Scalable Production of Norpsilocin: Advanced Bi-Cu Catalyzed Synthesis for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for complex indole alkaloids, and the preparation method disclosed in patent CN108314641B represents a significant leap forward in the synthesis of the natural product Norpsilocin. This innovative protocol leverages a synergistic Bismuth(III) and Copper(I) catalytic system to achieve direct oxidative functionalization of the indole core, bypassing the cumbersome multi-step sequences traditionally associated with alkaloid modification. By utilizing readily available tryptamine as the starting scaffold, this route dramatically simplifies the supply chain logistics while delivering a high-yielding transformation that is critical for downstream drug discovery and development programs. The strategic integration of iodine-mediated activation followed by reductive amination allows for precise control over the regioselectivity at the C4 position of the indole ring, a notoriously difficult site to functionalize without expensive directing groups.
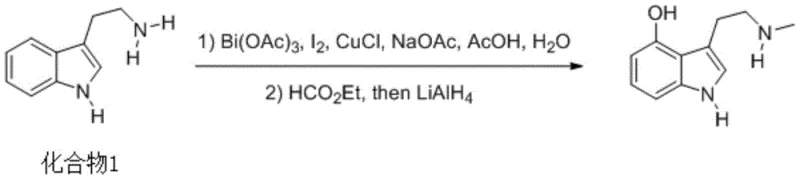
Furthermore, the operational simplicity of this method, which proceeds through a stable intermediate isolated via standard extraction techniques, offers a compelling value proposition for contract development and manufacturing organizations (CDMOs) aiming to optimize their portfolio of neuroactive compounds. The elimination of harsh halogenation conditions typically required for similar transformations reduces the environmental footprint and enhances the safety profile of the manufacturing process. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to offer clients not just a molecule, but a validated, IP-backed strategy for securing long-term supply continuity. The ability to transition from gram-scale laboratory synthesis to kilogram-level production with minimal process re-engineering is a hallmark of this technology, addressing the critical need for agility in the modern biochemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-substituted tryptamines like Norpsilocin has been plagued by significant synthetic hurdles that drive up costs and extend lead times for research teams. Traditional routes often rely on the Fischer indole synthesis or the modification of pre-existing indole scaffolds using strong electrophilic reagents that lack regioselectivity, leading to complex mixtures of 4-, 5-, 6-, and 7-substituted isomers that are difficult and expensive to separate. These conventional methods frequently necessitate the use of protecting groups on the indole nitrogen and the amine side chain to prevent unwanted side reactions, adding at least two additional synthetic steps that decrease overall atom economy and yield. Moreover, the reliance on cryogenic conditions or highly toxic heavy metal catalysts in older protocols poses substantial safety risks and waste disposal challenges, making them less attractive for green chemistry initiatives and large-scale commercial manufacturing. The cumulative effect of these inefficiencies is a high cost of goods sold (COGS) and a fragile supply chain vulnerable to raw material shortages and regulatory scrutiny regarding solvent and metal residues.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108314641B introduces a streamlined, catalytic approach that directly addresses these historical bottlenecks by employing a benign Bismuth/Copper dual-catalyst system. This novel pathway enables the direct installation of the oxygen functionality at the C4 position of the tryptamine skeleton without the need for prior protection of the amine side chain, effectively collapsing a multi-step sequence into a concise two-step operation. The use of aqueous acetic acid and sodium acetate buffers in the first step creates a mild reaction environment that tolerates the sensitive indole nucleus, preventing polymerization or degradation often seen under strongly acidic or basic conditions. Subsequent formylation and reduction with lithium aluminum hydride proceed with high efficiency, converting the intermediate directly into the target secondary amine with excellent selectivity. This approach not only reduces the consumption of solvents and reagents but also simplifies the purification workflow, as the byproducts are largely water-soluble or easily removed during the aqueous workup, resulting in a cleaner crude product profile.
Mechanistic Insights into Bi-Cu Catalyzed Oxidative Functionalization
The core innovation of this synthesis lies in the intricate interplay between Bismuth(III) acetate and Cuprous Chloride, which facilitates a unique oxidative activation of the indole ring. In the initial step, the iodine species, activated by the Lewis acidic bismuth center, likely generates an electrophilic iodinium ion that attacks the electron-rich C4 position of the tryptamine indole ring, favored by the electronic influence of the C3-ethylamine side chain. The copper co-catalyst plays a crucial role in mediating the redox cycle, potentially assisting in the regeneration of the active iodine species or stabilizing radical intermediates that prevent over-oxidation of the sensitive pyrrole ring. This catalytic cycle operates efficiently in a mixed aqueous-organic solvent system, where water acts not merely as a solvent but potentially as the oxygen source for the eventual hydroxyl group following hydrolysis of the iodonium intermediate. The precise stoichiometry of the catalysts, typically ranging from 0.1 to 0.8 equivalents relative to the substrate, ensures that the reaction proceeds to completion without excessive accumulation of heavy metal waste, aligning with modern principles of sustainable catalysis.
