Scalable Metal-Free Synthesis of Alpha-Aminoketones for Advanced Pharmaceutical Manufacturing
Scalable Metal-Free Synthesis of Alpha-Aminoketones for Advanced Pharmaceutical Manufacturing
The landscape of organic synthesis is continuously evolving towards greener, more atom-economical processes that reduce environmental impact while maintaining high efficiency. A significant breakthrough in this domain is detailed in patent CN116410098A, which discloses a novel preparation method for synthesizing alpha-aminoketone compounds directly from fatty aldehydes and secondary amines. This technology represents a paradigm shift from traditional multi-step sequences to a streamlined, one-pot oxidative rearrangement. The core innovation lies in the utilization of sodium percarbonate as a benign, commercially available oxidant that facilitates the transformation without the need for transition metal catalysts. This approach not only addresses the growing demand for sustainable chemistry in the pharmaceutical sector but also offers a robust pathway for generating structurally diverse alpha-aminoketones, which serve as critical scaffolds in medicinal chemistry.
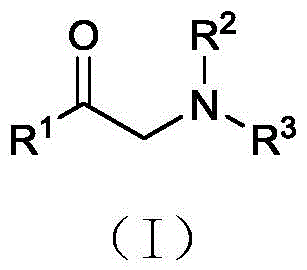
Alpha-aminoketones are privileged structures found in numerous natural products and bioactive molecules, possessing both amino and carbonyl active sites that allow for tunable interactions with biological receptors. Historically, accessing these motifs has been fraught with challenges, often requiring pre-functionalized starting materials or harsh reaction conditions. The method described in CN116410098A overcomes these hurdles by leveraging the inherent reactivity of simple fatty aldehydes and secondary amines. By operating at temperatures between 95°C and 115°C for 16 to 24 hours, the process achieves high conversion rates with remarkable substrate scope. This includes compatibility with linear and branched aliphatic chains, aromatic systems, and even complex steroid derivatives, making it a versatile tool for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to alpha-aminoketones have long been plagued by inefficiencies that hinder their widespread adoption in industrial settings. Conventional strategies typically rely on two-step operations involving the pre-preparation of reactive intermediates such as alpha-haloketones or silyl enol ethers. These precursors are often unstable, toxic, and expensive to produce, adding significant cost and safety burdens to the manufacturing process. Furthermore, alternative methods like metal-catalyzed carbene N-H insertion or the functionalization of unsaturated hydrocarbons frequently suffer from poor positional selectivity, particularly with linear ketones, leading to complex mixtures of regioisomers that are difficult to separate. The reliance on precious metal catalysts also introduces the risk of heavy metal contamination in the final product, necessitating costly and time-consuming purification steps to meet stringent regulatory standards for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a direct oxidative amination strategy that bypasses the need for pre-functionalized substrates or exotic catalysts. By employing sodium percarbonate, a stable and inexpensive solid source of hydrogen peroxide, the reaction proceeds through a clean oxidative rearrangement mechanism. This method allows for the direct coupling of readily available fatty aldehydes and secondary amines in a single reaction vessel. The operational simplicity is a major advantage; the reaction can be performed in common solvent systems like chloroform and dichloromethane under aerobic conditions. As demonstrated in the specific examples, such as the conversion of hexanal and dibenzylamine, the process delivers the desired alpha-aminoketone products in respectable yields without generating hazardous waste streams associated with halogenated reagents.
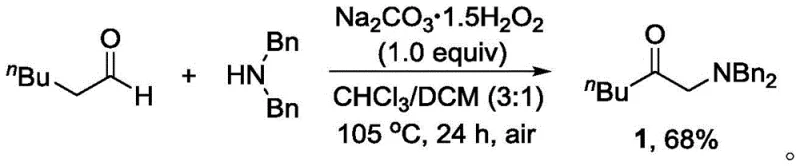
Mechanistic Insights into Sodium Percarbonate-Mediated Oxidative Rearrangement
The mechanistic pathway of this transformation is a testament to the elegance of green chemistry principles applied to complex molecule synthesis. While the exact catalytic cycle may involve radical or ionic intermediates generated by the decomposition of sodium percarbonate, the net result is the formal insertion of the amine nitrogen into the alpha-position of the aldehyde-derived carbonyl framework. The absence of transition metals suggests that the oxidation potential of the percarbonate species is sufficient to activate the aldehyde or the transient imine/enamine intermediate formed in situ. This metal-free nature is particularly advantageous for R&D directors focused on impurity profiles, as it eliminates the possibility of metal-ligand complexes persisting in the final API. The reaction conditions are optimized to balance reaction kinetics with substrate stability, ensuring that sensitive functional groups remain intact throughout the 16 to 24-hour heating period.
