Revolutionizing Antineoplastic Production: A Deep Dive into the Mild Oxidative Synthesis of Tegafur
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical antineoplastic agents, and the synthesis of Tegafur stands as a prime example of this evolution. Patent CN107235967B introduces a groundbreaking methodology that fundamentally alters the manufacturing landscape for this essential fluoropyrimidine derivative. By leveraging a novel oxidative coupling strategy between 5-fluorouracil and tetrahydrofuran, this technology bypasses the severe thermal and catalytic constraints that have plagued traditional production methods for decades. The core innovation lies in the utilization of a specific oxidant and base system that activates the relatively inert tetrahydrofuran ring, allowing for a direct and selective alkylation of the uracil nitrogen under remarkably mild conditions. This approach not only streamlines the synthetic sequence but also aligns perfectly with modern green chemistry principles by minimizing waste generation and eliminating the need for hazardous Lewis acids. For R&D directors and process chemists, this patent represents a significant leap forward in designing robust, scalable, and economically viable routes for complex heterocyclic pharmaceutical intermediates.
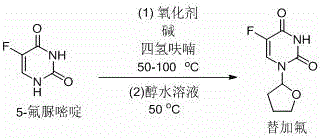
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tegafur has been fraught with significant operational challenges and economic inefficiencies that hinder optimal production scalability. Traditional protocols typically rely on the reaction of 5-fluorouracil with 2,3-dihydrofuran or silylated derivatives, necessitating extreme thermal conditions ranging from 150°C to 180°C to drive the reaction to completion. These high-temperature requirements impose severe stress on reactor materials, increase energy consumption drastically, and often lead to the formation of complex impurity profiles due to thermal degradation of sensitive fluorinated intermediates. Furthermore, conventional routes frequently depend on the addition of stoichiometric amounts of Lewis acids, organic protonic acids, or amine salts to facilitate the coupling, which introduces additional downstream purification burdens and generates substantial quantities of acidic waste streams. The necessity for pre-functionalized starting materials, such as silylated uracil derivatives, further inflates the raw material costs and adds extra synthetic steps, thereby reducing the overall atom economy and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN107235967B offers a streamlined and economically superior alternative that operates under significantly milder parameters. By employing tetrahydrofuran directly as both the solvent and the alkylating source in the presence of an oxidant and a base, the process eliminates the need for pre-activation of the furan ring or the uracil substrate. The reaction proceeds efficiently at temperatures between 50°C and 100°C, which drastically reduces the thermal load on the production equipment and enhances operational safety by avoiding high-pressure scenarios associated with volatile solvents at elevated temperatures. This oxidative strategy utilizes inexpensive and commercially abundant reagents, such as carbon tetrabromide or tert-butyl hydroperoxide, coupled with common inorganic bases like potassium carbonate or sodium hydride. The result is a cleaner reaction profile with fewer byproducts, simplifying the isolation procedure to a mere filtration and recrystallization step, which ultimately delivers the target molecule with exceptional purity levels exceeding 99% without the need for chromatographic purification.
Mechanistic Insights into Oxidative N-Alkylation
The mechanistic underpinning of this synthesis involves a sophisticated interplay between base-mediated deprotonation and oxidant-driven radical or electrophilic activation of the tetrahydrofuran solvent. Initially, the base abstracts the acidic proton from the N1 position of the 5-fluorouracil ring, generating a nucleophilic uracil anion that is primed for substitution. Simultaneously, the oxidant interacts with the tetrahydrofuran, likely generating an alpha-oxy radical or a reactive oxocarbenium ion species at the C2 position of the ether ring. This activation lowers the energy barrier for the nucleophilic attack by the uracil anion, facilitating the formation of the N-C bond that links the heterocyclic base to the tetrahydrofuran moiety. The choice of oxidant is critical, as it must be strong enough to activate the ether linkage but selective enough to avoid over-oxidation of the sensitive fluoropyrimidine core, ensuring that the fluorine atom at the C5 position remains intact throughout the transformation.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed thermal routes. The mild basic conditions prevent the hydrolysis of the urea linkage within the uracil ring, a common degradation pathway observed under harsh acidic conditions. Additionally, the use of tetrahydrofuran as the reaction medium ensures high solubility of the intermediates, promoting homogeneous reaction kinetics that minimize localized hot spots where polymerization or charring could occur. The subsequent workup involving aqueous ethanol serves a dual purpose: it quenches any remaining reactive oxidant species and selectively precipitates the product while keeping inorganic salts and polar byproducts in solution. This inherent selectivity in the reaction mechanism and workup design is what allows the process to achieve such high purity specifications consistently, making it highly attractive for the manufacture of high-purity pharmaceutical intermediates where strict impurity limits are mandated by regulatory bodies.
