Advanced Binuclear Ferrocene Triazole Ionic Complexes For Next-Generation Solid Propellant Systems
Advanced Binuclear Ferrocene Triazole Ionic Complexes For Next-Generation Solid Propellant Systems
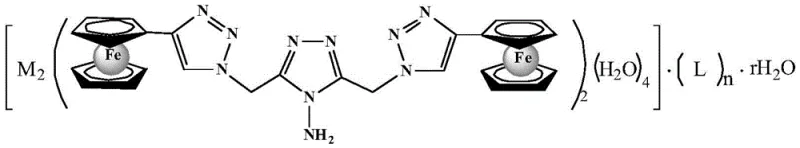
The development of high-performance solid propellants relies critically on the efficiency and stability of burning rate catalysts. Patent CN108558958B introduces a groundbreaking class of high-nitrogen binuclear ferrocene triazole ionic metal complexes that address the longstanding limitations of traditional ferrocene-based additives. This technology leverages a modular design strategy, combining the high iron content of ferrocene with the positive enthalpy of formation inherent to nitrogen-rich triazole heterocycles. By transitioning from neutral molecules to ionic coordination complexes, this innovation offers a pathway to significantly enhance the energy density and thermal stability of composite solid propellants while mitigating the migration issues that plague current commercial standards like Catocene.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional ferrocene-based burning rate catalysts, such as the widely used Catocene, have served the aerospace industry well but possess intrinsic chemical weaknesses that limit next-generation missile performance. The primary drawback of these neutral ferrocene derivatives is their tendency to migrate within the propellant matrix over time due to their non-polar nature and relatively high vapor pressure. This migration leads to uneven distribution of the catalyst, causing unpredictable burning rates and potentially catastrophic failures during operation. Furthermore, neutral ferrocenes often lack sufficient thermal stability, leading to volatilization during long-term storage which shortens the shelf-life of strategic munitions. The synthesis of earlier generation binuclear ferrocenes also tended to be chemically complex and costly, involving multi-step organic transformations that hindered large-scale economic production for defense applications.
The Novel Approach
The novel approach detailed in this patent fundamentally reengineers the catalyst architecture by introducing an ionic character to the binuclear ferrocene scaffold. By coordinating a nitrogen-rich bis-triazole ferrocene ligand with transition metal ions and high-energy polycyano anions, the resulting complexes exhibit drastically reduced volatility and migration rates. The ionic nature creates strong electrostatic interactions within the propellant binder, effectively locking the catalyst in place. Additionally, the incorporation of polycyano anions not only balances the charge but contributes additional chemical energy through their high heat of formation. This dual-function design ensures that the additive acts not merely as a passive catalyst but as an active energetic component, boosting the overall impulse of the propulsion system while maintaining exceptional thermal resilience under harsh environmental conditions.
Mechanistic Insights into Coordination-Driven Catalytic Enhancement
The catalytic efficacy of these complexes stems from a sophisticated synergistic mechanism between the central transition metal ions and the ferrocenyl-triazole ligands. The ligand, synthesized via a highly efficient copper-catalyzed azide-alkyne cycloaddition (click chemistry), provides a rigid, nitrogen-rich framework that stabilizes the metal center in a specific oxidation state. Upon thermal decomposition of the propellant, the metal center facilitates electron transfer processes that lower the activation energy for ammonium perchlorate (AP) decomposition. The presence of multiple triazole rings ensures a high nitrogen content, which decomposes exothermically to release gaseous nitrogen, further driving the combustion reaction forward. This cooperative effect between the iron from the ferrocene units and the coordinated transition metal creates a multi-site catalytic environment that is far more effective than simple physical mixtures of components.
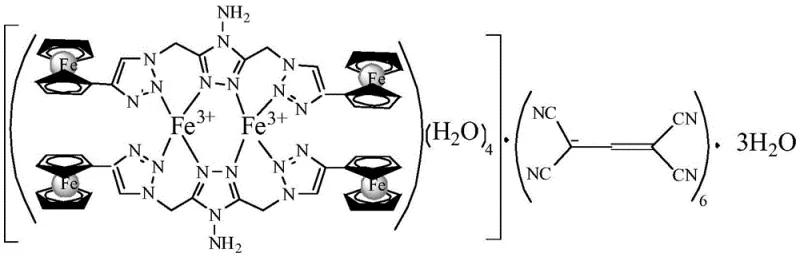
Impurity control and structural integrity are maintained through the robustness of the coordination bonds formed during synthesis. The use of polycyano anions, such as 1,1,3,3-tetracyanoacrylate, forms stable ionic lattices that resist hydrolysis and oxidation better than simple halide or nitrate salts. The crystal engineering aspects, evidenced by the specific unit cell parameters and dihedral angles observed in the X-ray diffraction data, suggest a tightly packed molecular arrangement. This dense packing contributes to the low volatility and high density of the material, which are critical parameters for maximizing the volumetric impulse of solid rocket motors. The ability to tune the metal center allows chemists to optimize the Lewis acidity of the catalyst, thereby tailoring the decomposition profile of the propellant to specific thrust requirements without compromising the mechanical properties of the cured grain.
