Advanced Synthesis of Cyclopentachromanone Spiro-Oxoindole Intermediates for Oncology Drug Discovery
The pharmaceutical industry is constantly seeking novel scaffolds that offer potent biological activity combined with synthetic accessibility. Patent CN112390804A, published on February 23, 2021, introduces a groundbreaking class of cyclopentachromanone spliced spiro-oxoindole compounds that merge two highly privileged structures in medicinal chemistry. This innovation provides a robust source of compounds for bioactive screening, specifically demonstrating significant inhibitory activity against human leukemia cells (K562). The strategic splicing of the cyclopentano-chromanone skeleton with the spiro-oxoindole framework creates a multi-scaffold molecule with potential applications in oncology drug discovery. For R&D directors and procurement specialists, this patent represents a critical opportunity to access high-value intermediates through a streamlined, metal-free synthetic pathway that ensures both high purity and supply chain reliability.
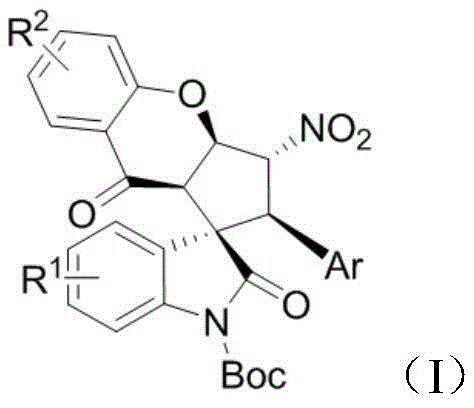
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex spiro-oxindole frameworks often rely heavily on transition metal catalysis, which introduces significant bottlenecks in pharmaceutical manufacturing. The use of precious metals such as palladium, rhodium, or iridium not only escalates raw material costs but also necessitates rigorous downstream processing to meet stringent regulatory limits on residual heavy metals in active pharmaceutical ingredients (APIs). Furthermore, many conventional methods suffer from poor atom economy and require harsh reaction conditions, including high temperatures or strong acidic environments, which can compromise the stability of sensitive functional groups. These factors collectively lead to extended production lead times and increased waste generation, posing substantial challenges for supply chain heads aiming to maintain cost-effective and environmentally compliant operations. The complexity of purifying reaction mixtures from metal residues often results in reduced overall yields, further impacting the commercial viability of these traditional approaches.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112390804A utilizes an organocatalytic Michael/Michael cycloaddition reaction that operates under remarkably mild conditions. By employing chiral thiourea or aromatic amide catalysts derived from readily available precursors like cinchona alkaloids or cyclohexyldiamine, this novel approach eliminates the need for transition metals entirely. The reaction proceeds efficiently in common organic solvents such as diethyl ether, acetonitrile, or toluene at temperatures ranging from -10°C to 40°C, demonstrating excellent air stability and operational simplicity. This shift to organocatalysis not only drastically simplifies the work-up procedure by removing metal scavenging steps but also enhances the safety profile of the manufacturing process. The ability to tolerate a wide range of substituent groups without compromising yield or selectivity makes this route exceptionally versatile for generating diverse libraries of analogues for structure-activity relationship (SAR) studies.
Mechanistic Insights into Organocatalytic Michael/Michael Cycloaddition
The core of this synthetic breakthrough lies in the precise activation of substrates through a dual hydrogen-bonding network facilitated by the chiral organocatalyst. The reaction initiates with the activation of the bifunctional oxoindole-chromone 3C synthon, where the catalyst simultaneously coordinates with the carbonyl groups to enhance electrophilicity. Concurrently, the nitrostyrene nucleophile is activated through hydrogen bonding interactions, orienting it for a highly stereoselective attack. This cooperative activation mode ensures that the carbon-carbon bond formation occurs with exceptional control over the stereochemical outcome, leading to the formation of multiple chiral centers in a single operational step. The cascade nature of the Michael/Michael sequence allows for the rapid construction of the complex spirocyclic core, minimizing the number of isolation steps required and maximizing overall process efficiency.
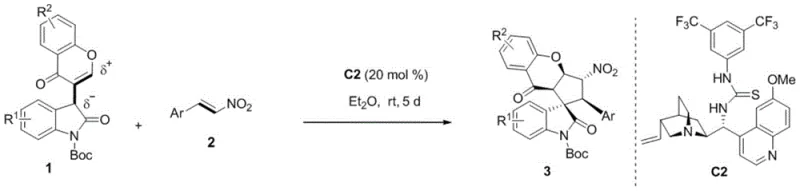
Impurity control is inherently built into the mechanism due to the high specificity of the organocatalyst. The rigid transition state imposed by the chiral catalyst framework effectively suppresses the formation of unwanted diastereomers and enantiomers, resulting in products with enantiomeric excess (ee) values consistently exceeding 99% and diastereomeric ratios (dr) greater than 20:1. This high level of stereocontrol is crucial for pharmaceutical applications, as it reduces the burden on chiral separation processes which are often the most costly and time-consuming stages of API manufacturing. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups, ensuring that the final product profile remains clean and free from decomposition byproducts. The robustness of this catalytic system against moisture and oxygen further contributes to batch-to-batch consistency, a key metric for supply chain reliability.
