Advanced Manufacturing of Phenylalkanoic Acid Amide Intermediates for High-Efficiency Fungicides
The chemical landscape for producing high-performance fungicides is constantly evolving, driven by the need for more efficient and cost-effective synthetic routes. Patent CN1304399A introduces a groundbreaking methodology for the preparation of phenylalkanoic acid amide compounds, which serve as critical intermediates in the manufacture of potent fungicidal agents. This technology addresses significant bottlenecks in traditional synthesis by leveraging a novel intramolecular ring-closure reaction of nitrile compounds to form oxazolinone intermediates. Unlike conventional methods that rely on scarce or expensive precursors, this approach utilizes widely available N-acyl aminonitriles, transforming them through a streamlined acid-catalyzed cyclization. The subsequent reaction of these oxazolinone compounds with carboxyl derivatives in the presence of a base offers a robust pathway to complex amide structures. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing processes for agrochemical intermediates.
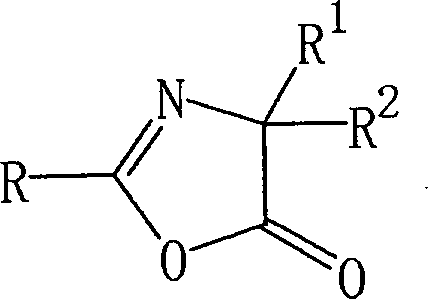
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxazolinone compounds, which are essential precursors for various bioactive molecules, has been fraught with industrial challenges. Traditional methodologies often involve the dehydration cyclization of amino acids where the amino group is protected by an acyl group. While chemically feasible, this route is severely limited by the availability and high cost of the corresponding protected amino acids, making it economically unviable for large-scale production. Alternatively, methods utilizing oxalyl chloride or chloroglyoxylate to produce oxazolinones from acetylated aminonitriles have been documented. However, these reagents are not only hazardous to handle on a commercial scale but also introduce significant safety risks and waste disposal issues. The difficulty in sourcing high-quality starting materials and the harsh reaction conditions associated with these legacy processes often result in lower overall yields and inconsistent product quality, creating substantial supply chain vulnerabilities for manufacturers of fungicidal active ingredients.
The Novel Approach
The innovative process described in the patent data fundamentally reimagines the synthesis trajectory by starting with highly accessible nitrile compounds. By reacting these nitriles with an acid, such as sulfuric acid or hydrochloric acid, an efficient intramolecular ring closure occurs to generate the oxazolinone core. This step bypasses the need for expensive amino acid derivatives entirely. Following the formation of the oxazolinone ring, the process employs a base-mediated reaction with carboxyl compounds, such as malonate half-esters, to construct the final amide backbone. This two-step sequence is characterized by its operational simplicity and the use of common industrial solvents like acetic acid, toluene, and acetonitrile. The ability to tune reaction parameters, such as temperature ranges from 0°C to 80°C and precise molar ratios of acid catalysts, allows for exceptional control over the reaction outcome. This novel approach not only simplifies the synthetic route but also significantly enhances the economic feasibility of producing high-purity agrochemical intermediates.
Mechanistic Insights into Acid-Catalyzed Cyclization and Base-Mediated Acylation
The core of this technological advancement lies in the mechanistic efficiency of the acid-catalyzed cyclization of N-acyl aminonitriles. In this step, the nitrile nitrogen acts as a nucleophile, attacking the carbonyl carbon of the adjacent amide group under acidic conditions. The presence of a strong acid catalyst, preferably sulfuric acid used in amounts ranging from 0.1 to 2 moles per mole of nitrile, facilitates protonation and activates the nitrile group for cyclization. The reaction is typically conducted in a solvent system that can dissolve both the organic substrate and the acid, with glacial acetic acid being a preferred medium. Crucially, the addition of small amounts of water, specifically between 0.0005 to 1 mole per mole of nitrile, has been found to surprisingly enhance the yield of the desired oxazolinone product without causing hydrolysis. This delicate balance of acidity and hydration ensures that the ring closure proceeds rapidly while minimizing the formation of by-products, thereby establishing a clean impurity profile from the very first step of the synthesis.
Following the formation of the oxazolinone intermediate, the second critical mechanistic phase involves the nucleophilic attack by a carboxyl compound in the presence of a base. The oxazolinone ring is susceptible to ring-opening when treated with bases such as alkali metal hydrides, organic amines like triethylamine, or pyridines. In the presence of a Lewis acid co-catalyst like magnesium chloride, the enolate formed from the carboxyl compound (e.g., monopotassium ethyl malonate) attacks the carbonyl carbon of the oxazolinone. This results in the cleavage of the C-O bond of the heterocycle and the formation of a new C-C bond, yielding the target beta-keto amide structure. The reaction conditions are carefully optimized, typically requiring temperatures between 0°C and 100°C and reaction times of 1 to 24 hours. This mechanism allows for the introduction of diverse R-groups at the alpha-position, providing significant structural flexibility for generating a wide library of fungicidal intermediates with tailored biological activity.
