Advanced 1-Phospha Norbornene Chiral Phosphine Catalysts for Asymmetric Synthesis and Commercial Scale-Up
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel phosphorus-based architectures that address long-standing stability issues. Patent CN112851710B discloses a groundbreaking class of 1-phospha norbornene chiral phosphine catalysts, designed specifically to overcome the inherent instability of traditional P-chiral centers. Historically, the tendency of phosphorus chiral centers to racemize at elevated temperatures has severely limited their utility in robust industrial processes. This new technology embeds the phosphorus atom into a rigid bicyclic framework, effectively locking the stereochemistry and preventing epimerization. 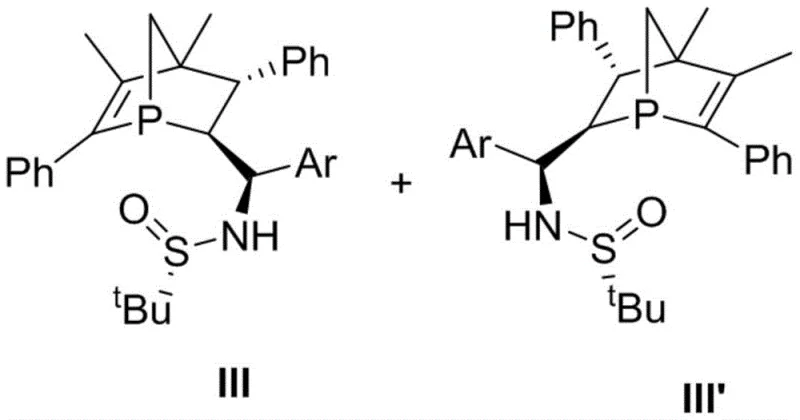 As a reliable pharmaceutical intermediate supplier, understanding these structural advancements is critical for developing next-generation synthetic routes. The catalysts, represented by formulas III and III', feature a unique arrangement where three P-C bonds are fully constrained within the norbornene skeleton. This design not only enhances thermal stability but also provides a well-defined chiral environment for substrate binding. For R&D directors seeking to optimize asymmetric cyclization reactions, this platform offers a compelling solution that combines high stereoselectivity with operational robustness, paving the way for more efficient manufacturing of complex chiral molecules.
As a reliable pharmaceutical intermediate supplier, understanding these structural advancements is critical for developing next-generation synthetic routes. The catalysts, represented by formulas III and III', feature a unique arrangement where three P-C bonds are fully constrained within the norbornene skeleton. This design not only enhances thermal stability but also provides a well-defined chiral environment for substrate binding. For R&D directors seeking to optimize asymmetric cyclization reactions, this platform offers a compelling solution that combines high stereoselectivity with operational robustness, paving the way for more efficient manufacturing of complex chiral molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to designing chiral organophosphine catalysts have predominantly focused on modifying the chiral backbone while leaving the phosphorus coordination sphere relatively flexible. In many conventional P-chiral ligands, the phosphorus atom is prone to inversion or racemization, especially under the thermal stress often required for demanding organic transformations. This instability arises because the lone pair of electrons on the trivalent phosphorus can facilitate pyramidal inversion, leading to a loss of optical purity over time. Furthermore, the synthesis of such ligands often involves cumbersome multi-step sequences with poor atom economy, resulting in significant waste generation and higher production costs. The reliance on sensitive precursors and the difficulty in purifying diastereomeric mixtures further complicate the supply chain for these critical reagents. Consequently, despite their theoretical potential, many P-chiral catalysts have failed to transition from academic curiosity to commercial viability due to these reproducibility and stability challenges.
The Novel Approach
The innovation described in the patent introduces a paradigm shift by utilizing a 1-phospha norbornene scaffold to rigidify the phosphorus center. By embedding the phosphorus atom within a strained bicyclic system, the catalyst effectively eliminates the degrees of freedom that lead to racemization. This structural rigidity ensures that the chiral information encoded in the molecule is preserved even under rigorous reaction conditions. The synthesis strategy employs a modular approach, starting from readily available phosphacyclopentadiene derivatives and utilizing a phospha-Diels-Alder reaction to construct the core framework. This method allows for the systematic variation of substituents on the aromatic rings, enabling fine-tuning of the electronic and steric properties of the catalyst. For procurement managers, this modularity translates to a versatile platform where a single synthetic route can generate a library of catalysts tailored for specific substrates, thereby reducing the need for developing entirely new processes for each new target molecule.