Following the oxidative functionalization, the second phase of the mechanism involves the conversion of the primary amine side chain into the N-methyl secondary amine characteristic of Norpsilocin. The intermediate, possessing a free primary amine, undergoes nucleophilic attack on ethyl formate to generate a formamide species, a transformation driven by the thermal energy provided at 90-100°C. This formyl group serves as a masked methyl equivalent; upon treatment with the potent reducing agent lithium aluminum hydride (LiAlH4), the carbonyl oxygen is completely removed, and the carbon-nitrogen bond is reduced to a methylene linkage. This reductive amination strategy is highly chemoselective, leaving the newly installed C4-hydroxyl group and the indole aromatic system intact, which is critical for preserving the biological activity of the final natural product. The robustness of this reduction step ensures that even if minor amounts of unreacted formamide remain, they are efficiently converted, contributing to the high overall yield and purity observed in the experimental data.
How to Synthesize Norpsilocin Efficiently
Executing this synthesis requires careful attention to temperature control and reagent addition rates to maximize the formation of the desired C4-isomer while minimizing side reactions. The process begins with the preparation of the catalytic iodine solution, where precise molar ratios of iodine, bismuth acetate, and cuprous chloride are maintained to ensure consistent batch-to-batch reproducibility. Operators must monitor the exotherm during the dropwise addition of the iodine solution to the tryptamine mixture, keeping the temperature strictly between -10°C and 0°C to prevent thermal degradation of the intermediate. Following the isolation of the intermediate, the subsequent formylation and reduction steps demand anhydrous conditions, particularly during the LiAlH4 reduction, to ensure safety and reaction efficiency. For a detailed breakdown of the specific operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Dissolve tryptamine in an acetic acid/sodium acetate/water mixture, cool to -10 to 0°C, and add an iodine-containing solution with Bi(OAc)3 and CuCl catalysts. React to room temperature to form the intermediate.
- Dissolve the intermediate in THF, heat to 90-100°C, and add ethyl formate. After reaction, cool to -10 to 0°C and reduce with LiAlH4 to obtain Norpsilocin.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple yield improvements. By shifting from complex, multi-step traditional syntheses to this concise catalytic protocol, manufacturers can significantly reduce the inventory burden of specialized reagents and intermediates, thereby freeing up working capital and warehouse space. The reliance on commodity chemicals such as tryptamine, ethyl formate, and acetic acid means that sourcing risks are minimized, as these materials are available from multiple global vendors, ensuring supply continuity even during market fluctuations. Furthermore, the simplified purification process reduces the consumption of chromatography media and solvents, leading to substantial cost savings in waste management and utility consumption. This operational efficiency translates directly into a more competitive pricing structure for the final API intermediate, allowing downstream partners to optimize their own cost structures without compromising on quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group chemistry and the reduction in total step count drastically lower the labor and material costs associated with production. By avoiding the use of precious metal catalysts like Palladium or Platinum, which are subject to volatile market pricing and require rigorous removal protocols to meet ppm limits, this Bismuth/Copper system utilizes base metals that are economically stable and easier to manage. The high atom economy of the reductive amination step ensures that the majority of the starting mass is incorporated into the final product, minimizing waste disposal fees. Additionally, the ability to perform reactions in mixed aqueous-organic solvents reduces the volume of pure organic solvents required, further driving down utility and solvent recovery costs.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials like tryptamine mitigates the risk of supply disruptions caused by the scarcity of custom-synthesized building blocks. Since the reaction conditions are relatively mild and do not require specialized high-pressure equipment or extreme cryogenic infrastructure, the process can be easily transferred between different manufacturing sites or CDMO partners without significant capital expenditure. This flexibility ensures that production schedules can be maintained even if one facility faces downtime, providing a robust contingency plan for critical drug development timelines. The simplified workflow also shortens the manufacturing cycle time, allowing for faster turnaround on custom orders and enabling just-in-time delivery models for research clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with exotherms that are manageable through standard cooling jackets and reaction profiles that do not exhibit dangerous runaway potentials. The reduced use of chlorinated solvents and the avoidance of toxic heavy metals simplify the environmental permitting process and reduce the regulatory burden associated with wastewater treatment. By generating fewer byproducts and utilizing a cleaner reaction profile, the facility can achieve higher throughput within existing waste treatment capacity limits. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the corporate sustainability profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and practical manufacturing considerations. Understanding these details is crucial for R&D directors and procurement managers evaluating the feasibility of integrating Norpsilocin into their pipeline. The answers reflect the specific advantages of the Bi-Cu catalytic system and the operational parameters defined in the intellectual property.
Q: What are the key advantages of the Bi-Cu catalyzed route for Norpsilocin?
A: The method utilizes commercially available tryptamine and common reagents, eliminating the need for complex protection groups and reducing overall synthesis costs while maintaining high selectivity.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the process operates under relatively mild conditions with simple workup procedures like extraction and column chromatography, facilitating easier scale-up compared to traditional multi-step alkaloid syntheses.
Q: What is the expected purity profile of the final product?
A: The patent data indicates high selectivity and good separability, with NMR analysis confirming stable peak positions and absence of significant impurity peaks, ensuring a high-purity profile suitable for research applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Norpsilocin Supplier
The technical elegance of the Bi-Cu catalyzed synthesis of Norpsilocin underscores the immense potential for scaling this natural product to meet growing global demand in neuroscience research and pharmaceutical development. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs, guaranteeing that every batch of Norpsilocin meets the highest standards required for clinical and preclinical applications. We understand that consistency is key in drug development, and our process validation protocols are designed to deliver reproducible results batch after batch.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your project economics. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your supply chain objectives. Let us be your partner in turning complex chemical challenges into commercial successes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
