Advanced Iridium-Catalyzed Synthesis of N-Phenyl-7-Azaindole Derivatives for Commercial Scale
Advanced Iridium-Catalyzed Synthesis of N-Phenyl-7-Azaindole Derivatives for Commercial Scale
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those featuring the 7-azaindole core, which is prevalent in numerous bioactive molecules targeting cancer, bacterial infections, and diabetes. A significant breakthrough in this domain is detailed in Chinese Patent CN113461681A, which discloses a highly efficient iridium-catalyzed preparation method for N-phenyl-7-azaindole derivatives. This technology leverages the unique directing ability of the 7-azaindole nitrogen to facilitate precise C-H amination on the N-phenyl ring. By utilizing specialized iridium complexes in conjunction with ionic liquids, the process achieves superior conversion rates and selectivity under remarkably mild conditions. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of N-aryl-7-azaindoles has relied heavily on transition metal catalysis, yet existing protocols often suffer from significant inefficiencies that hinder commercial viability. Prior art, such as rhodium-catalyzed systems reported in earlier literature, frequently necessitates harsh reaction conditions, including elevated temperatures and extended reaction times, which can lead to thermal degradation of sensitive substrates. Furthermore, these conventional methods often struggle with poor regioselectivity, resulting in complex mixtures of C-N and C-C coupled products that require extensive and costly downstream purification. The reliance on volatile organic solvents in these traditional processes also exacerbates environmental concerns and increases waste disposal costs, creating a substantial burden for supply chain managers aiming to meet increasingly stringent green chemistry standards.
The Novel Approach
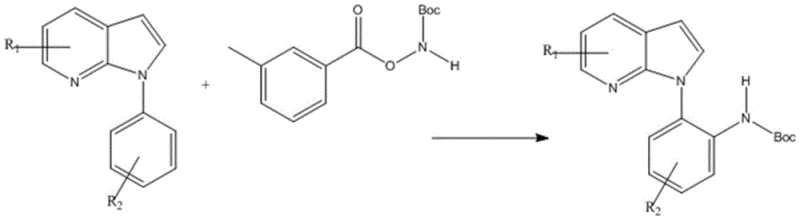
The innovative methodology described in the patent overcomes these hurdles by employing a sophisticated iridium catalytic system combined with task-specific ionic liquids. As illustrated in the general reaction scheme below, the process utilizes N-phenyl-7-azaindole and acyloxy carbamate compounds as key starting materials. The use of ionic liquids such as 1-butyl-3-methylimidazolium tetrafluoroborate (BMIMBF4) not only serves as a green solvent but also actively participates in lowering the activation energy barrier of the C-H bond. This synergistic effect allows the reaction to proceed efficiently at temperatures as low as 25-30°C, drastically reducing energy consumption. The result is a streamlined synthesis that delivers high-purity N-phenyl-7-azaindole derivatives with excellent yields, providing a reliable pathway for the production of complex drug candidates.
Mechanistic Insights into Iridium-Catalyzed C-H Amination
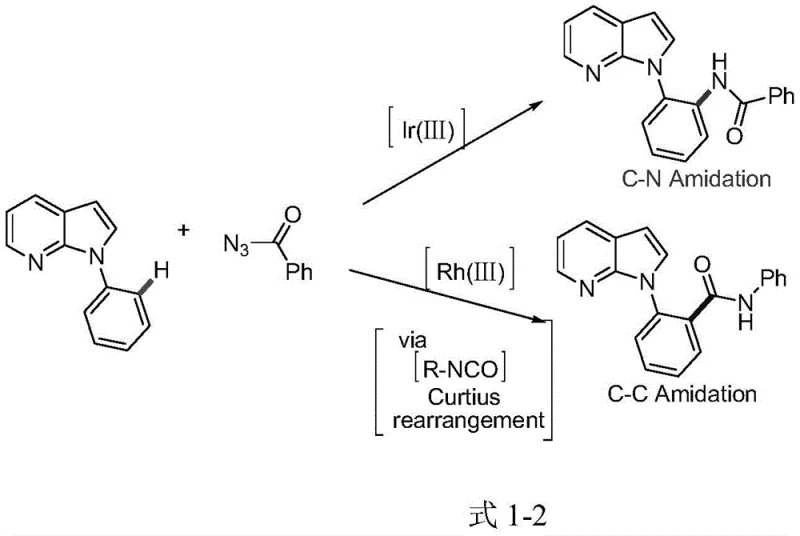
The success of this transformation lies in the precise mechanistic interplay between the iridium catalyst, the silver salt additive, and the 7-azaindole directing group. The 7-azaindole moiety acts as a powerful bidentate ligand, coordinating with the iridium center to form a stable cyclometalated intermediate. This coordination directs the metal specifically to the ortho-C-H bond of the N-phenyl ring, ensuring high regioselectivity for the subsequent amination step. Unlike rhodium systems, which may diverge into C-C bond formation pathways via Curtius rearrangement intermediates, the iridium catalyst favors the formation of the C-N bond directly. This mechanistic divergence is critical for pharmaceutical synthesis, where structural fidelity is paramount. The inclusion of silver salts, such as AgSbF6 or AgOTf, further enhances catalytic activity by abstracting halide ligands from the iridium precursor, generating a highly electrophilic cationic iridium species that is more reactive towards C-H activation.
