Scalable Green Synthesis of Ferrocenyl Dihydropyrimidinones for Pharma Applications
Scalable Green Synthesis of Ferrocenyl Dihydropyrimidinones for Pharma Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for complex heterocyclic compounds that offer both high efficiency and environmental sustainability. Patent CN111217862B introduces a groundbreaking methodology for the preparation of 4-aryl-6-ferrocenyl-3,4-dihydropyrimidin-2(1H)-one derivatives, which are critical scaffolds in medicinal chemistry known for their diverse biological activities including anticancer and antiviral properties. This innovation leverages a choline chloride-urea deep eutectic solvent (DES) system combined with Lewis acid catalysis to achieve exceptional yields under mild conditions. By replacing traditional volatile organic solvents with a recyclable, non-toxic eutectic mixture, this process addresses the growing regulatory pressure for greener manufacturing practices while maintaining rigorous quality standards required for pharmaceutical intermediates. The integration of ferrocenyl moieties further enhances the electrochemical and structural stability of the final compounds, making them highly valuable for advanced drug discovery programs.
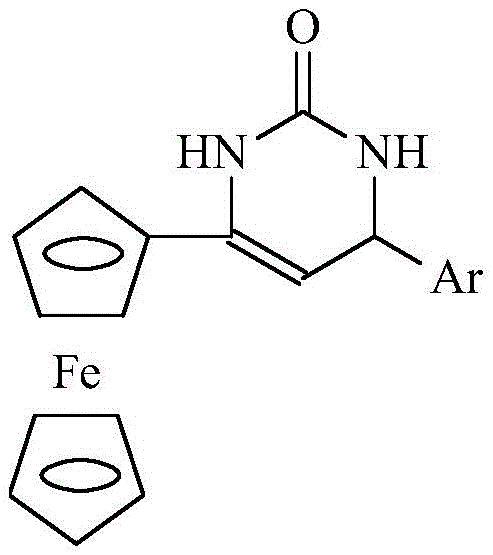
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for dihydropyrimidin-2(1H)-one (DHPM) derivatives, such as the classic Biginelli reaction, often rely heavily on hazardous organic solvents and strong mineral acids that pose significant safety and disposal challenges in a commercial setting. These conventional methods frequently require harsh reaction conditions, prolonged heating, and complex workup procedures involving multiple extraction steps to remove acidic residues and solvent traces, which inevitably increases the overall production cost and carbon footprint. Furthermore, the use of stoichiometric amounts of corrosive catalysts can lead to equipment degradation and the generation of substantial chemical waste, complicating compliance with increasingly stringent environmental regulations. The difficulty in recovering and reusing these organic solvents also contributes to resource inefficiency, making the traditional approach less economically viable for large-scale manufacturing of high-value fine chemicals where margin optimization is critical.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a deep eutectic solvent composed of choline chloride and urea, which serves a dual function as both the reaction medium and a promoter for the cyclocondensation reaction. This green solvent system operates effectively at a moderate temperature of 80°C, significantly reducing energy consumption compared to high-temperature reflux conditions often required in organic solvents. The simplicity of the post-reaction workup, which involves merely pouring the mixture into water to precipitate the solid product, eliminates the need for extensive chromatographic purification or solvent evaporation steps. Additionally, the filtrate containing the deep eutectic solvent can be recovered and reused multiple times without a significant drop in catalytic efficiency, offering a sustainable cycle that aligns perfectly with the principles of green chemistry and circular economy in modern chemical manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization in DES
The reaction mechanism involves a synergistic interaction between the Lewis acid catalyst and the hydrogen-bonding network of the deep eutectic solvent, which activates the carbonyl groups of the acetylferrocene and aromatic aldehyde for nucleophilic attack by urea. The Lewis acid, such as ZnCl2 or FeCl3, coordinates with the oxygen atoms of the reactants, increasing their electrophilicity and facilitating the formation of the key C-C and C-N bonds required to construct the dihydropyrimidine ring. The unique polarity and viscosity of the DES medium help stabilize the transition states and intermediates, preventing side reactions and ensuring high regioselectivity towards the desired 4-aryl-6-ferrocenyl product. This controlled environment minimizes the formation of polymeric byproducts or decomposition of the sensitive ferrocenyl group, which is crucial for maintaining the integrity of the organometallic functionality throughout the synthesis.
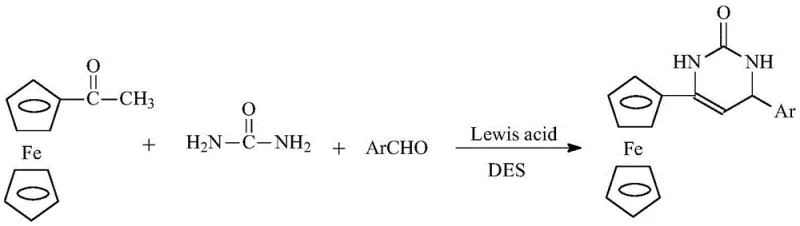
Impurity control is inherently managed through the precise tuning of the catalyst loading and the stoichiometric balance of the reactants within the eutectic phase. The homogeneous nature of the catalytic system ensures uniform heat distribution and mass transfer, reducing localized hot spots that could lead to thermal degradation or charring of the organic materials. Upon completion, the addition of water disrupts the eutectic structure, causing the hydrophobic product to precipitate while the hydrophilic solvent components remain in the aqueous phase, allowing for a clean separation. This physical separation mechanism effectively removes unreacted starting materials and soluble impurities without the need for additional washing with organic solvents, resulting in a crude product with high purity that often requires only simple recrystallization to meet pharmaceutical grade specifications.
