Revolutionizing Benzofuran Functionalization: A High-Yield Route for Complex Pharmaceutical Intermediates
Revolutionizing Benzofuran Functionalization: A High-Yield Route for Complex Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for safer, more efficient, and scalable chemical processes. A significant breakthrough in this domain is documented in Chinese Patent CN103333144A, which introduces a novel method for synthesizing 2-sulfenyl-3-halo benzofuran compounds. These heterocyclic structures are critical scaffolds in medicinal chemistry, often serving as key building blocks for antiviral, antitumor, and antioxidant agents. The patent details a transformative one-pot reaction strategy that bypasses the severe limitations of classical organolithium chemistry, offering a pathway to high-purity products with exceptional yields. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards greener and more cost-effective manufacturing protocols that align with modern industrial standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the benzofuran ring at the 2-position has been a challenging endeavor, predominantly relying on harsh organometallic strategies. As illustrated in prior art, such as the methods reported by Marino et al. in 1981, the standard protocol involved the use of n-Butyl Lithium (n-BuLi) under strict nitrogen protection and cryogenic conditions.
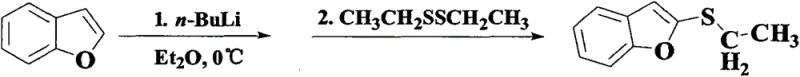
These traditional routes typically require cooling the reaction system to extremely low temperatures, often around -78°C, to generate the necessary organolithium intermediates before introducing sulfur sources like ethyl disulfide or elemental sulfur. Such conditions impose significant burdens on industrial infrastructure, necessitating specialized cryogenic equipment and rigorous safety measures to handle pyrophoric reagents. Furthermore, these multi-step sequences often suffer from poor atom economy and moderate yields, frequently hovering around 64% or lower, which drastically increases the cost of goods sold (COGS) and complicates waste management protocols in large-scale facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN103333144A eliminates the need for hazardous lithiation steps entirely. The invention utilizes a direct reaction between benzofuran compounds, N-halosuccinimides (such as NBS or NIS), and aryl disulfides in a common organic solvent like dichloromethane. This single-stage process operates under remarkably mild thermal conditions, typically ranging from 30°C to 70°C, which dramatically reduces energy consumption and operational complexity. By avoiding moisture-sensitive reagents and cryogenic temperatures, the new method simplifies the reactor setup and enhances process safety, making it an ideal candidate for cost reduction in API manufacturing. The result is a streamlined workflow that delivers 2-sulfenyl-3-halo benzofuran derivatives with yields often exceeding 95%, providing a robust foundation for downstream synthetic applications.
Mechanistic Insights into Electrophilic Substitution and Thiolation
The core innovation of this patent lies in its unique activation mechanism, which facilitates simultaneous halogenation and thiolation of the benzofuran core. Unlike radical-based thiolations that might require transition metal catalysts or harsh initiators, this reaction likely proceeds through an electrophilic activation pathway mediated by the N-halosuccinimide. The N-halosuccinimide serves a dual purpose: it acts as a source of electrophilic halogen species to activate the electron-rich C3 position of the benzofuran ring, while simultaneously facilitating the nucleophilic attack or radical coupling of the disulfide species at the C2 position.
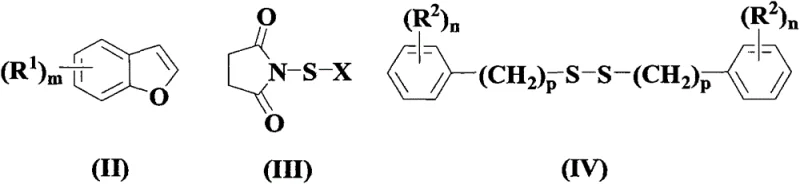
This synergistic interaction allows for precise regioselectivity, ensuring that the halogen and sulfur groups are installed at the desired 3 and 2 positions, respectively, without significant formation of regioisomers. The use of aryl disulfides as sulfur donors is particularly advantageous due to their stability and commercial availability compared to thiols, which can be malodorous and prone to oxidation. The reaction tolerates a wide range of substituents on both the benzofuran ring and the disulfide moiety, including alkyl, alkoxy, and halo groups, demonstrating remarkable substrate scope. This mechanistic efficiency not only maximizes the conversion of raw materials but also minimizes the generation of complex impurity profiles, thereby reducing the burden on purification steps and ensuring the delivery of high-purity OLED material or pharmaceutical precursors.
Furthermore, the resulting 2-sulfenyl-3-halo benzofurans are not merely end-products but serve as highly reactive intermediates for further diversification. The presence of the halogen atom at the 3-position enables subsequent cross-coupling reactions, such as Suzuki-Miyaura couplings, to introduce diverse aryl groups. Additionally, the sulfur moiety can be selectively oxidized to sulfoxides or sulfones using oxidants like m-CPBA, expanding the chemical space accessible from this single scaffold. This versatility is crucial for medicinal chemists aiming to explore structure-activity relationships (SAR) rapidly.
