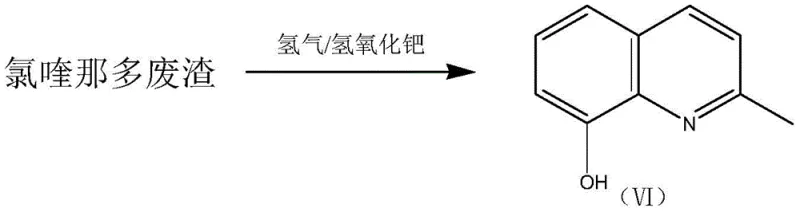
Transforming Chloroquinaldine Waste into High-Purity 8-Hydroxy-2-Methylquinoline via Advanced Hydrogenation
The pharmaceutical and fine chemical industries are increasingly focused on sustainable manufacturing practices that transform hazardous by-products into valuable resources. Patent CN108409651B introduces a groundbreaking methodology for recovering 8-hydroxy-2-methylquinoline from chloroquinaldine waste residue, addressing both economic inefficiency and environmental concerns. This innovative approach utilizes a specialized catalytic hydrogenation process that effectively dechlorinates complex polychlorinated impurities inherent in the waste stream. By leveraging high-activity palladium hydroxide on carbon, the method achieves superior conversion rates under remarkably mild reaction conditions compared to traditional catalysts. This technological advancement not only mitigates the ecological burden of chemical waste but also establishes a circular economy model for reliable pharmaceutical intermediate supplier networks seeking to optimize their raw material sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for handling chloroquinaldine waste often involve incineration or landfilling, which represent a significant loss of potential chemical value and impose heavy environmental compliance costs. Furthermore, attempting to synthesize 8-hydroxy-2-methylquinoline from scratch via direct chlorination of 2-methylquinoline frequently results in the formation of difficult-to-separate polychlorinated by-products, creating a cycle of waste generation. Conventional hydrogenation catalysts such as standard palladium on carbon or Raney nickel have historically shown inadequate reactivity towards the stubborn carbon-chlorine bonds present in these conjugated heterocyclic systems. These older catalytic systems often require harsh conditions involving high temperatures and excessive pressures, which can degrade the sensitive quinoline ring structure and lead to poor selectivity. Consequently, the resulting crude products typically suffer from low purity, necessitating energy-intensive and costly purification steps that erode profit margins. The inability of standard catalysts to efficiently process these specific chlorinated residues has long been a bottleneck in cost reduction in fine chemical manufacturing.
The Novel Approach
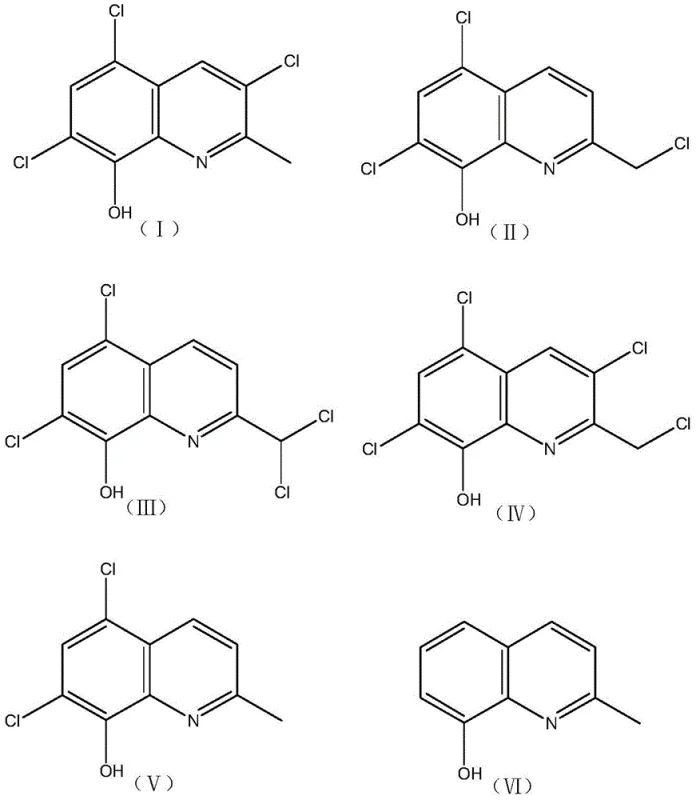
The patented method revolutionizes this landscape by employing palladium hydroxide on carbon, a catalyst uniquely suited for the hydrogenolysis of aryl chlorides in conjugated systems. This novel approach allows for the direct conversion of the waste residue, which contains compounds (I) through (V), into the target molecule (VI) with exceptional efficiency. The process operates at moderate temperatures ranging from 30 to 50°C and hydrogen pressures between 0.3 and 1.5 MPa, significantly reducing energy consumption and safety risks associated with high-pressure reactors. By integrating an acid-binding agent like triethylamine, the reaction neutralizes the hydrochloric acid generated during dechlorination, preventing catalyst poisoning and equipment corrosion. This streamlined workflow transforms a liability into a high-value asset, demonstrating the feasibility of commercial scale-up of complex quinoline derivatives from waste streams. The visual representation of this transformation highlights the structural simplification from polychlorinated precursors to the clean final product.