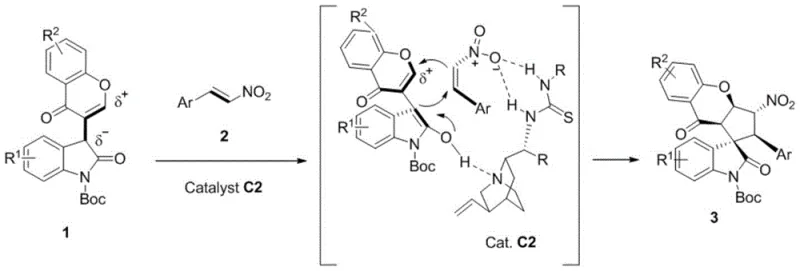
How to Synthesize Cyclopentachromanone Spiro-Oxoindole Efficiently
The synthesis of these high-value intermediates is designed for practical implementation in both laboratory and pilot plant settings. The protocol involves the sequential addition of the bifunctional synthon and nitrostyrene derivative into a reaction vessel containing the chosen organic solvent, followed by the introduction of the chiral catalyst. The mixture is then stirred at ambient temperature for a period ranging from 2 to 10 days, depending on the specific electronic nature of the substituents. Reaction progress is monitored via thin-layer chromatography (TLC), and upon completion, the crude product can be purified directly by column chromatography using standard eluent systems such as petroleum ether and ethyl acetate. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Prepare the reaction mixture by adding bifunctional oxoindole-chromone 3C synthon and substituted nitrostyrene into an organic solvent such as diethyl ether.
- Introduce the chiral thiourea catalyst (e.g., catalyst C2) at a loading of 20 mol% to initiate the asymmetric cascade reaction.
- Stir the reaction at room temperature for 2 to 10 days, monitor by TLC, and purify the resulting spiro-oxoindole compound via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this organocatalytic technology offers transformative benefits for procurement managers and supply chain leaders seeking to optimize their sourcing strategies. The elimination of expensive transition metal catalysts directly translates to significant cost reductions in raw material procurement, while the simplified purification workflow reduces solvent consumption and waste disposal costs. The use of cheap and easily obtained starting materials ensures a stable supply base, mitigating the risks associated with sourcing specialized reagents. Moreover, the operational simplicity and air stability of the reaction allow for flexible manufacturing schedules, reducing the dependency on highly specialized equipment or inert atmosphere conditions. These factors collectively enhance the overall economic feasibility of producing these complex intermediates at a commercial scale.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the synthesis route eliminates the need for costly metal scavengers and extensive purification steps required to meet regulatory limits on heavy metal residues. This streamlined process significantly lowers the cost of goods sold (COGS) by reducing both material expenses and processing time. Additionally, the high yields and exceptional stereoselectivity minimize the loss of valuable intermediates during purification, further enhancing the economic efficiency of the manufacturing process. The ability to use common organic solvents instead of specialized anhydrous or degassed solvents also contributes to substantial operational savings.
- Enhanced Supply Chain Reliability: The reliance on readily available organic small molecule catalysts and common chemical building blocks ensures a robust and resilient supply chain. Unlike processes dependent on scarce precious metals, this method is less susceptible to market volatility and supply disruptions. The mild reaction conditions and high air stability of the reagents simplify logistics and storage requirements, allowing for safer and more efficient transportation of materials. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: The organocatalytic nature of this synthesis aligns perfectly with green chemistry principles, offering a more environmentally sustainable alternative to traditional metal-catalyzed methods. The absence of toxic heavy metals reduces the environmental footprint of the manufacturing process and simplifies compliance with increasingly stringent environmental regulations. The reaction's compatibility with various solvents and its tolerance to functional groups make it highly scalable, facilitating the transition from gram-scale laboratory synthesis to multi-kilogram commercial production without significant process re-engineering. This scalability ensures that supply can be rapidly ramped up to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel spiro-oxoindole compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing drug discovery or manufacturing pipelines. We encourage stakeholders to review these insights to fully appreciate the strategic value of this synthetic methodology.
Q: What is the primary advantage of this organocatalytic route over traditional metal-catalyzed methods?
A: The primary advantage is the elimination of transition metal catalysts, which removes the need for expensive and complex heavy metal removal steps, thereby significantly simplifying the purification process and reducing overall manufacturing costs while ensuring high pharmaceutical purity.
Q: What level of stereoselectivity can be achieved with this synthesis method?
A: The method demonstrates exceptional stereoselectivity, consistently achieving enantiomeric excess (ee) values greater than 99% and diastereomeric ratios (dr) exceeding 20:1, which is critical for the development of single-enantiomer drug candidates.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction utilizes cheap and easily obtained raw materials, operates under mild conditions with good air stability, and avoids sensitive reagents, making it highly amenable to commercial scale-up for the production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopentachromanone Spiro-Oxoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercially viable realities. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in patent CN112390804A can be effectively realized on an industrial scale. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and consistency makes us the ideal partner for companies looking to secure a stable supply of these advanced oncology intermediates.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how this organocatalytic route can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us help you navigate the complexities of commercializing this cutting-edge chemistry. Together, we can drive the next generation of cancer therapeutics forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
