Revolutionizing Axial Chiral Aryl Olefin Production with Advanced Palladium Catalysis
The landscape of asymmetric synthesis is constantly evolving, driven by the demand for more efficient and sustainable routes to complex chiral molecules. A significant breakthrough in this domain is documented in Chinese Patent CN108586284B, which introduces a robust methodology for the construction of axial chiral aryl olefin compounds. These structures are pivotal in the development of advanced pharmaceutical intermediates and specialized ligands, yet their synthesis has historically been plagued by challenges such as low rotational energy barriers and susceptibility to racemization. The patented technology addresses these critical pain points by leveraging a palladium-catalyzed intermolecular asymmetric carbon-hydrogen bond alkenylation strategy. This approach not only streamlines the synthetic pathway but also ensures high levels of stereoselectivity, making it a highly attractive option for industrial-scale production where purity and consistency are paramount.
Traditionally, the synthesis of axially chiral compounds relied heavily on the coupling of pre-functionalized aryl halides with organometallic reagents. This conventional paradigm often necessitated lengthy multi-step sequences to install the requisite halogen handles, resulting in poor atom economy and substantial waste generation. Furthermore, controlling the stereochemistry of the resulting axis was frequently difficult, leading to mixtures of enantiomers that required costly and time-consuming separation processes. The reliance on sensitive organometallic reagents also imposed strict operational constraints, typically requiring inert atmospheres and anhydrous conditions, which complicates scale-up and increases infrastructure costs for manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The legacy methods for constructing aryl-olefin axes often suffer from inherent inefficiencies that hinder their commercial viability. The prerequisite for aryl halides means that every batch starts with a more expensive and less available starting material compared to simple arenes. Additionally, the use of stoichiometric amounts of metal reagents generates significant heavy metal waste, posing environmental compliance challenges and increasing disposal costs. From a process safety perspective, handling large quantities of reactive organometallics introduces hazards that require specialized equipment and rigorous safety protocols. These factors collectively contribute to a higher cost of goods sold (COGS) and longer lead times, creating bottlenecks in the supply chain for downstream drug development projects that rely on these chiral building blocks.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a direct C-H activation strategy that fundamentally reshapes the synthetic logic. By employing readily available 3-alkyl-2-aryl-1-cyclohexenyl oximes and simple olefin derivatives, the process bypasses the need for pre-halogenation entirely. The reaction proceeds under the catalysis of a chiral palladium complex in the presence of an oxidant, facilitating the direct formation of the C-C bond with exceptional stereocontrol. Notably, the protocol is designed to operate in an air atmosphere, removing the burden of stringent inert gas management. This operational simplicity translates directly into reduced capital expenditure and lower operating costs. The versatility of the method is further demonstrated by its tolerance to a wide range of functional groups, allowing for the synthesis of diverse derivatives without the need for extensive protecting group chemistry.
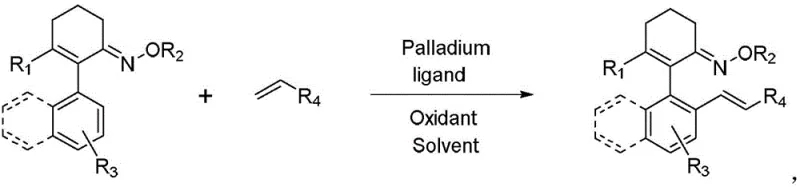
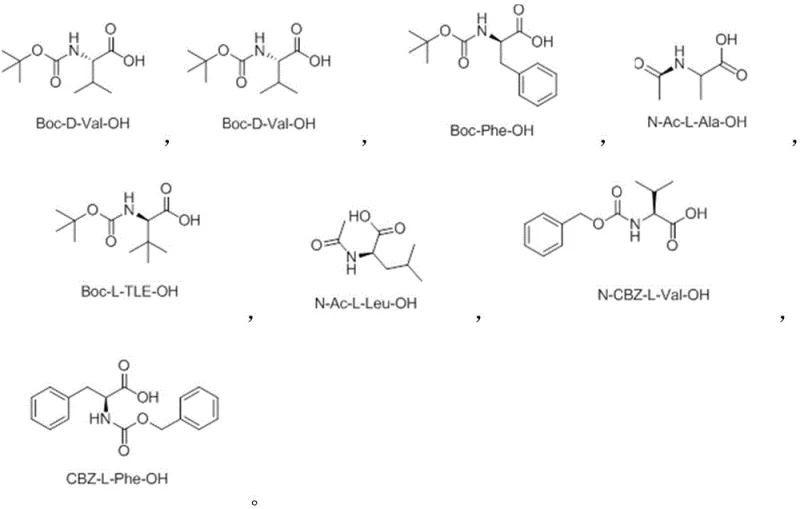
The core of this innovation lies in the precise orchestration of the catalytic cycle. The use of monoprotected chiral amino acids as ligands plays a decisive role in differentiating the prochiral faces of the substrate, thereby locking the conformational freedom of the biaryl-like axis. This ligand-directed stereocontrol ensures that the desired enantiomer is formed with high fidelity, often achieving enantiomeric excess values exceeding 95%. The mechanism involves a concerted metalation-deprotonation or electrophilic palladation pathway, followed by olefin insertion and beta-hydride elimination, all governed by the chiral environment created by the ligand. Understanding these mechanistic nuances is crucial for process chemists aiming to optimize reaction parameters for specific substrates.