Mechanistic Insights into Grignard Addition and Desulfurization
The synthesis of these advanced catalysts relies on a precise two-step sequence that ensures high stereochemical fidelity. The first critical stage involves the nucleophilic addition of aryl Grignard reagents to a chiral phosphine sulfide imine precursor (Compound I). This reaction is conducted at cryogenic temperatures, specifically -78°C, in anhydrous tetrahydrofuran to control the reactivity of the Grignard species and prevent side reactions. 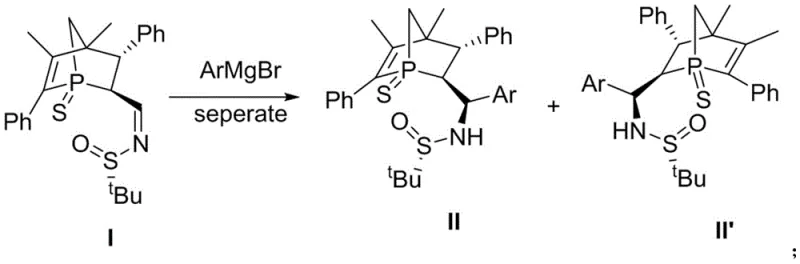 The low temperature is essential for maintaining the integrity of the chiral sulfinamide auxiliary, which directs the formation of the new carbon-phosphorus bond with high diastereoselectivity. Following the addition, the reaction mixture is quenched with ammonium chloride and extracted, yielding the intermediate sulfinamide compounds (II-a to II'-g) in yields ranging from 39% to 46%. This step establishes the foundational stereochemistry of the side chain, which is crucial for the subsequent catalytic performance. The use of Schlenk techniques throughout this process underscores the sensitivity of the intermediates to moisture and oxygen, necessitating strict inert atmosphere protocols during manufacturing.
The low temperature is essential for maintaining the integrity of the chiral sulfinamide auxiliary, which directs the formation of the new carbon-phosphorus bond with high diastereoselectivity. Following the addition, the reaction mixture is quenched with ammonium chloride and extracted, yielding the intermediate sulfinamide compounds (II-a to II'-g) in yields ranging from 39% to 46%. This step establishes the foundational stereochemistry of the side chain, which is crucial for the subsequent catalytic performance. The use of Schlenk techniques throughout this process underscores the sensitivity of the intermediates to moisture and oxygen, necessitating strict inert atmosphere protocols during manufacturing.
The second pivotal transformation is the desulfurization of the phosphine sulfide moiety to generate the active trivalent phosphine species. This is achieved using Raney Nickel, a cost-effective and widely available hydrogenation catalyst, in a mixture of anhydrous ethanol and tetrahydrofuran. 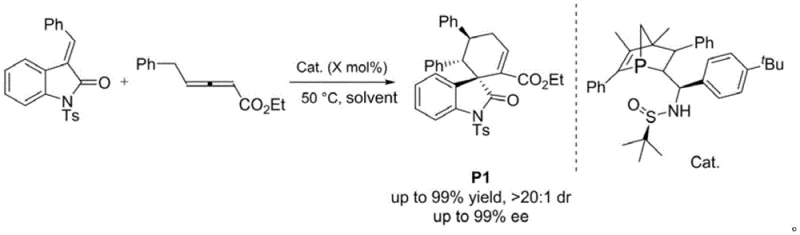 The removal of the sulfur atom is a critical activation step that converts the stable pentavalent phosphorus sulfide into the nucleophilic trivalent phosphine required for catalysis. This step proceeds with excellent efficiency, delivering the final catalysts (III-a to III'-g) in yields between 71% and 87%. The resulting trivalent phosphorus center is now free to act as a nucleophilic catalyst, attacking electron-deficient alkenes to initiate asymmetric transformations. In practical applications, such as the (4+2) cycloaddition of substituted allenoates with alkenyl oxindoles, these catalysts demonstrate exceptional performance, achieving yields above 99% with enantiomeric excess values exceeding 99% ee. This high level of stereocontrol validates the design hypothesis that a rigid framework effectively transmits chiral information to the reaction center.
The removal of the sulfur atom is a critical activation step that converts the stable pentavalent phosphorus sulfide into the nucleophilic trivalent phosphine required for catalysis. This step proceeds with excellent efficiency, delivering the final catalysts (III-a to III'-g) in yields between 71% and 87%. The resulting trivalent phosphorus center is now free to act as a nucleophilic catalyst, attacking electron-deficient alkenes to initiate asymmetric transformations. In practical applications, such as the (4+2) cycloaddition of substituted allenoates with alkenyl oxindoles, these catalysts demonstrate exceptional performance, achieving yields above 99% with enantiomeric excess values exceeding 99% ee. This high level of stereocontrol validates the design hypothesis that a rigid framework effectively transmits chiral information to the reaction center.