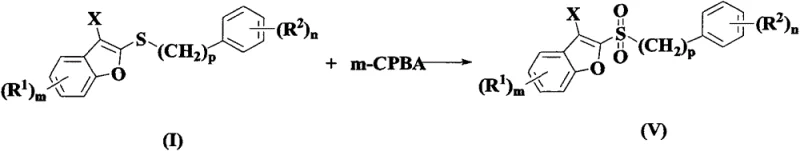
How to Synthesize 2-Sulfenyl-3-Halo Benzofuran Efficiently
The practical implementation of this synthesis is straightforward and designed for reproducibility in both laboratory and pilot plant settings. The process begins with the preparation of a reaction vessel containing dichloromethane, into which the benzofuran substrate, N-halosuccinimide, and the chosen aryl disulfide are introduced in specific molar ratios optimized for maximum conversion. The reaction mixture is then heated to a moderate temperature, typically around 50°C, and stirred for a period of 4 to 10 hours until monitoring indicates complete consumption of the starting materials. Following the reaction, the workup procedure is exceptionally simple, involving filtration to remove succinimide byproducts, followed by solvent removal and purification via standard silica gel chromatography. Detailed standardized synthesis steps see the guide below.
- Charge a dried flask with dichloromethane solvent and add benzofuran compound, N-bromosuccinimide (NBS), and aryl disulfide in a molar ratio of approximately 3.5: 7:1.
- Stir the reaction mixture at a controlled temperature between 30°C and 70°C, preferably around 50°C, for a duration of 4 to 10 hours.
- Upon completion, filter the mixture, remove solvent via rotary evaporation, and purify the residue using silica gel column chromatography to obtain the target product with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible strategic benefits beyond mere chemical elegance. The shift from cryogenic lithiation to ambient thermal processing fundamentally alters the cost structure of producing these valuable intermediates. By eliminating the requirement for expensive, hazardous reagents like n-Butyl Lithium and the associated infrastructure for maintaining ultra-low temperatures, manufacturers can achieve substantial cost savings in raw material procurement and facility maintenance. The simplified operational protocol also reduces the risk of batch failures due to moisture ingress or temperature fluctuations, thereby enhancing overall production reliability and consistency.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and cryogenic reagents directly lowers the input costs for every batch produced. Traditional methods often require stoichiometric amounts of expensive organometallics and specialized quenching procedures, whereas this new route utilizes commodity chemicals like NBS and disulfides which are readily available in bulk quantities. The high atom economy and superior yields mean that less raw material is wasted, effectively reducing the cost per kilogram of the final active pharmaceutical ingredient precursor. Additionally, the simplified workup reduces solvent usage and waste disposal fees, contributing to a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents that have long lead times or limited suppliers poses a significant risk to supply chain continuity. This novel method relies on widely sourced chemicals that are stable and easy to transport, mitigating the risk of production stoppages due to material shortages. The robustness of the reaction conditions ensures that production can be maintained consistently across different seasons and geographical locations, providing a stable supply of high-purity pharmaceutical intermediates to downstream partners. This reliability is critical for meeting tight project timelines and maintaining trust with global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but this one-pot method is inherently scalable due to its lack of exothermic hazards and sensitive intermediates. The use of dichloromethane, a common industrial solvent, allows for easy integration into existing solvent recovery systems, minimizing environmental impact. Furthermore, the absence of heavy metal catalysts simplifies the regulatory compliance landscape, as there is no need for extensive testing and validation for residual metal limits in the final product. This facilitates faster regulatory approval and market entry for new drug candidates utilizing these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-sulfenyl-3-halo benzofuran compounds. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners and technical evaluators.
Q: What are the primary advantages of this new synthesis method over traditional lithiation routes?
A: Unlike traditional methods requiring cryogenic conditions (-78°C) and hazardous n-Butyl Lithium, this novel approach operates at mild temperatures (30-70°C) using stable reagents like NBS and disulfides, significantly improving safety and operational simplicity while achieving yields exceeding 95%.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the process utilizes common solvents like dichloromethane and avoids sensitive organometallic reagents, making it highly suitable for commercial scale-up. The simple workup involving filtration and chromatography ensures robust quality control for large-scale manufacturing.
Q: What downstream derivatives can be prepared from these 2-sulfenyl-3-halo benzofurans?
A: These compounds serve as versatile precursors for further functionalization, including oxidation to sulfoxides and sulfones using m-CPBA, or cross-coupling reactions to form biaryl structures, expanding their utility in drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Sulfenyl-3-Halo Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the technology described in CN103333144A and is fully equipped to leverage this methodology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss your project specifics,索取 specific COA data, and receive comprehensive route feasibility assessments that demonstrate how our advanced synthesis capabilities can drive value for your organization.
