Advanced Synthesis of Fluorinated Oxime Ethers for Scalable Pharmaceutical Intermediate Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures with high efficiency and minimal environmental impact. Patent CN110698393A introduces a groundbreaking approach to the synthesis of novel fluorine-containing oxime ethers, specifically structures designated as I through V. These compounds are not merely incremental improvements but represent a fundamental shift in how radical precursors are generated and utilized in organic synthesis. The core innovation lies in the incorporation of a pentafluoropyridine moiety, which acts as a powerful electron-withdrawing group. This structural feature dramatically lowers the bond dissociation energy of the nitrogen-oxygen (N-O) bond, allowing for facile homolysis under either thermal or photocatalytic conditions. For R&D directors and process chemists, this patent offers a versatile toolkit for generating carbon-centered radicals without the reliance on stoichiometric oxidants or toxic transition metals, paving the way for greener and more scalable synthetic routes for active pharmaceutical ingredients (APIs) and advanced intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the activation of C-H bonds for alkylation purposes has heavily relied on transition metal catalysis, often involving precious metals such as palladium, rhodium, or iridium. While effective, these conventional methods present significant challenges for large-scale manufacturing. The primary concern is the residual metal content in the final product, which requires rigorous and expensive purification processes to meet stringent regulatory standards for pharmaceutical applications. Furthermore, many traditional radical generation methods utilize hazardous reagents or harsh reaction conditions that pose safety risks and generate substantial chemical waste. The reliance on stoichiometric amounts of metal catalysts or toxic radical initiators also complicates the supply chain, introducing volatility in raw material costs and availability. Additionally, the functional group tolerance in these older methods is often limited, restricting the structural diversity that can be achieved in a single synthetic step, thereby necessitating longer linear synthesis sequences that reduce overall yield and increase production time.
The Novel Approach
In stark contrast, the methodology disclosed in CN110698393A leverages the unique electronic properties of the tetrafluoropyridyl group to enable a metal-free or photocatalytic radical relay mechanism. This novel approach allows for the generation of reactive radical species under remarkably mild conditions, typically at room temperature for the precursor synthesis and under visible light irradiation for the subsequent coupling reactions. The process utilizes readily available starting materials such as oximes and pentafluoropyridine, reacting them in the presence of a mild base like potassium carbonate. This simplifies the operational complexity significantly, as it avoids the need for inert atmosphere gloveboxes or cryogenic temperatures often required for sensitive organometallic reagents. The resulting fluorinated oxime ethers are stable enough to be isolated and stored, yet reactive enough to serve as efficient radical precursors when triggered, offering a level of control and modularity that is highly desirable for process optimization.

The versatility of this system is further demonstrated by its ability to accommodate various structural motifs, including cyclic and acyclic oximes, allowing for the introduction of diverse alkyl chains into target molecules. This flexibility is crucial for medicinal chemists exploring structure-activity relationships (SAR) during drug discovery phases.
Mechanistic Insights into Photocatalytic Radical Relay Activation
The mechanistic elegance of this technology centers on the homolytic cleavage of the N-O bond, which is activated by the strong electron-withdrawing nature of the -C5F4N group. Upon exposure to thermal energy or visible light in the presence of a photocatalyst such as Ir(ppy)3, the N-O bond undergoes homolysis to generate an iminyl radical intermediate. For cyclic oxime ethers like structures I through IV, the inherent ring strain provides an additional thermodynamic driving force for the subsequent ring-opening reaction. This cascade event releases the ring tension and generates a distal cyanoalkyl radical, which is a highly valuable synthon for constructing nitrile-containing pharmacophores. The nitrile group is a privileged motif in medicinal chemistry, often serving as a bioisostere for other functional groups or participating in critical binding interactions within biological targets. The ability to install this group via a radical mechanism expands the chemical space accessible to synthetic chemists beyond what is possible with traditional nucleophilic substitutions.
For acyclic variants such as structure V, the mechanism diverges slightly but remains equally powerful. Following the initial N-O bond cleavage, the resulting iminyl radical undergoes a 1,5-hydrogen atom transfer (HAT) process. This intramolecular migration generates a carbon-centered alkyl radical at a specific position along the chain, dictated by the geometry of the transition state. This remote functionalization capability is particularly noteworthy, as it allows for the modification of unactivated C-H bonds that are typically inert to standard chemical reagents. The generated alkyl radical can then engage in addition reactions with electron-deficient olefins or heteroaromatic systems, such as the quinoxalinone derivatives shown in the patent examples. This radical relay strategy effectively bypasses the need for pre-functionalized halides or organometallic reagents, streamlining the synthesis of complex molecules and reducing the number of synthetic steps required to reach the final target.
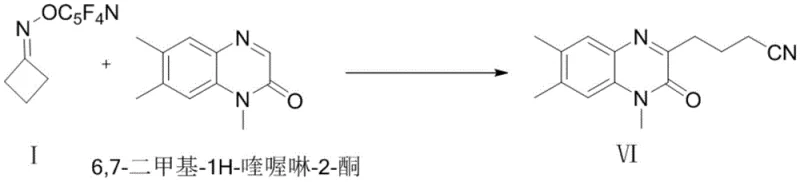
The successful application of this mechanism is evidenced by the efficient synthesis of quinoxaline derivatives like compound VI, where the cyanoalkyl chain is installed directly onto the heterocyclic core with high regioselectivity. This level of precision is essential for ensuring the purity and efficacy of the final pharmaceutical product.