From an impurity control perspective, the simplicity of the reagent system translates to a cleaner crude reaction mixture. Unlike metal-catalyzed processes that often generate a plethora of side products due to competing coordination pathways, this oxidative rearrangement is highly selective for the alpha-aminoketone motif. The purification process, typically involving standard silica gel chromatography with ethyl acetate and petroleum ether, is straightforward and scalable. This ease of purification is critical for maintaining high throughput in a manufacturing environment. The method's tolerance for diverse functional groups, including esters, silyl ethers, and heterocycles, further underscores its robustness. For instance, the successful synthesis of derivatives containing pyridine rings or long-chain unsaturated fatty acids indicates that the oxidative conditions are mild enough to prevent over-oxidation or degradation of sensitive moieties.
How to Synthesize Alpha-Aminoketones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and purity. The protocol involves mixing the fatty aldehyde, secondary amine, and sodium percarbonate in a defined molar ratio, typically ranging from 0.75 to 1.5 equivalents of aldehyde relative to the amine and oxidant. The choice of solvent, a mixture of chloroform and dichloromethane, plays a crucial role in solubilizing the organic substrates while allowing the solid oxidant to interact effectively at the interface or in suspension. Maintaining the temperature within the 95-115°C window is essential; lower temperatures may result in incomplete conversion, while higher temperatures could lead to decomposition of the percarbonate or the product. Detailed standardized synthesis steps for specific substrates are provided in the guide below.
- Mix fatty aldehyde, secondary amine, and sodium percarbonate in a chloroform/dichloromethane solvent system within a sealed reaction vessel.
- Heat the reaction mixture to a temperature range of 95-115°C and maintain stirring for a duration of 16 to 24 hours to ensure complete conversion.
- Purify the resulting crude mixture using standard chromatographic techniques, such as thin-layer chromatography with an ethyl acetate and petroleum ether eluent system, to isolate the pure alpha-aminoketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are profound. The shift away from precious metal catalysts and pre-functionalized intermediates directly impacts the cost of goods sold (COGS). Sodium percarbonate is a commodity chemical produced on a massive scale for detergent applications, ensuring a stable and low-cost supply chain that is immune to the geopolitical volatility often seen with rare earth metals or specialized organometallic reagents. Furthermore, the reduction in synthetic steps from two or more down to a single pot operation drastically reduces labor costs, energy consumption, and solvent usage. This consolidation of the process flow enhances overall manufacturing efficiency, allowing facilities to produce more material in less time with existing infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium, rhodium, or iridium removes a significant variable cost from the production budget. Additionally, the avoidance of hazardous alpha-haloketone intermediates reduces the need for specialized containment equipment and waste disposal protocols associated with halogenated byproducts. The use of simple, commercially available fatty aldehydes and secondary amines as starting materials ensures that raw material sourcing is straightforward and competitive, driving down the overall input costs for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Reliance on commodity chemicals like sodium percarbonate and bulk solvents mitigates the risk of supply disruptions. Unlike specialized reagents that may have single-source suppliers or long lead times, the inputs for this process are widely available from multiple global vendors. This diversification of the supply base enhances resilience against market fluctuations and logistical bottlenecks. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain and ensuring consistent delivery of high-purity alpha-aminoketones to downstream customers.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with modern environmental, social, and governance (ESG) goals. The generation of minimal waste and the absence of heavy metals simplify the regulatory approval process for new drug applications. From a scalability standpoint, the reaction does not require high-pressure reactors or cryogenic cooling, making it easily adaptable from gram-scale discovery to multi-ton commercial production. The simple workup and purification procedures facilitate rapid turnover in manufacturing suites, optimizing asset utilization and reducing the carbon footprint of the synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative rearrangement technology. These insights are derived directly from the experimental data and scope limitations defined in the patent literature, providing a realistic overview of what process development teams can expect when integrating this method into their workflows. Understanding these nuances is key to successful technology transfer and scale-up.
Q: What are the primary advantages of using sodium percarbonate over traditional metal catalysts for alpha-aminoketone synthesis?
A: The use of sodium percarbonate eliminates the need for expensive and potentially toxic transition metal catalysts, significantly reducing raw material costs and simplifying the downstream purification process by removing the requirement for heavy metal scavenging steps.
Q: Does this synthetic method tolerate sensitive functional groups commonly found in drug molecules?
A: Yes, the methodology exhibits excellent functional group tolerance, successfully accommodating substrates containing olefins, silyl ethers, esters, and heterocycles without the need for additional protective group strategies, thereby enhancing atomic economy.
Q: Is this process suitable for large-scale industrial production of pharmaceutical intermediates?
A: Absolutely. The reaction utilizes commercially available, low-cost reagents and operates under relatively mild thermal conditions without requiring specialized high-pressure equipment, making it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aminoketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient, green synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively evaluated the methodology described in CN116410098A and confirmed its potential for generating high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from the bench to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies, guaranteeing that every batch of alpha-aminoketone delivered meets the highest standards of quality and consistency.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules. Whether you require route feasibility assessments for novel derivatives or need to secure a long-term supply of established intermediates, we are committed to being your partner in success. Contact our technical procurement team today to request specific COA data and discuss how our advanced synthesis capabilities can accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