How to Synthesize 1-Phospha Norbornene Chiral Phosphine Catalyst Efficiently
The preparation of these high-value catalysts follows a streamlined protocol that balances stereochemical control with operational simplicity. The process begins with the careful handling of air-sensitive reagents under nitrogen atmosphere, utilizing standard Schlenk line techniques to ensure reproducibility. The initial Grignard addition requires precise temperature management to avoid thermal degradation of the chiral auxiliary, while the subsequent desulfurization step leverages the robust nature of Raney Nickel to drive the reaction to completion. Detailed standardized synthesis steps are provided in the guide below to assist process chemists in replicating these results at scale.
- React the phosphine sulfide imine precursor (Compound I) with aryl Grignard reagents at -78°C in anhydrous THF to form the intermediate sulfinamide (Compound II).
- Purify the intermediate via column chromatography after quenching with ammonium chloride and extraction.
- Perform desulfurization using Raney Nickel in anhydrous ethanol/THF to yield the final trivalent phosphine catalyst (Compound III).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial benefits that extend beyond mere technical performance, addressing key pain points in the supply chain for chiral catalysts. The reliance on commodity chemicals and standard unit operations significantly lowers the barrier to entry for large-scale production, making it an attractive option for cost-sensitive manufacturing environments.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal complexes often required in traditional cross-coupling or hydrogenation catalysts. By utilizing organocatalysis based on phosphorus, the process avoids the stringent regulatory requirements and costly removal steps associated with heavy metal residues in pharmaceutical products. Furthermore, the use of Raney Nickel for desulfurization is a well-established, low-cost industrial process that does not require specialized noble metals like palladium or rhodium. This substitution of precious metals with base metals and organic frameworks leads to a drastic simplification of the bill of materials and a corresponding reduction in raw material expenditures.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as cinnamaldehyde derivatives and common Grignard reagents, are globally sourced commodities with stable supply chains. Unlike specialized chiral ligands that may depend on single-source suppliers or complex natural product extraction, the precursors for these 1-phospha norbornene catalysts can be produced by multiple chemical manufacturers. This diversification of the supply base mitigates the risk of shortages and price volatility, ensuring consistent availability for continuous manufacturing campaigns. Additionally, the intermediates are stable enough to be stored and transported under standard conditions, reducing the logistical complexities associated with cold chain management.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in batch reactors using standard glassware that can be directly translated to stainless steel vessels. The absence of toxic heavy metals in the final catalyst structure simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The high atom economy of the Diels-Alder construction step and the efficient desulfurization process minimize waste generation, supporting green chemistry initiatives. For supply chain heads, this means faster regulatory approval times and reduced costs associated with environmental compliance and waste management, facilitating a smoother path from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and potential applications for prospective partners.
Q: Why is the 1-phospha norbornene framework superior for P-chiral catalysts?
A: The rigid bicyclic norbornene skeleton locks the phosphorus atom in place, forming three completely bound P-C bonds. This structural rigidity successfully prevents the thermal racemization that typically plagues flexible P-chiral centers, ensuring high stereochemical stability during reactions.
Q: What are the typical yields and selectivity for this catalyst system?
A: The synthesis of the catalyst intermediates proceeds with yields ranging from 39% to 46% for the Grignard addition step, followed by 71% to 87% for the desulfurization step. In application, such as the (4+2) cycloaddition of substituted allenoates, the catalyst achieves yields above 99% with diastereoselectivity greater than 20:1 and enantioselectivity exceeding 99% ee.
Q: Is this catalyst suitable for large-scale pharmaceutical manufacturing?
A: Yes, the synthesis utilizes commercially available starting materials like cinnamaldehyde and standard reagents such as Grignard reagents and Raney Nickel. The process avoids exotic transition metals in the final catalytic step and uses standard purification techniques like column chromatography, making it highly amenable to scale-up for industrial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phospha Norbornene Catalyst Supplier
The development of 1-phospha norbornene chiral phosphine catalysts represents a significant leap forward in the field of asymmetric organocatalysis, offering a robust solution for the synthesis of complex chiral intermediates. NINGBO INNO PHARMCHEM stands at the forefront of this technological advancement, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of catalyst delivered meets the highest standards of quality and performance required by the global pharmaceutical industry.
We invite you to collaborate with us to explore how this novel catalyst system can optimize your existing synthetic routes. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific process needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your journey from discovery to commercial success.