How to Synthesize Fluorinated Oxime Ethers Efficiently
The synthesis of these valuable intermediates is designed to be operationally simple and scalable, making it suitable for both laboratory discovery and industrial production. The process begins with the deprotonation of the oxime hydroxyl group using a mild inorganic base, followed by nucleophilic aromatic substitution with pentafluoropyridine. This reaction proceeds smoothly in polar aprotic solvents like acetonitrile at ambient temperature, eliminating the need for energy-intensive heating or cooling cycles. The workup procedure involves standard aqueous extraction and chromatographic purification, techniques that are universally available in chemical manufacturing facilities. For downstream applications, the purified oxime ethers can be employed in radical coupling reactions simply by mixing them with the desired substrate and a photocatalyst under LED irradiation. This modular approach allows chemists to rapidly assemble diverse libraries of compounds for biological screening.
- Mix the raw material oxime with potassium carbonate in acetonitrile solvent at room temperature and stir for 2-3 hours.
- Add pentafluoropyridine to the mixture and continue stirring at room temperature for 11-12 hours to complete the substitution reaction.
- Extract the reaction mixture with ethyl acetate and saturated sodium chloride, then purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this fluorinated oxime ether technology offers compelling advantages that directly impact the bottom line and operational resilience. The elimination of expensive and scarce transition metal catalysts represents a significant shift in cost structure. Traditional noble metal catalysts are subject to volatile market pricing and geopolitical supply risks; by replacing them with organic photocatalysts or thermal conditions, manufacturers can stabilize their raw material costs and reduce dependency on critical mineral supply chains. Furthermore, the removal of heavy metals from the process flow drastically simplifies the purification workflow. In conventional API manufacturing, metal scavenging resins and extensive recrystallization steps are often required to meet strict ppm limits for metal residues. By avoiding these metals entirely, the new method reduces the consumption of auxiliary materials, shortens cycle times, and minimizes waste generation, leading to substantial cost savings in both materials and disposal.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the synthetic route and the reduction in auxiliary material consumption. By utilizing inexpensive reagents like potassium carbonate and acetonitrile instead of costly organometallic complexes, the direct material cost per kilogram of product is significantly lowered. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to lower utility costs. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproducts. This efficiency translates directly into improved margins for high-volume production, making the technology attractive for cost-sensitive generic drug manufacturing as well as high-value specialty chemicals.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for pharmaceutical manufacturers, and this technology enhances reliability by relying on commodity chemicals rather than specialized reagents. Pentafluoropyridine and simple oximes are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions also means that the process is less susceptible to variations in environmental conditions or minor fluctuations in reagent quality, ensuring consistent batch-to-batch performance. This reliability allows supply chain managers to forecast production timelines with greater accuracy and maintain leaner inventory levels without compromising on delivery commitments to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden challenges, but this radical relay method is inherently scalable due to its homogeneous nature and lack of exothermic hazards associated with strong oxidants. The use of visible light photocatalysis is particularly amenable to continuous flow processing, a technology that is increasingly favored for safe and efficient large-scale manufacturing. From an environmental standpoint, the process aligns with green chemistry principles by minimizing waste and avoiding toxic heavy metals. This compliance simplifies the regulatory approval process for new drug filings and reduces the environmental footprint of the manufacturing facility, which is increasingly important for meeting corporate sustainability goals and regulatory requirements in major markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this pathway for their own production needs. Understanding these details is crucial for assessing the feasibility of integrating this chemistry into existing manufacturing workflows.
Q: What is the primary chemical advantage of the -C5F4N group in these oxime ethers?
A: The tetrafluoropyridyl (-C5F4N) group possesses strong electron-withdrawing capabilities. This electronic effect significantly weakens the N-O bond, facilitating homolytic cleavage under mild heating or photocatalytic conditions to generate reactive iminyl or alkyl radicals efficiently.
Q: How does this method improve upon traditional transition metal catalysis?
A: This protocol utilizes a radical relay mechanism that replaces traditional transition metal catalysts. By avoiding heavy metals, the process eliminates the need for complex and costly metal removal steps, resulting in a cleaner impurity profile and reduced environmental impact for pharmaceutical manufacturing.
Q: What types of chemical transformations can these intermediates facilitate?
A: These fluorinated oxime ethers serve as versatile radical precursors. They can undergo ring-opening reactions to form cyanoalkyl radicals or 1,5-hydrogen migrations to form alkyl radicals, enabling the efficient C-H alkylation of heterocycles like quinoxalinones and functionalization of olefins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Oxime Ether Supplier
As the demand for advanced synthetic methodologies grows, partnering with an experienced CDMO becomes essential for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our technical team is well-versed in handling fluorine chemistry and photocatalytic processes, equipped with state-of-the-art reactors and rigorous QC labs to guarantee stringent purity specifications for every batch. We understand the critical nature of supply chain stability and are committed to providing consistent, high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to explore the potential of this technology for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us collaborate to optimize your synthesis strategy, reduce your time to market, and achieve your commercial objectives with confidence and efficiency.