Furthermore, the choice of the aminating reagent, specifically the acyloxy carbamate, plays a vital role in the reaction kinetics. Upon activation by the iridium catalyst, this reagent generates a reactive nitrenoid species that inserts efficiently into the activated C-H bond. The stability of this intermediate is finely tuned by the electronic properties of the acyloxy group, preventing premature decomposition and ensuring smooth conversion to the final carbamate-protected amine product. This level of control minimizes the formation of side products and impurities, simplifying the purification process. For R&D teams, understanding this mechanism allows for the rational design of substrate libraries, enabling the rapid exploration of structure-activity relationships (SAR) around the 7-azaindole core without being limited by synthetic bottlenecks.
How to Synthesize N-Phenyl-7-Azaindole Derivatives Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol involves mixing the N-phenyl-7-azaindole substrate with 1.5 equivalents of the amination reagent in the presence of 5 mol% iridium catalyst and 20 mol% silver additive within an ionic liquid medium. The reaction is typically conducted at ambient pressure and mild temperatures ranging from 25°C to 30°C for a duration of 12 to 36 hours. Following the reaction, the product is isolated through extraction with cyclohexane and purified via silica gel column chromatography using a gradient of petroleum ether and ethyl acetate. The detailed standardized synthesis steps are provided in the guide below.
- Mix N-phenyl-7-azaindole substrate with acyloxy carbamate compound (1: 1.5 molar ratio) in an ionic liquid solvent such as BMIMBF4.
- Add iridium catalyst (5 mol%) and silver salt additive (20 mol%) to the mixture under inert atmosphere.
- Stir the reaction at mild temperatures (25-30°C) for 12-36 hours, then extract with cyclohexane and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this iridium-catalyzed process offers transformative advantages that directly address the pain points of modern chemical manufacturing. The shift from traditional volatile solvents to recyclable ionic liquids represents a significant step towards sustainability, potentially reducing the environmental footprint of the manufacturing facility. Moreover, the mild reaction conditions eliminate the need for expensive heating infrastructure and reduce the risk of thermal runaway incidents, enhancing overall plant safety. These factors collectively contribute to a more resilient and cost-efficient supply chain, ensuring consistent availability of high-quality intermediates for downstream drug development.
- Cost Reduction in Manufacturing: The utilization of ionic liquids as both solvent and co-catalyst eliminates the need for large volumes of disposable organic solvents, thereby significantly reducing raw material costs and waste disposal fees. Additionally, the high selectivity of the iridium catalyst minimizes the formation of byproducts, which reduces the complexity and cost of downstream purification processes such as chromatography or recrystallization. The ability to operate at near-room temperature also translates to substantial energy savings compared to high-temperature reflux conditions required by older technologies.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system allows for the use of diverse substrates with varying electronic properties, ensuring that supply chains are not bottlenecked by the availability of specific, hard-to-synthesize starting materials. The high conversion rates reported in the patent examples indicate a reliable process that can consistently meet production targets without frequent batch failures. This reliability is crucial for maintaining continuous flow in the production of active pharmaceutical ingredients (APIs), preventing costly delays in drug development timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at atmospheric pressure and mild temperatures, removing the engineering challenges associated with high-pressure reactors. The non-volatile nature of the ionic liquid solvent facilitates easier containment and potential recycling, aligning with strict environmental regulations regarding VOC emissions. This compliance reduces the regulatory burden on manufacturers and future-proofs the production process against tightening environmental laws, making it a sustainable long-term solution for industrial synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iridium-catalyzed technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this synthesis route for potential adoption.
Q: What are the advantages of using ionic liquids in this C-H amination reaction?
A: Ionic liquids serve dual roles as both solvent and co-catalyst, significantly accelerating reaction rates compared to conventional organic solvents while allowing for potential recycling and reducing volatile organic compound (VOC) emissions.
Q: How does the iridium catalyst improve selectivity over rhodium systems?
A: The iridium catalytic system specifically promotes C-N bond formation (amidation) at the ortho-position of the N-phenyl ring, avoiding the competing C-C amidation pathways often observed with rhodium catalysts, thus ensuring higher regioselectivity.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process operates at mild temperatures (25-30°C) and atmospheric pressure with high conversion rates, making it energetically efficient and safer for scale-up compared to high-temperature traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenyl-7-Azaindole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-purity intermediates for accelerating drug discovery programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of N-phenyl-7-azaindole derivative meets the exacting standards required by global pharmaceutical clients. Our commitment to quality assurance ensures that your research and production timelines remain on track without compromise.
We invite you to leverage our technical expertise to optimize your supply chain for these valuable heterocyclic compounds. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced iridium-catalyzed manufacturing capabilities can drive value and efficiency in your upcoming projects.