Mechanistic Insights into Pd(OH)2/C Catalyzed Hydrogenolysis
The core of this technological breakthrough lies in the unique electronic and structural properties of palladium hydroxide supported on carbon, which facilitates the activation of molecular hydrogen and its subsequent transfer to the carbon-chlorine bond. Unlike metallic palladium surfaces which may be sterically hindered or electronically deactivated by the nitrogen atom in the quinoline ring, the hydroxide species offers a distinct active site geometry that promotes oxidative addition into the C-Cl bond. The presence of the acid-binding agent is critical, as it scavenges the liberated chloride ions, shifting the equilibrium towards product formation and maintaining the catalyst in its active state throughout the reaction duration of 3 to 8 hours. This mechanism ensures that even heavily chlorinated impurities, such as the dichloro- and trichloro-quinoline derivatives found in the waste, are sequentially dechlorinated without saturating the aromatic rings. The result is a highly selective process that preserves the integrity of the quinoline scaffold while removing unwanted halogen substituents. Understanding this mechanistic pathway is essential for reducing lead time for high-purity intermediates by minimizing side reactions and maximizing yield.
Impurity control is inherently built into this catalytic system, as the specific activity of the catalyst favors the removal of chlorine atoms over other potential reduction pathways. The waste residue typically contains a complex mixture of isomers and polychlorinated species, as illustrated by the structural diversity of compounds (I) through (V). The robust nature of the Pd(OH)2/C catalyst ensures that these varied precursors are all funneled towards the single desired product, 8-hydroxy-2-methylquinoline (VI), rather than forming a spectrum of partially dechlorinated by-products. This convergence simplifies the downstream purification process, allowing for the achievement of purity levels exceeding 99.0% through standard extraction and distillation techniques. The ability to handle such a heterogeneous feedstock with consistent output quality underscores the reliability of this method for industrial applications. The structural complexity of the starting materials is effectively managed by the catalyst's specificity.
How to Synthesize 8-Hydroxy-2-Methylquinoline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this waste-to-value strategy in a production environment. It begins with the preparation of the reaction mixture, where the chloroquinaldine waste residue is suspended in a polar protic solvent such as methanol or ethanol alongside a stoichiometric amount of triethylamine.
- Mix chloroquinaldine waste residue with an acid-binding agent like triethylamine and a solvent such as methanol or ethanol in a hydrogenation reactor.
- Add 5-10 wt.% palladium hydroxide on carbon catalyst and introduce hydrogen gas at 0.3-1.5 MPa pressure while maintaining temperature between 30-50°C.
- Filter the reaction mixture, dissolve the solid in dilute hydrochloric acid, adjust pH to 5-6 with sodium bicarbonate, extract with organic solvent, and distill to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this technology offers a compelling value proposition by decoupling production costs from volatile raw material markets. By utilizing waste residue as the primary feedstock, manufacturers can significantly lower the direct material costs associated with producing 8-hydroxy-2-methylquinoline, a key intermediate for various antimicrobial and chelating agents. The elimination of expensive precursor synthesis steps and the reduction in waste disposal fees contribute to a substantially improved cost structure. Furthermore, the mild reaction conditions reduce the demand for specialized high-pressure equipment, lowering capital expenditure requirements for facility upgrades. This process enhances supply security by creating an internal loop for material recovery, reducing dependence on external suppliers of fresh raw materials.
- Cost Reduction in Manufacturing: The utilization of waste residue as a feedstock fundamentally alters the cost basis of production, turning a disposal cost into a revenue-generating input. The high selectivity of the palladium hydroxide catalyst minimizes the formation of by-products, which reduces the load on purification units and lowers solvent and energy consumption during downstream processing. Additionally, the extended catalyst life and reduced need for frequent replacement further contribute to operational savings. These factors combine to deliver substantial cost savings without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Implementing this recycling method diversifies the source of raw materials, making the supply chain more resilient to disruptions in the global market for quinoline derivatives. Since the waste residue is often generated onsite or locally during the production of chloroquinaldine, logistics costs and lead times associated with transporting bulk chemicals are drastically reduced. This localized sourcing strategy ensures a steady and predictable flow of materials, enabling better production planning and inventory management. It effectively insulates the manufacturing process from external supply shocks and price fluctuations.
- Scalability and Environmental Compliance: The process is designed for easy industrialization, operating at temperatures and pressures that are standard in most chemical plants, which facilitates seamless scale-up from pilot to commercial production. From an environmental perspective, the method aligns with green chemistry principles by reducing the volume of hazardous waste requiring treatment and lowering the overall carbon footprint of the manufacturing operation. This compliance with stringent environmental regulations mitigates the risk of fines and shutdowns, ensuring long-term operational continuity. It positions the manufacturer as a leader in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this waste recovery technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature. They provide clarity on catalyst selection, reaction parameters, and the quality of the resulting product.
Q: Why is palladium hydroxide on carbon preferred over standard palladium on carbon for this reaction?
A: Palladium hydroxide on carbon demonstrates significantly higher activity for dechlorinating conjugated ring chlorinated substances found in the waste residue, achieving yields of 55-65% compared to only 26% with standard Pd/C, while also ensuring purity levels above 99%.
Q: What are the typical reaction conditions for converting chloroquinaldine waste?
A: The process operates under mild conditions, specifically at temperatures between 30-50°C and hydrogen pressures of 0.3-1.5 MPa, using solvents like methanol or ethanol and triethylamine as an acid-binding agent.
Q: How does this method impact environmental compliance?
A: By recycling hazardous polychlorinated waste residues into valuable pharmaceutical intermediates, this method drastically reduces the volume of chemical waste requiring disposal, thereby lowering environmental treatment costs and improving overall sustainability metrics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Hydroxy-2-Methylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and sustainable synthesis routes for critical pharmaceutical intermediates like 8-hydroxy-2-methylquinoline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to offer high-purity 8-hydroxy-2-methylquinoline that supports the development of next-generation therapeutics.
We invite you to collaborate with us to explore how this advanced recycling technology can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this approach for your business. Let us help you turn your chemical challenges into competitive advantages.