How to Synthesize 4-aryl-6-ferrocenyl-3,4-dihydropyrimidin-2(1H)-one Efficiently
The standardized protocol for this synthesis begins with the preparation of the deep eutectic solvent by mixing choline chloride and urea in a specific molar ratio and heating the mixture to 80°C until a clear liquid forms. Once the solvent matrix is established, the reactants including acetylferrocene, urea, and the chosen aromatic aldehyde are introduced along with a catalytic amount of Lewis acid to initiate the cyclocondensation. The reaction progress is closely monitored using thin-layer chromatography (TLC) to ensure complete conversion before proceeding to the isolation stage, which guarantees consistent batch-to-batch quality. Detailed standardized synthesis steps see the guide below.
- Prepare deep eutectic solvent by stirring choline chloride and urea at 80°C.
- Add acetylferrocene, urea, aromatic aldehyde, and Lewis acid catalyst to the solvent.
- React at 80°C monitored by TLC, then pour into water to precipitate and filter the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial cost reductions by eliminating the purchase and disposal costs associated with large volumes of volatile organic solvents and corrosive mineral acids. The ability to recycle the deep eutectic solvent multiple times significantly lowers the raw material cost per kilogram of the final product, providing a competitive edge in pricing strategies for bulk chemical supply contracts. Moreover, the use of commercially available and inexpensive starting materials like choline chloride and urea ensures a stable supply chain that is not subject to the volatility of specialized reagent markets. This stability allows for more accurate long-term budgeting and reduces the risk of production delays caused by raw material shortages, which is a critical factor for supply chain managers planning annual procurement cycles.
- Cost Reduction in Manufacturing: The elimination of organic solvents removes the need for expensive solvent recovery distillation units and reduces the energy load associated with solvent evaporation, leading to drastic operational expenditure savings. The recyclability of the catalytic solvent system means that the effective cost of the reaction medium is amortized over multiple batches, further driving down the variable cost of goods sold. Additionally, the simplified workup procedure reduces labor hours and consumable usage during the purification phase, contributing to overall manufacturing efficiency. These cumulative savings allow for a more aggressive pricing model while maintaining healthy profit margins in the competitive fine chemical market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the solvent system and readily available aromatic aldehydes ensures that the supply chain is resilient against geopolitical or logistical disruptions affecting specialty reagents. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without compromising yield, reduces the risk of batch failures that could interrupt supply continuity. This reliability is paramount for downstream pharmaceutical customers who require just-in-time delivery of intermediates to maintain their own production schedules without interruption. Consequently, suppliers adopting this technology can offer higher service level agreements and build stronger long-term partnerships with key accounts.
- Scalability and Environmental Compliance: The process is inherently scalable as it avoids exothermic runaways common in strong acid catalysis, allowing for safe operation in larger reactors without complex cooling infrastructure. The green nature of the solvent system simplifies waste treatment protocols, as the aqueous waste stream is less hazardous and easier to treat than organic solvent mixtures, ensuring compliance with strict environmental discharge regulations. This ease of compliance reduces the administrative burden and potential fines associated with environmental permitting, making the facility more attractive for investment and expansion. Furthermore, the high atom economy of the reaction minimizes waste generation at the source, aligning with corporate sustainability goals and enhancing the brand reputation of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology in an industrial setting. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the advantages of using Deep Eutectic Solvents (DES) in this synthesis?
A: DES eliminates the need for volatile organic solvents, reduces toxicity, and allows for solvent recycling up to five times without significant yield loss, drastically lowering environmental impact and operational costs.
Q: Can this process be scaled for industrial production?
A: Yes, the process operates at mild temperatures (80°C) with simple workup procedures (water precipitation), making it highly suitable for large-scale commercial manufacturing without complex equipment requirements.
Q: What is the purity profile of the resulting intermediates?
A: The method yields high-purity products (yields over 91% reported in examples) with minimal impurities due to the selective catalytic nature of the Lewis acid in the DES medium, simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-aryl-6-ferrocenyl-3,4-dihydropyrimidin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green synthetic route for producing high-value ferrocenyl intermediates required in next-generation therapeutic applications. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and impurity profiles of these complex organometallic compounds to meet global pharmacopoeia standards. We are committed to delivering consistent quality and supply security for our clients' most critical drug development projects.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this process can optimize your bill of materials. Please contact us to request specific COA data and route feasibility assessments that will validate the commercial viability of this synthesis for your portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
