Advanced Cu-Catalyzed Synthesis of Polysubstituted Benzothiazoles for Commercial Scale-Up
Advanced Cu-Catalyzed Synthesis of Polysubstituted Benzothiazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly benzothiazoles, which serve as critical cores in numerous bioactive molecules and optoelectronic materials. A groundbreaking approach detailed in patent CN111187233B introduces a highly efficient, copper-catalyzed strategy for synthesizing polysubstituted benzothiazoles and their derivatives. This innovation specifically targets the formation of N-(2-(benzo[d]thiazol-2-yl)phenyl)morpholine-4-carboxamide derivatives and 2-(benzo[d]thiazol-2-yl)aniline derivatives through a streamlined one-pot process. By leveraging inexpensive copper catalysts and molecular oxygen as a green oxidant, this method addresses long-standing challenges regarding cost, environmental impact, and operational complexity in heterocycle synthesis.
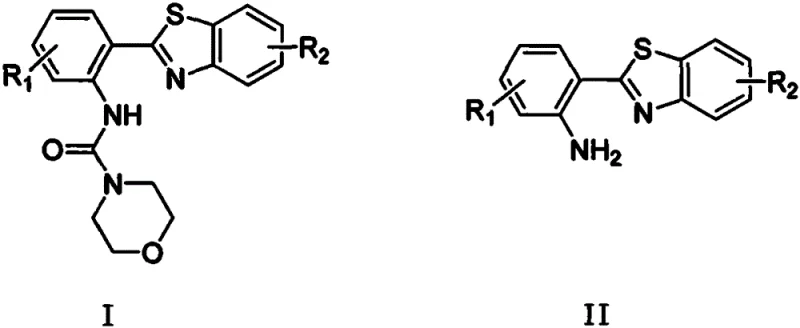
The significance of this technology lies in its ability to produce molecularly stable compounds with excellent chemical properties, filling a notable gap in existing synthetic literature where such specific derivatives were previously difficult to access. For R&D directors and process chemists, the availability of a reliable pathway to these structures opens new avenues for drug discovery and material science applications. The reaction system is designed to be simple yet powerful, utilizing readily available starting materials that do not require extensive pretreatment, thereby enhancing the overall atom economy and reducing the carbon footprint associated with multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of benzothiazole derivatives has often relied on harsh reaction conditions, expensive noble metal catalysts, or multi-step sequences that generate significant waste. Conventional routes frequently involve the use of stoichiometric oxidants or toxic reagents, which pose serious safety hazards and complicate waste disposal protocols in large-scale manufacturing. Furthermore, many existing methods suffer from limited substrate scope, failing to tolerate sensitive functional groups that are often essential for biological activity in pharmaceutical intermediates. The reliance on precious metals like palladium or platinum not only drives up the raw material costs but also necessitates rigorous purification steps to remove trace metal residues, which is a critical quality control hurdle in API production.
The Novel Approach
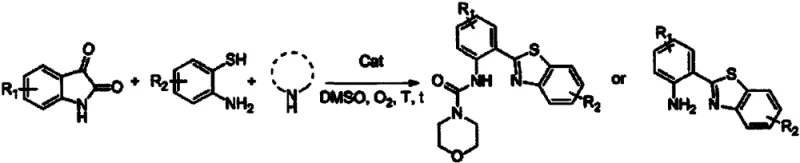
In stark contrast, the methodology disclosed in patent CN111187233B revolutionizes this landscape by employing a cost-effective copper catalytic system under an oxygen atmosphere. This novel approach enables the direct conversion of indole diketones, o-aminothiophenols, and amines into the target benzothiazole structures in a single reaction vessel. The use of molecular oxygen as the terminal oxidant is a standout feature, offering a green and sustainable alternative to traditional chemical oxidants. This shift not only simplifies the reaction setup but also drastically reduces the generation of hazardous byproducts. The process operates under relatively mild thermal conditions, typically between 100°C and 140°C, which enhances energy efficiency and minimizes the risk of thermal degradation of sensitive substrates.