Mechanistic Insights into Pd-Catalyzed Asymmetric Alkenylation
The catalytic system relies on the synergistic interaction between the palladium center and the chiral ligand to overcome the low rotational barrier inherent to aryl-olefin systems. The ligand, typically derived from natural amino acids, coordinates to the palladium species, creating a rigid chiral pocket that directs the approach of the olefin substrate. This steric guidance is essential for establishing the axial chirality during the bond-forming event. The choice of oxidant, such as silver acetate or benzoquinone, is also critical for regenerating the active Pd(II) species from the Pd(0) intermediate formed after product release, thus sustaining the catalytic turnover. The ability to perform this transformation in solvents like methanol or toluene further enhances the practicality of the method, as these are common, cost-effective industrial solvents.
Impurity control is another significant advantage of this catalytic system. The high selectivity of the C-H activation minimizes the formation of regioisomers, which are common byproducts in non-directed coupling reactions. Furthermore, the mild reaction conditions help preserve sensitive functional groups that might otherwise degrade under harsher traditional conditions. The post-reaction workup is straightforward, typically involving filtration to remove silver salts followed by standard chromatographic purification. This simplicity in isolation reduces the risk of product degradation during processing and ensures a high-purity final product, which is a critical quality attribute for pharmaceutical applications. The robustness of the catalyst system also suggests a potential for recycling or continuous flow adaptation, offering further opportunities for process intensification.
How to Synthesize Axial Chiral Aryl Olefins Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the oxidant and the loading of the chiral ligand. The patent provides detailed embodiments showing that molar ratios of olefin to oxime ranging from 1.1:1 to 3:1 are effective, allowing for flexibility depending on the cost and availability of the olefin partner. Reaction temperatures between 40°C and 80°C provide a balance between reaction rate and selectivity, ensuring that the process remains energy-efficient. For those looking to adopt this technology, the standardized protocol offers a clear path from gram-scale experimentation to larger production batches, minimizing the risk associated with technology transfer.
- Prepare the reaction mixture by combining 3-alkyl-2-aryl-1-cyclohexenyl oxime and an olefin derivative in a suitable solvent such as methanol or toluene.
- Add the catalytic system consisting of a palladium salt (e.g., palladium acetate) and a monoprotected chiral amino acid ligand, along with an oxidant like silver acetate.
- Stir the reaction in an air atmosphere at temperatures between 40-80°C for 10-48 hours, followed by purification via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic benefits beyond mere technical elegance. The shift from halide-based coupling to direct C-H activation represents a fundamental optimization of the raw material basket. By utilizing simpler, more abundant starting materials, manufacturers can mitigate the risks associated with the supply volatility of specialized halogenated intermediates. This diversification of the supply base enhances resilience against market fluctuations and geopolitical disruptions that often affect the availability of fine chemical building blocks. Moreover, the reduction in synthetic steps directly correlates to a shorter overall production cycle, enabling faster response times to customer demands and accelerating time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps significantly lowers the direct material costs associated with the synthesis. Without the need to purchase or synthesize aryl halides, the bill of materials is streamlined, leading to substantial savings. Additionally, the improved atom economy means less waste is generated per kilogram of product, which reduces the costs associated with waste treatment and disposal. The use of air-stable conditions further cuts down on utility costs related to nitrogen or argon consumption, contributing to a leaner and more cost-effective manufacturing profile that improves overall margin potential.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is vital for maintaining a reliable supply of critical intermediates. The tolerance for air and moisture simplifies the logistical requirements for storage and transport of reagents, reducing the likelihood of spoilage or degradation before use. This reliability allows supply chain planners to maintain lower safety stock levels while still meeting production targets, freeing up working capital. The scalability of the process ensures that supply can be ramped up quickly to meet surges in demand without compromising on the stringent purity specifications required by regulatory bodies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this green chemistry approach aligns perfectly with modern sustainability goals. The reduction in heavy metal waste and hazardous reagents simplifies compliance with increasingly strict environmental regulations. The process is inherently safer due to the absence of pyrophoric reagents, lowering the insurance and safety infrastructure costs for the manufacturing site. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, which is becoming an increasingly important factor for multinational partners when selecting vendors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this axial chiral synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows.
Q: What is the primary advantage of this C-H activation method over traditional cross-coupling?
A: This method eliminates the need for pre-functionalized aryl halides, significantly reducing synthetic steps and waste generation while maintaining high enantioselectivity.
Q: Can this reaction be performed under standard atmospheric conditions?
A: Yes, the patent specifies that the reaction can be completed in an air atmosphere, which simplifies operational requirements compared to inert gas protocols.
Q: What types of ligands are effective for stereocontrol in this process?
A: Monoprotected chiral amino acids, such as N-acetyl-L-alanine or N-CBZ-L-valine, are utilized to induce high levels of axial chirality during the alkenylation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Aryl Olefin Supplier
As the pharmaceutical industry continues to demand more complex and potent chiral molecules, the ability to deliver high-purity intermediates efficiently is a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging advanced catalytic technologies like the one described in CN108586284B to serve our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage development through to full-scale commercialization. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest international standards.
We invite you to explore the potential of this innovative synthesis route for your specific projects. By partnering with us, you gain access to not just a product, but a comprehensive Customized Cost-Saving Analysis tailored to your unique supply chain requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to optimize your manufacturing process, reduce costs, and accelerate the delivery of life-saving therapies to the market.
