Advanced Green Synthesis of Beta-Cyano Ketones for Commercial Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving towards greener, safer, and more economically viable pathways, particularly in the production of complex pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN114456086A, which details a novel method for the synthesis of beta-cyano ketones. This class of compounds serves as a critical structural motif in numerous bioactive molecules, acting as versatile precursors for amides, amines, and heterocycles essential in modern drug discovery. The patented technology fundamentally shifts the paradigm by replacing hazardous traditional cyanating agents with acetone cyanohydrin, a reagent that combines operational safety with high atom economy. For R&D directors and procurement specialists seeking reliable supply chains for high-value intermediates, this innovation represents a substantial leap forward in process chemistry, offering a route that mitigates environmental risks while maintaining exceptional product purity standards exceeding 99%.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group into an alpha,beta-unsaturated carbonyl system has been fraught with significant safety and economic challenges. Traditional methodologies frequently rely on hydrogen cyanide (HCN) or inorganic metal cyanides such as sodium cyanide and potassium cyanide. These reagents are notoriously volatile and acutely toxic, necessitating rigorous containment protocols and specialized infrastructure that drastically inflate capital expenditure and operational costs. Furthermore, the use of metal cyanides generates stoichiometric amounts of heavy metal waste, creating a severe burden on wastewater treatment facilities and complicating regulatory compliance for environmental discharge. Alternatively, while reagents like trimethylsilyl cyanide (TMSCN) offer improved safety profiles, their prohibitive cost and poor atom utilization render them unsuitable for cost-sensitive commercial manufacturing. These legacy methods often suffer from harsh reaction conditions and prolonged reaction times, leading to inconsistent yields and complex purification workflows that hinder scalability.
The Novel Approach
In stark contrast, the methodology outlined in CN114456086A utilizes acetone cyanohydrin as a benign and efficient cyanide source, activated by lithium hydroxide in a mixed solvent system of water and DMF. This approach elegantly circumvents the toxicity issues associated with HCN and the waste generation of metal salts. The reaction proceeds under mild thermal conditions, typically around 60°C, and demonstrates remarkable tolerance for a wide array of functional groups on the aromatic rings of the chalcone substrates. As illustrated in the reaction scope below, this protocol consistently delivers high yields across diverse electronic environments, from electron-donating methoxy groups to electron-withdrawing halogens.
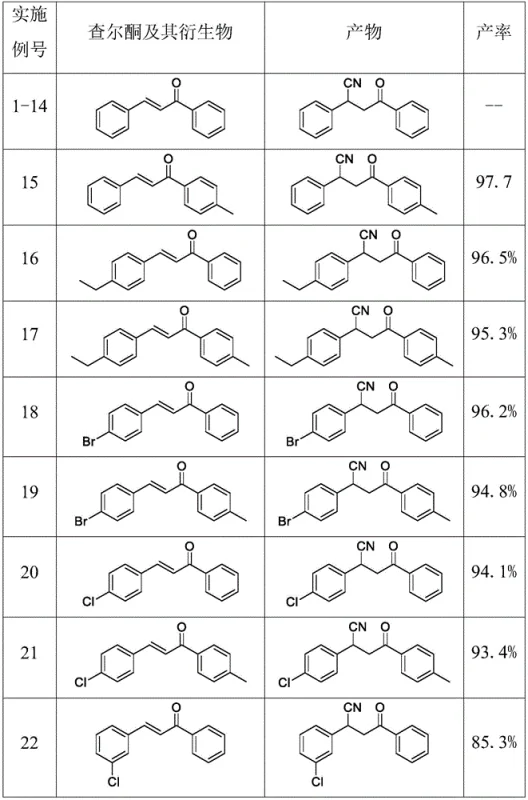
The data presented confirms the robustness of this synthetic strategy, with isolated yields frequently surpassing 95% for substituted chalcones. The byproduct of the reaction is acetone, a common solvent that is non-toxic and easily removed during workup, thereby simplifying downstream processing. This transition to a greener reagent system not only enhances the safety profile of the manufacturing process but also aligns with the increasing global demand for sustainable chemical production practices, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into LiOH-Catalyzed Michael Addition
The core of this synthetic advancement lies in the mechanistic efficiency of the lithium hydroxide-mediated Michael addition. In this catalytic cycle, lithium hydroxide acts as a mild yet effective base to deprotonate the acetone cyanohydrin, generating a reactive cyanide anion in situ without the need for anhydrous or oxygen-free conditions. This activation step is crucial as it lowers the energy barrier for the nucleophilic attack on the beta-carbon of the chalcone double bond. The use of a water/DMF co-solvent system is particularly ingenious; water facilitates the dissolution of the inorganic base, while DMF ensures the solubility of the organic substrates, creating a homogeneous reaction environment that maximizes molecular collisions and reaction kinetics. This biphasic-mimicking homogeneity allows for rapid mass transfer, which is often a limiting factor in heterogeneous cyanation reactions involving solid metal cyanides.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of heavy metal catalysts eliminates the risk of metal leaching into the final product, a critical quality attribute for pharmaceutical intermediates intended for human consumption. Consequently, the need for expensive and time-consuming metal scavenging steps is entirely removed. Furthermore, the mild basicity of lithium hydroxide minimizes side reactions such as polymerization of the alpha,beta-unsaturated system or hydrolysis of the nitrile group, which are common pitfalls in harsher alkaline conditions. The result is a cleaner crude reaction profile that facilitates straightforward purification via flash column chromatography or recrystallization, consistently achieving purity levels greater than 99% as verified by NMR and HPLC analysis.
