Advanced One-Pot Synthesis of Fluroxypyr-Monoctyl Ester for Industrial Scale-Up
The global demand for high-efficiency, low-residue herbicides continues to drive innovation in agrochemical intermediate manufacturing. A pivotal advancement in this sector is detailed in patent CN103172561A, which discloses a novel one-pot method for synthesizing [(4-amino-3,5-dichloro-6-fluoro-2-pyridinyl)oxy]acetic acid esters, specifically the fluroxypyr-monoctyl ester. This compound serves as a critical active ingredient precursor for broadleaf weed control in major crops like wheat, corn, and grapes. The technical breakthrough lies in the consolidation of multiple reaction stages into a single continuous process, fundamentally altering the economic and environmental footprint of production. By integrating esterification and condensation without intermediate isolation, this methodology addresses long-standing inefficiencies in fine chemical synthesis, offering a robust pathway for reliable agrochemical intermediate suppliers seeking to optimize their portfolios.
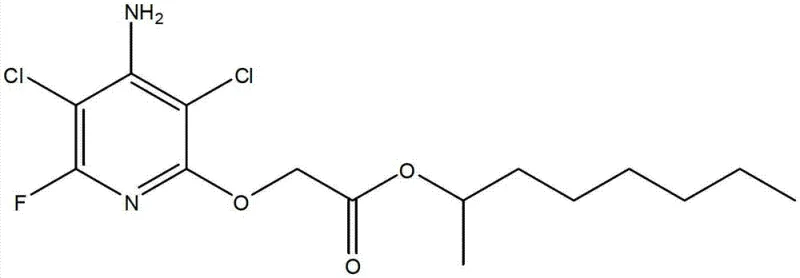
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of fluroxypyr-monoctyl ester has been plagued by complex, multi-step protocols that hinder scalability and cost-efficiency. Prior art, including various US patents, typically relies on a disjointed sequence involving the separate synthesis of glycolic acid esters followed by a distinct condensation step with substituted pyridines. These conventional routes often necessitate rigorous separation and drying procedures between stages, which not only consume substantial energy but also lead to significant material loss during transfer operations. Furthermore, traditional methods frequently generate hazardous pyridol byproducts, creating severe environmental compliance challenges and increasing waste disposal costs. The reliance on harsh derivatization steps, such as methyl sulphonyl activation, introduces unnecessary operational complexity and raw material consumption, ultimately inflating the cost of goods sold and limiting the ability to achieve consistent high-purity outputs required by top-tier pharmaceutical and agrochemical clients.
The Novel Approach
In stark contrast, the one-pot synthesis strategy described in the patent data revolutionizes the production workflow by merging the esterification of glycolic acid with DL-2-octanol and the subsequent condensation with 4-amino-3,5-dichloro-2,6-difluoropyridine into a unified reactor system. This approach effectively reduces the main reaction steps from five down to three, streamlining the entire manufacturing timeline. By eliminating the need for intermediate separation and drying, the process drastically cuts down on equipment occupancy time and energy expenditure. The continuous nature of the reaction allows for the immediate consumption of the esterification product in the condensation phase, minimizing degradation and side reactions. Moreover, the optimized conditions prevent the formation of polluting pyridol compounds, aligning the process with modern green chemistry principles. This seamless integration not only enhances the overall yield to approximately 90% but also simplifies the post-treatment workflow, making it highly attractive for large-scale industrial application.
Mechanistic Insights into One-Pot Esterification and Copper-Catalyzed Condensation
The core of this technological advancement rests on a sophisticated dual-catalyst system that facilitates two distinct chemical transformations within the same reaction medium. Initially, the esterification of glycolic acid with DL-2-octanol is driven by a strong acid catalyst, such as sulfuric acid or p-toluenesulfonic acid, under reduced pressure (0.05-0.4 atm) and elevated temperatures (100-150°C). This step efficiently generates the reactive glycolic acid octyl ester in situ. Following this, the system transitions to a nucleophilic substitution mechanism where the fluorine atom at the 2-position of the pyridine ring is displaced by the oxygen of the glycolic ester. This critical condensation step is accelerated by a specialized catalyst system comprising copper species (Cu, Cu2O, CuO, or Cu halides) potentially synergized with iodide sources. The copper catalyst activates the carbon-halogen bond, lowering the activation energy required for the substitution, while the acid binding agent neutralizes the generated hydrogen fluoride, driving the equilibrium towards product formation.
Impurity control is inherently managed through the precise regulation of reaction parameters and the avoidance of intermediate handling. In traditional multi-step processes, exposure of intermediates to air and moisture during isolation can lead to hydrolysis and oxidation, introducing difficult-to-remove impurities. The one-pot method mitigates these risks by maintaining a closed system throughout the transformation. The specific molar ratios employed—such as a 1:3 to 1:10 ratio of glycolic acid to octanol and a 1.5:1 to 1:1 ratio relative to the pyridine derivative—ensure that reactants are consumed efficiently without excessive accumulation of unreacted starting materials. Furthermore, keeping the condensation temperature below 200°C prevents thermal decomposition and the formation of polymeric tars, which are common contaminants in high-temperature heterocyclic synthesis. This tight control over the reaction environment ensures that the final crude product possesses a purity profile that requires minimal downstream purification, thereby preserving yield and reducing solvent usage.
