Advanced Copper-Catalyzed Cascade Synthesis for Complex Nitrogen Heterocyclic Scaffolds
Introduction to Patent CN102140096A Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex nitrogen-containing heterocyclic frameworks, which serve as critical backbones for numerous bioactive molecules. Patent CN102140096A introduces a groundbreaking synthetic methodology that addresses these needs through a highly efficient copper-catalyzed cascade reaction. This technology enables the rapid assembly of pyrrolo[3,2-c]quinoline and indeno[1,2-b]pyrrole skeletons directly from readily available N-(2-halophenyl)alkynamides or analogous ketone precursors and isocyano compounds. The significance of this invention lies in its ability to perform these transformations under relatively mild conditions using inexpensive copper salts, thereby offering a distinct advantage over traditional palladium-catalyzed processes which often suffer from higher costs and metal residue issues. For R&D directors and process chemists, this patent represents a valuable tool for accelerating the discovery and development of new drug candidates by providing a robust route to privileged structures.
Furthermore, the reaction kinetics described in the patent are exceptionally fast, with many transformations reaching completion within merely ten minutes at temperatures ranging from 80°C to 100°C. This rapid turnover not only enhances laboratory throughput but also suggests significant potential for energy savings during large-scale manufacturing. The method utilizes common organic solvents such as DMF or DMSO and employs bases like cesium carbonate or potassium carbonate, which are standard reagents in industrial settings. By streamlining the synthesis of these complex polycyclic systems into a single operational step, the technology minimizes waste generation and reduces the overall environmental footprint of the production process. This aligns perfectly with the growing global demand for greener chemistry solutions in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fused nitrogen heterocycles like pyrroloquinolines has relied on multi-step sequences that often involve harsh reaction conditions, expensive noble metal catalysts, and tedious purification protocols. Traditional approaches might require the pre-functionalization of substrates, protection and deprotection steps for sensitive amine groups, or the use of stoichiometric amounts of toxic reagents. These conventional pathways frequently result in low overall yields due to cumulative losses at each stage, making them economically unviable for commercial scale-up. Additionally, the reliance on palladium or rhodium catalysts introduces the challenge of removing trace heavy metals from the final active pharmaceutical ingredient (API), a regulatory hurdle that adds significant time and cost to the manufacturing timeline. The slow reaction rates associated with older methodologies further exacerbate production bottlenecks, limiting the ability of supply chains to respond quickly to market demands.
The Novel Approach
In stark contrast, the novel approach detailed in CN102140096A utilizes a tandem cyclization strategy driven by earth-abundant copper catalysts. This method bypasses the need for multiple isolation steps by constructing two rings simultaneously in a single pot. The reaction tolerates a wide array of substituents on both the alkyne and the isocyanide components, allowing for the rapid generation of diverse chemical libraries for structure-activity relationship (SAR) studies. As illustrated in the reaction scheme below, the transformation proceeds smoothly with various alkyl and aryl groups attached to the alkyne terminus, demonstrating remarkable versatility.
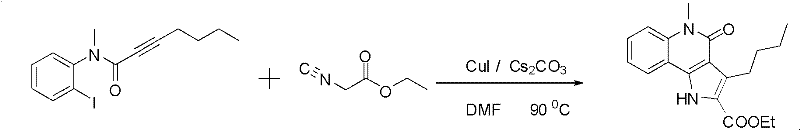
The use of simple copper salts like CuI or CuBr significantly lowers the raw material costs compared to precious metal alternatives. Moreover, the reaction conditions are optimized to proceed rapidly, often completing in as little as ten minutes, which drastically reduces energy consumption per batch. This efficiency translates directly into improved process economics, making it an attractive option for procurement managers looking to optimize the cost of goods sold (COGS). The simplicity of the workup procedure, typically involving standard extraction and column chromatography, further enhances the practicality of this method for both small-scale discovery and larger pilot plant operations.
Mechanistic Insights into Copper-Catalyzed Cascade Cyclization
The underlying mechanism of this transformation involves a sophisticated interplay between the copper catalyst, the halogenated substrate, and the isocyanide functionality. It is hypothesized that the copper species initially activates the carbon-halogen bond of the N-(2-iodophenyl)alkynamide, facilitating an intramolecular carbocupration or a similar activation of the alkyne moiety. Subsequently, the nucleophilic attack by the isocyanide carbon onto the activated intermediate triggers a cascade of cyclization events. This sequence effectively builds the pyrrole ring fused to the existing quinoline or indene framework. The choice of base, such as Cs2CO3, plays a crucial role in deprotonating intermediates and driving the equilibrium towards the final aromatized product. Understanding this mechanistic pathway allows chemists to fine-tune reaction parameters, such as ligand selection or temperature, to maximize yields for specific substrate classes.
Beyond the core mechanism, the patent highlights the exceptional functional group tolerance of this catalytic system, which is a critical factor for R&D teams designing complex molecules. The reaction successfully accommodates electron-withdrawing groups like cyano (-CN) and trifluoromethyl (-CF3) on the aromatic ring, as well as electron-donating groups like methoxy (-OMe). Furthermore, the methodology is not limited to amide substrates; it extends effectively to ketone-based precursors to synthesize indeno[1,2-b]pyrroles, as shown in the following example. This versatility ensures that the process can be adapted for a wide range of target molecules without requiring extensive re-optimization, thereby reducing development timelines.