How to Synthesize High-Nitrogen Binuclear Ferrocene Triazole Complexes Efficiently
The preparation method outlined in the patent represents a significant simplification over previous multi-step organic syntheses required for functionalized ferrocenes. The process utilizes readily available metal nitrates and a pre-synthesized triazole ligand, combining them in a straightforward one-pot coordination reaction. This streamlined approach reduces the number of purification steps and minimizes solvent waste, aligning with modern green chemistry principles essential for sustainable energetic material manufacturing. The reaction conditions are mild, typically requiring temperatures between 60°C and 80°C, which lowers energy consumption and reduces the risk of thermal runaway during production. Detailed standardized synthesis steps for scaling this process are provided in the guide below.
- Dissolve the selected transition metal salt (e.g., Iron(III) nitrate, Copper(II) nitrate) completely in distilled water within a reaction vessel.
- Prepare separate solutions of the 4-amino-3,5-bis-(4-ferrocenyl-1,2,3-triazole-1-methyl)-1,2,4-triazole ligand in DMF and the polycyanoalkene compound in methanol.
- Combine the solutions at 60-80°C, stir for 3-4 hours to facilitate coordination, then filter, wash with solvents, and vacuum dry to isolate the final ionic complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this ionic ferrocene technology offers compelling strategic advantages rooted in process efficiency and material stability. The simplified synthesis route eliminates the need for expensive and hazardous reagents often associated with traditional ferrocene functionalization, leading to a more robust and cost-effective supply chain. The high thermal stability of the final product reduces the need for specialized cold-chain logistics during transportation and storage, significantly lowering overhead costs associated with temperature-controlled warehousing. Furthermore, the non-volatile nature of these ionic complexes ensures that inventory retains its specification integrity over extended periods, reducing waste due to shelf-life expiration and enhancing the overall reliability of the raw material supply for critical defense programs.
- Cost Reduction in Manufacturing: The synthetic pathway described avoids the use of precious metal catalysts in the final coordination step, relying instead on abundant transition metal salts like iron, copper, or zinc nitrates. This substitution drastically reduces the raw material cost profile compared to catalysts requiring noble metals or complex organometallic precursors. Additionally, the high yield reported in the experimental examples indicates efficient atom economy, meaning less starting material is wasted as byproducts. The simplified workup procedure, involving basic filtration and washing, reduces labor hours and solvent consumption, contributing to substantial operational expenditure savings in large-scale production facilities.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including ethynylferrocene and simple metal salts, are commercially available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the ionic product against environmental degradation means that safety stock can be held for longer durations without quality deterioration. This stability provides procurement teams with greater flexibility in ordering cycles and inventory management, ensuring continuous availability of critical burning rate modifiers even during periods of geopolitical supply disruption or raw material scarcity.
- Scalability and Environmental Compliance: The reaction operates in common industrial solvents like methanol and DMF, which are easily recovered and recycled using standard distillation infrastructure, facilitating seamless scale-up from laboratory to multi-ton production. The absence of heavy metal catalysts in the final formulation simplifies the regulatory compliance landscape regarding toxic residue limits in exhaust plumes. This environmental advantage aligns with increasingly stringent global regulations on energetic material emissions, future-proofing the supply chain against potential regulatory bans on older, more toxic ferrocene derivatives.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and performance of these novel ionic ferrocene complexes in energetic formulations. These answers are derived directly from the experimental data and characterization results presented in the patent documentation, providing a factual basis for evaluating the technology's suitability for specific application requirements. Understanding these nuances is essential for formulators aiming to optimize propellant performance while maintaining strict safety and quality standards.
Q: How does the ionic nature of these complexes improve propellant storage stability?
A: Unlike neutral ferrocene derivatives which suffer from high volatility and migration, the ionic structure of these binuclear complexes significantly reduces vapor pressure and mobility, ensuring long-term storage reliability and consistent burning rates.
Q: What is the thermal stability range of these new catalysts compared to Catocene?
A: Thermogravimetric analysis indicates that these complexes maintain structural integrity up to high temperatures with significantly lower weight loss rates compared to traditional Catocene, attributed to their high molecular weight and extensive hydrogen bonding networks.
Q: Can the catalytic performance be tuned for specific propellant formulations?
A: Yes, by varying the central metal ion (Fe, Cu, Cr, Zn) and the polycyano counter-anion, the heat of combustion and decomposition temperature can be precisely modulated to meet diverse energetic material requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solid Propellant Additive Supplier
The technological potential of high-nitrogen binuclear ferrocene triazole complexes represents a significant leap forward in energetic material science, offering a balance of high energy and stability that is rare in this sector. As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated coordination complexes to market. Our facility is equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of catalyst meets the exacting demands of solid propellant manufacturers, guaranteeing consistent burning rates and ballistic performance in the final munition systems.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this technology can be integrated into your existing supply chain. By requesting a Customized Cost-Saving Analysis, you can evaluate the specific economic benefits of switching to these ionic catalysts for your programs. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your unique formulation requirements, ensuring a smooth transition to this next-generation catalytic technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
