism?
A: Rodamin 6G acts as a photocatalyst that facilitates single electron transfer (SET) processes. Upon excitation by visible light, it oxidizes the thiophenol anion to a thiophenol radical and, in its reduced state, reduces the aryl diazonium salt to generate an aryl radical, enabling the radical coupling that forms the S-aryl thiosulfone bond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Aryl Thiosulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photoredox catalysis in modernizing the synthesis of complex sulfur-containing scaffolds. As a leading CDMO partner, we possess the technical expertise to translate laboratory-scale innovations like the one described in CN109134326B into robust, commercial-scale processes. Our facilities are equipped with advanced photochemical reactors capable of handling visible-light mediated reactions efficiently, ensuring that the benefits of mild conditions and high selectivity are retained during scale-up. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee the quality of every batch.
We invite pharmaceutical and agrochemical companies to leverage our capabilities for the production of high-purity S-aryl thiosulfones and related intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this novel route can optimize your COGS. We encourage you to contact our technical procurement team to request specific COA data for similar compounds and to discuss route feasibility assessments for your target molecules, ensuring a seamless transition from development to commercial supply.