How to Synthesize Fluroxypyr-Monoctyl Ester Efficiently
The implementation of this one-pot synthesis route requires careful attention to the sequential addition of reagents and the management of thermodynamic conditions to maximize efficiency. The process begins with the charging of glycolic acid, DL-2-octanol, and the acid catalyst into a reactor equipped for water removal, typically via azeotropic distillation. Once the esterification is deemed complete, the reaction mixture is not cooled or isolated; instead, the pyridine substrate, acid binding agent, and copper catalyst are introduced directly into the hot mixture. The detailed standardized synthetic steps, including specific workup procedures and recrystallization protocols using ethanol, are outlined in the technical guide below.
- Perform esterification of glycolic acid and DL-2-octanol using an acid catalyst (e.g., H2SO4) at 100-150°C under reduced pressure.
- Without isolation, add 4-amino-3,5-dichloro-2,6-difluoropyridine, an acid binding agent, and a copper-based catalyst to the reaction mixture.
- Heat the mixture to 100-200°C to complete the condensation reaction, followed by filtration and solvent removal to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis technology translates into tangible strategic benefits that extend beyond simple yield metrics. The consolidation of reaction steps fundamentally alters the cost structure of manufacturing this high-value agrochemical intermediate. By removing unit operations such as filtration, drying, and re-dissolution between steps, the process significantly reduces the consumption of utilities like steam and electricity. This energy efficiency directly contributes to a lower cost base, allowing suppliers to offer more competitive pricing in a volatile raw material market. Additionally, the reduction in processing time means that reactor turnover rates are improved, effectively increasing production capacity without the need for capital-intensive infrastructure expansion. This enhanced throughput capability ensures a more reliable supply of fluroxypyr-monoctyl ester, mitigating the risk of stockouts during peak agricultural seasons.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive solvent usage and the associated recovery costs. Traditional methods often require large volumes of solvents for crystallization and washing at each stage, which incurs both purchase and disposal expenses. By performing the reaction in a single pot, solvent consumption is drastically minimized, and the overall material balance is improved. Furthermore, the high yield of approximately 90% ensures that expensive starting materials, particularly the fluorinated pyridine derivative, are utilized with maximum efficiency, reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Simplified processes are inherently more robust and less prone to operational failures. Multi-step syntheses involve multiple transfer points where human error or equipment malfunction can compromise a batch. The one-pot method reduces these touchpoints, leading to higher batch success rates and more predictable lead times. The use of readily available catalysts and standard reaction conditions (temperatures below 200°C) means that the process can be easily replicated across different manufacturing sites, diversifying the supply base and reducing dependency on single-source producers. This flexibility is crucial for maintaining continuity in the global agrochemical supply chain.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, the environmental profile of a manufacturing process becomes a key determinant of its long-term viability. This novel method avoids the generation of toxic pyridol byproducts, which are difficult to treat and pose significant environmental hazards. The absence of these pollutants simplifies wastewater treatment requirements and reduces the regulatory burden on manufacturing facilities. Consequently, scaling this process from pilot plant to commercial production (100 kgs to 100 MT) is smoother, with fewer permitting obstacles. The eco-friendly nature of the process also aligns with the sustainability goals of major agrochemical corporations, making it a preferred choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of fluroxypyr-monoctyl ester using this advanced one-pot methodology. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of the one-pot synthesis method for fluroxypyr-monoctyl ester?
A: The one-pot method reduces the traditional five-step process to three steps, eliminating intermediate separation and drying. This significantly lowers energy consumption, reduces equipment usage, and minimizes environmental pollution by avoiding the generation of toxic pyridol byproducts.
Q: What catalysts are utilized in this novel synthesis route?
A: The process employs a dual-catalyst system: an acid catalyst such as sulfuric acid or p-toluenesulfonic acid for the initial esterification, followed by a copper-based catalyst (e.g., Cu, Cu2O, CuI) combined with an iodide source for the subsequent nucleophilic substitution condensation.
Q: How does this method impact the overall yield and purity of the agrochemical intermediate?
A: By maintaining the reaction in a single vessel and optimizing conditions (100-200°C, 0.01-1 atm), the method achieves yields approaching 90%. The elimination of transfer steps prevents material loss, ensuring high purity suitable for downstream herbicide formulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluroxypyr-Monoctyl Ester Supplier
The technical potential of this one-pot synthesis route represents a significant leap forward in the manufacturing of herbicide intermediates, offering a blend of efficiency, purity, and environmental stewardship. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to life. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of this synthesis, ensuring stringent purity specifications are met consistently. With our rigorous QC labs and commitment to process safety, we are uniquely positioned to deliver high-quality fluroxypyr-monoctyl ester that meets the exacting standards of the global agrochemical industry.
We invite you to explore how this optimized synthesis route can enhance your supply chain resilience and cost structure. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