Mechanistic Insights into Cu-Catalyzed Oxidative Cyclization
The core of this synthetic breakthrough is the copper-catalyzed oxidative coupling mechanism, which facilitates the simultaneous formation of carbon-sulfur and carbon-nitrogen bonds essential for the benzothiazole ring closure. The reaction initiates with the coordination of the copper catalyst to the sulfur and nitrogen centers of the o-aminothiophenol and the carbonyl oxygen of the indole diketone. Under an oxygen atmosphere, the copper species undergoes redox cycling, activating the C-H bonds adjacent to the heteroatoms and promoting the dehydrogenative coupling required for cyclization. This mechanistic pathway ensures high selectivity for the desired benzothiazole core while suppressing side reactions that typically plague non-catalytic thermal condensations.
From an impurity control perspective, the use of a well-defined copper catalyst system allows for precise tuning of the reaction kinetics, leading to cleaner reaction profiles. The oxidative nature of the process means that hydrogen is removed as water, avoiding the accumulation of reduced byproducts that can be difficult to separate. Additionally, the compatibility of this system with various amine nucleophiles, such as morpholine, suggests a flexible mechanism where the amine participates in the final amidation step without interfering with the initial heterocycle formation. This mechanistic robustness is crucial for ensuring consistent batch-to-batch quality, a key metric for supply chain reliability in the production of high-purity pharmaceutical intermediates.
How to Synthesize Polysubstituted Benzothiazole Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the reactants and the selection of the appropriate solvent system to maximize yield and purity. The patent outlines a straightforward procedure where indole diketones, o-aminothiophenols, and amines are mixed with a copper catalyst in solvents like DMSO or DMF. The reaction is then heated under an oxygen atmosphere for a duration ranging from 8 to 24 hours, depending on the specific substituents involved. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical guide below, ensuring reproducibility for process development teams.
- Mix indole diketone compounds, o-aminothiophenol compounds, and amines or bases with a copper catalyst and organic solvent in a reaction vessel.
- Heat the reaction mixture uniformly to 100°C-140°C under an oxygen atmosphere for 8 to 24 hours to facilitate the cyclization.
- Upon completion, purify the reaction mixture to isolate the target polysubstituted benzothiazole derivatives with high structural stability.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology represents a strategic opportunity to optimize cost structures and enhance supply security. The elimination of precious metal catalysts removes a major variable cost driver and mitigates the supply risks associated with fluctuating prices of rare earth elements. Furthermore, the simplified one-pot nature of the reaction reduces the number of unit operations required, directly translating to lower capital expenditure on equipment and reduced labor costs per kilogram of product. The use of commodity chemicals like isatins and o-aminothiophenols ensures a stable and diverse supplier base, preventing bottlenecks in raw material sourcing.
- Cost Reduction in Manufacturing: The substitution of expensive noble metals with abundant copper salts significantly lowers the catalyst cost per batch, while the high atom economy minimizes raw material waste. This qualitative shift in reagent selection leads to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on yield or quality standards.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and a robust catalytic system, the risk of supply disruption due to specialized reagent shortages is drastically reduced. The mild reaction conditions also allow for the use of standard stainless steel reactors, increasing the number of qualified manufacturing sites capable of producing these intermediates.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily water and benign organic byproducts, simplifying effluent treatment and ensuring compliance with stringent environmental regulations. The straightforward workup and purification steps facilitate easy scale-up from pilot plant to commercial production volumes, ensuring a continuous supply of high-purity products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, catalyst loading, and product stability for potential partners and licensees.
Q: What are the key advantages of this Cu-catalyzed benzothiazole synthesis?
A: The method utilizes cheap copper catalysts instead of precious metals, operates under mild conditions (100°C-140°C), and achieves high atom economy in a one-pot process, significantly reducing environmental pollution and raw material costs.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process requires simple reaction equipment, uses widely available raw materials like isatins and o-aminothiophenols, and involves straightforward operational steps, making it highly suitable for commercial scale-up and mass production.
Q: What types of substituents are tolerated in this reaction?
A: The reaction demonstrates broad substrate scope, tolerating various groups such as hydrogen, halogens, alkyls, alkoxy groups, and substituted aryl or heterocyclic rings on both the indole and thiophenol components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Benzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the global pharmaceutical and fine chemical markets. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our state-of-the-art facilities are equipped to handle the specific solvent systems and oxygen atmosphere requirements of this process, maintaining stringent purity specifications through our rigorous QC labs to meet the highest international standards.
We invite you to collaborate with us to leverage this innovative technology for your next-generation drug candidates or advanced materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your bill of materials. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the supply of high-purity polysubstituted benzothiazoles.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