![Synthesis of indeno[1,2-b]pyrrole derivatives using ketone substrates and isocyanides](/insights/img/nitrogen-heterocycles-copper-catalysis-pharma-supplier-20260305064603-017.png)
Impurity control is another area where this method excels. The high selectivity of the copper-catalyzed cascade minimizes the formation of side products such as homocoupling dimers or incomplete cyclization intermediates. The clean reaction profiles observed in the experimental data suggest that the transition state is well-defined and energetically favorable. For quality control departments, this means simpler purification processes and higher purity specifications for the final intermediates. The ability to achieve high yields (often exceeding 80% and reaching up to 95% in optimized cases) with minimal byproduct formation underscores the robustness of this chemistry for GMP manufacturing environments where consistency and purity are paramount.
How to Synthesize Pyrrolo[3,2-c]quinoline Efficiently
To implement this synthesis in a laboratory or pilot setting, operators should follow a standardized protocol that ensures safety and reproducibility. The process begins with the precise weighing of the N-(2-iodophenyl)alkynamide substrate and the isocyano compound, typically ethyl isocyanoacetate, in a molar ratio that favors complete conversion of the limiting reagent. The reaction is conducted under an inert atmosphere, such as argon, to prevent oxidation of the copper catalyst or the sensitive isocyanide species. Detailed standard operating procedures regarding the addition order of reagents, heating ramps, and quenching methods are essential for maintaining batch-to-batch consistency. Please refer to the specific synthesis guide below for the step-by-step technical instructions.
- Combine N-(2-iodophenyl)-N-methylalkynamide substrate with ethyl isocyanoacetate, cuprous iodide catalyst (1-20 mol%), and cesium carbonate base in DMF solvent.
- Heat the sealed reaction mixture to 80-100°C for approximately 10 minutes to facilitate the cascade cyclization.
- Quench the reaction, extract with ethyl acetate, dry over sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed technology offers substantial strategic benefits for procurement and supply chain management. The primary advantage stems from the drastic simplification of the synthetic route, which eliminates multiple unit operations associated with traditional multi-step syntheses. By consolidating the ring-forming steps into a single reaction vessel, manufacturers can significantly reduce labor costs, solvent usage, and reactor occupancy time. This streamlined workflow directly contributes to cost reduction in API manufacturing, allowing companies to remain competitive in price-sensitive markets. Furthermore, the use of non-precious metal catalysts removes the dependency on volatile precious metal markets, stabilizing raw material costs and mitigating supply risks associated with scarce resources like palladium.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive copper salts like CuI results in immediate savings on catalyst procurement. Additionally, the short reaction times (approximately 10 minutes) lead to massive reductions in energy consumption for heating and stirring, lowering the utility costs per kilogram of product. The high atom economy and yield of the reaction minimize the amount of starting material wasted, further optimizing the material cost structure. These factors combine to create a highly economical process that improves the overall margin profile for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as substituted 2-iodoanilines, propiolic acids, and isocyano esters, are commercially available from multiple global suppliers. This diversity in the supply base reduces the risk of single-source bottlenecks and ensures continuity of supply even during market disruptions. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, enhancing the reliability of production schedules. Consequently, supply chain managers can plan inventory levels with greater confidence and reduce the need for excessive safety stock.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the use of common solvents like DMF make this process highly scalable from gram to ton quantities without significant engineering challenges. The reduced generation of hazardous waste due to fewer synthetic steps aligns with increasingly stringent environmental regulations and corporate sustainability goals. Easier waste treatment and lower emissions contribute to a smaller environmental footprint, facilitating smoother regulatory approvals and community relations for manufacturing sites. This scalability ensures that the technology can support the transition from clinical trial materials to full commercial production seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances helps in making informed decisions about process adoption and optimization strategies.
Q: What represents the optimal catalytic system for this heterocycle synthesis?
A: The patent data indicates that Cuprous Iodide (CuI) combined with Cesium Carbonate (Cs2CO3) in DMF provides superior yields compared to other copper salts or bases, operating efficiently at temperatures between 80°C and 100°C.
Q: Does this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the methodology demonstrates robust functional group tolerance, successfully accommodating electron-withdrawing groups like cyano (-CN) and trifluoromethyl (-CF3), as well as electron-donating groups like methoxy (-OMe) and methyl (-Me).
Q: Can this protocol be adapted for synthesizing indeno-pyrrole scaffolds?
A: Absolutely. By substituting the amide linkage with a ketone linkage in the starting alkyne material, the same copper-catalyzed conditions effectively construct the indeno[1,2-b]pyrrole backbone with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolo[3,2-c]quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102140096A for accelerating drug development pipelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nitrogen heterocycles adheres to the highest industry standards. We are committed to leveraging our technical expertise to bring complex synthetic routes to life efficiently and safely.
We invite you to collaborate with us to explore how this efficient copper-catalyzed synthesis can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data for similar scaffolds and discuss route feasibility assessments for your upcoming programs. Let us be your trusted partner in turning innovative chemistry into commercial success.