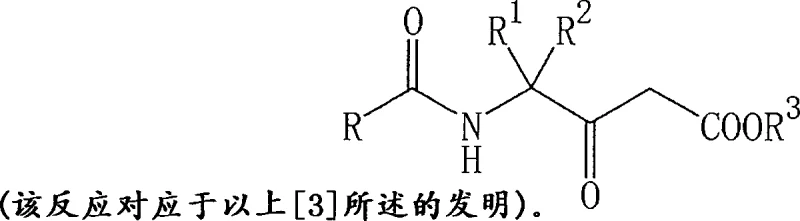
How to Synthesize Phenylalkanoic Acid Amides Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize yield and purity. The process begins with the preparation of the oxazolinone intermediate by mixing the N-acyl aminonitrile with an acid catalyst in a suitable solvent, followed by heating to induce cyclization. Once the intermediate is isolated and purified, it is subjected to the coupling reaction with the chosen carboxyl component under basic conditions. The detailed standardized synthesis steps, including specific workup procedures like pH adjustment and extraction protocols, are critical for reproducibility. For a comprehensive guide on executing this protocol with precision, please refer to the technical instructions below.
- Cyclize N-acyl aminonitrile compounds using an acid catalyst like sulfuric acid in a solvent such as glacial acetic acid at temperatures between 0-80°C to form the oxazolinone intermediate.
- React the isolated oxazolinone compound with a carboxyl compound, such as a malonate half-ester, in the presence of a base like triethylamine and a Lewis acid like magnesium chloride.
- Perform hydrolysis and decarboxylation under acidic or basic conditions with water to convert the resulting amide intermediate into the final ketone compound if required for downstream fungicide synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this nitrile-based synthesis route offers profound advantages over traditional amino acid-dependent methods. The primary driver for cost optimization is the substitution of expensive, protected amino acids with readily available and inexpensive nitrile starting materials. This shift in raw material sourcing drastically reduces the direct material costs associated with the production of these critical intermediates. Furthermore, the elimination of hazardous reagents like oxalyl chloride simplifies the safety compliance requirements and reduces the overhead costs related to waste treatment and environmental protection. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent systems, ensures high process reliability, minimizing the risk of batch failures that can disrupt supply chains. Consequently, manufacturers can achieve substantial cost savings while maintaining a consistent and reliable supply of high-quality intermediates for the agrochemical sector.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the use of low-cost commodity chemicals as starting materials. By avoiding the complex synthesis and protection/deprotection steps associated with amino acids, the overall number of unit operations is reduced. Additionally, the use of common solvents and catalysts that can be potentially recovered and recycled further drives down the operational expenditure. The high yields reported in the experimental examples, often exceeding 80% for the cyclization step, indicate a highly atom-economical process that minimizes raw material waste. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for fungicide intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the accessibility of the raw materials required for this synthesis. Nitrile compounds and simple carboxylic acid derivatives are produced on a massive industrial scale globally, ensuring that supply shortages are unlikely to occur. Unlike specialized chiral amino acids which may have limited suppliers and long lead times, the precursors for this route are commoditized. This abundance allows procurement managers to negotiate better terms and secure long-term contracts with multiple vendors. Moreover, the synthetic route's tolerance to variations in reaction conditions means that production can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand, ensuring uninterrupted continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations and workup techniques such as liquid-liquid extraction and crystallization. The absence of heavy metal catalysts or exotic reagents simplifies the purification process and reduces the burden on wastewater treatment facilities. The reaction generates minimal hazardous by-products, aligning with increasingly stringent environmental regulations and sustainability goals. This green chemistry profile not only reduces the environmental footprint of the manufacturing process but also mitigates regulatory risks. For supply chain heads, this means a future-proof production method that is less likely to face shutdowns due to environmental non-compliance, ensuring long-term viability and stability for the production of these essential agrochemical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the versatility and robustness of the chemistry involved.
Q: What are the primary advantages of the oxazolinone route over traditional amino acid dehydration?
A: The oxazolinone route utilizes readily available and cost-effective nitrile starting materials, avoiding the high cost and supply constraints associated with protected amino acids. Furthermore, the reaction conditions are milder and avoid hazardous reagents like oxalyl chloride, leading to a safer and more scalable industrial process.
Q: How does this process impact the purity profile of the final agrochemical intermediate?
A: By employing specific acid catalysts like sulfuric acid and controlled temperatures (0-80°C), the process minimizes side reactions and decomposition of the sensitive oxazolinone ring. Subsequent purification via crystallization or column chromatography ensures high-purity intermediates suitable for stringent pharmaceutical and agrochemical applications.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly scalable due to the use of common industrial solvents like toluene and acetonitrile, and robust reagents such as magnesium chloride and triethylamine. The workup procedures involve standard extraction and crystallization techniques, facilitating seamless transition from laboratory to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylalkanoic Acid Amides Supplier
The synthesis pathway outlined in patent CN1304399A demonstrates immense potential for the efficient production of high-value fungicidal intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the oxazolinone ring closure and subsequent acylation are executed with precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Phenylalkanoic Acid Amides meets the exacting standards required by the global agrochemical industry. Our commitment to quality and process optimization makes us an ideal partner for bringing advanced intermediate technologies to market.
We invite you to explore how our manufacturing capabilities can optimize your supply chain and reduce costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this novel synthesis can enhance your product portfolio. Let us collaborate to drive efficiency and innovation in your agrochemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