How to Synthesize Tegafur Efficiently
The practical implementation of this synthesis route is designed for ease of operation and reproducibility, making it an ideal candidate for technology transfer from the laboratory to the pilot plant. The process begins with the suspension of 5-fluorouracil in tetrahydrofuran, followed by the sequential addition of the base and the oxidant under an inert atmosphere to prevent moisture interference. The reaction mixture is then heated to a moderate temperature, typically around 60°C, and maintained for a period of approximately 8 hours to ensure complete conversion of the starting material. Upon completion, the reaction is cooled, and the insoluble inorganic salts are removed via simple filtration, leaving a clear solution of the crude product. The solvent is evaporated, and the residue is subjected to a recrystallization process using a mixture of ethanol and water, which yields the final white crystalline solid of Tegafur. For detailed standardized operating procedures and specific parameter optimizations, please refer to the guide below.
- Combine 5-fluorouracil, a suitable base (e.g., potassium carbonate), and an oxidant (e.g., carbon tetrabromide) in tetrahydrofuran solvent.
- Heat the reaction mixture to 50-100°C under nitrogen protection and stir for approximately 8 hours to facilitate the coupling reaction.
- Filter the solid byproducts, remove the solvent, and recrystallize the residue from an aqueous ethanol solution at 50°C to obtain high-purity Tegafur.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this oxidative synthesis technology translates into tangible improvements in cost structure and supply reliability. The shift away from specialized, high-cost reagents like silylated precursors and Lewis acids towards commodity chemicals such as tetrahydrofuran and potassium carbonate creates a much more resilient supply chain that is less susceptible to market volatility. The elimination of high-temperature processing steps not only reduces utility costs but also extends the lifespan of production assets by minimizing thermal stress and corrosion, leading to lower maintenance expenditures and reduced downtime. Furthermore, the simplified purification protocol reduces the consumption of organic solvents and silica gel typically required for column chromatography, resulting in substantial cost savings in waste disposal and material usage. These factors collectively contribute to a more competitive cost of goods sold (COGS), enabling manufacturers to offer high-quality intermediates at more aggressive price points while maintaining healthy margins.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts and pre-functionalized starting materials with bulk industrial chemicals significantly lowers the direct material costs associated with production. By operating at lower temperatures, the process also achieves significant energy savings, as less heating power is required to maintain reaction conditions compared to traditional methods that operate near 180°C. The high atom economy of the reaction ensures that a larger proportion of the raw materials end up in the final product, reducing the effective cost per kilogram of the active pharmaceutical ingredient. Additionally, the simplified workup reduces labor hours and solvent recovery costs, further enhancing the overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key raw materials, including 5-fluorouracil and tetrahydrofuran, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures or safety incidents related to high-pressure or high-temperature operations. This reliability ensures consistent delivery timelines for downstream API manufacturers, allowing for better inventory planning and reduced safety stock requirements. The stability of the process also facilitates long-term supply agreements, providing partners with the confidence needed to scale their own production volumes without fear of supply interruptions.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, such as reduced waste generation and the absence of heavy metal catalysts, align perfectly with increasingly stringent environmental regulations. Scaling this reaction from kilogram to tonnage levels is straightforward because the exothermic profile is manageable and does not require specialized high-pressure reactors. The use of ethanol and water for recrystallization minimizes the release of volatile organic compounds (VOCs) into the atmosphere, supporting corporate sustainability goals. This environmental compatibility reduces the regulatory burden and permitting time for new production lines, accelerating the time-to-market for new generic formulations or clinical supplies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and practical application data. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this oxidative synthesis method over traditional high-temperature routes?
A: Unlike conventional methods requiring temperatures of 150-180°C and Lewis acid catalysts, this patented process operates at mild temperatures (50-100°C) without expensive additives, significantly reducing energy consumption and equipment corrosion risks while improving atom economy.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The method utilizes a simple filtration step followed by recrystallization in aqueous ethanol, which effectively removes inorganic salts and side products, consistently achieving HPLC purity levels greater than 99% suitable for API manufacturing.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the use of common industrial solvents like tetrahydrofuran and ethanol, combined with mild reaction conditions and readily available raw materials like 5-fluorouracil, makes this process highly amenable to large-scale commercial manufacturing with minimal safety hazards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tegafur Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN107235967B for the production of high-value oncology intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from process development to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative coupling reactions, including rigorous safety protocols for handling oxidants and specialized containment for fluorinated compounds. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay every batch.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of high-purity Tegafur for your global markets.