How to Synthesize Beta-Cyano Ketone Efficiently
Implementing this synthesis requires precise control over reagent stoichiometry and solvent ratios to maximize efficiency. The protocol dictates dissolving lithium hydroxide in a specific volume of deionized water, followed by the addition of N,N-dimethylformamide to create the optimal mixed solvent environment. Acetone cyanohydrin is introduced first to allow for adequate activation before the addition of the chalcone substrate. The reaction is then heated to maintain a steady 60°C, ensuring complete conversion while preventing thermal degradation. Detailed standardized operating procedures regarding specific molar equivalents and workup sequences are provided in the technical documentation below.
- Dissolve lithium hydroxide in deionized water and mix with N,N-dimethylformamide (DMF) to form a homogeneous solvent system.
- Add acetone cyanohydrin to the mixture and stir at room temperature for 30 minutes to activate the cyanide source.
- Introduce the chalcone substrate, heat the reaction to 60°C, and monitor until completion before standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic reduction of raw material costs and hazard management expenses. By substituting expensive reagents like TMSCN or highly regulated toxic cyanides with commodity-grade acetone cyanohydrin, the direct material cost of goods sold (COGS) is significantly optimized. Additionally, the elimination of heavy metal waste streams reduces the financial burden associated with hazardous waste disposal and environmental compliance auditing. This streamlined waste profile not only lowers operational expenditures but also accelerates the timeline for regulatory approvals in different jurisdictions, facilitating faster market entry for new drug candidates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials. Acetone cyanohydrin is produced on a massive industrial scale, ensuring stable pricing and availability, unlike specialized silyl reagents which are subject to supply chain volatility. Furthermore, the simplified workup procedure, which avoids complex extraction steps required for metal salt removal, reduces labor hours and solvent consumption. The high atom economy of the reaction means that less raw material is wasted, directly contributing to a leaner and more cost-effective manufacturing process that enhances overall profit margins for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on non-restricted, dual-use chemical precursors. Unlike hydrogen cyanide or certain metal cyanides, which are subject to strict transportation regulations and security clearances, acetone cyanohydrin can be sourced and shipped with fewer logistical hurdles. This ease of logistics ensures consistent raw material flow, minimizing the risk of production stoppages due to regulatory delays. The robustness of the reaction conditions also implies that the process is less sensitive to minor variations in utility supplies, such as cooling water temperature fluctuations, further stabilizing the production schedule and ensuring on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is seamless due to the mild thermal requirements and the use of standard solvent systems. The reaction does not require cryogenic cooling or high-pressure equipment, allowing it to be run in existing glass-lined or stainless steel reactors without significant capital investment. From an environmental standpoint, the generation of acetone as the sole organic byproduct aligns perfectly with green chemistry principles, simplifying solvent recovery and recycling loops. This sustainability profile is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies who prioritize suppliers with strong environmental, social, and governance (ESG) credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-cyano ketone synthesis platform. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on process parameters and scalability potential. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is acetone cyanohydrin preferred over traditional cyanide sources?
A: Acetone cyanohydrin offers significantly lower toxicity and volatility compared to hydrogen cyanide or metal cyanides, while being more cost-effective than trimethylsilyl cyanide (TMSCN), with acetone as the only non-toxic byproduct.
Q: What represents the optimal reaction temperature for this synthesis?
A: While the reaction proceeds between 20°C and 100°C, experimental data indicates that 60°C provides the optimal balance between reaction rate and yield, often achieving over 95% conversion.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of inexpensive reagents, mild conditions, and the elimination of heavy metal waste makes this protocol highly amenable to commercial scale-up for API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Cyano Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends heavily on the reliability and quality of the supply chain. We have extensively evaluated the synthetic pathway described in CN114456086A and possess the technical expertise to execute this green chemistry protocol at an industrial level. Our facility is equipped to handle the specific solvent systems and thermal requirements of this reaction, ensuring that we can deliver high-purity beta-cyano ketones that meet stringent pharmacopeial standards. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we offer a partnership model that guarantees supply continuity and consistent quality for your critical drug development programs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to validate the feasibility of this route for a new molecule, our experts are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
