Scalable Green Synthesis of Phenylglycine Using Superparamagnetic Ionic Liquid Catalysts
Introduction to Next-Generation Phenylglycine Manufacturing
The global demand for high-quality pharmaceutical intermediates continues to surge, driven by the expanding antibiotic and polypeptide hormone markets. A pivotal development in this sector is detailed in patent CN111269134B, which discloses a highly efficient preparation method for phenylglycine and its derivatives. This technology leverages a novel superparamagnetic nanoparticle loaded ionic liquid catalyst to facilitate the reaction between benzaldehyde and sodium cyanide under remarkably mild conditions. Unlike conventional synthetic routes that often struggle with environmental compliance and operational complexity, this innovation offers a streamlined pathway to phenylhydantoin, which is subsequently hydrolyzed to yield phenylglycine. For R&D directors and procurement specialists alike, this represents a significant leap forward in process chemistry, promising not only enhanced reaction kinetics but also a drastic simplification of downstream processing through magnetic separation techniques.
The strategic importance of phenylglycine cannot be overstated, as it serves as a critical building block for beta-lactam antibiotics such as ampicillin, cephalexin, and cefradine. As the antibiotic industry evolves, the pressure to secure reliable sources of these key intermediates intensifies. The methodology outlined in CN111269134B addresses these supply chain vulnerabilities by introducing a catalytic system that combines high specific surface area with exceptional reusability. By operating at temperatures between 50-60°C and normal pressure, the process mitigates the safety risks associated with high-energy reactions while maintaining impressive conversion rates. This balance of safety, efficiency, and environmental stewardship positions the technology as a cornerstone for modern, sustainable pharmaceutical manufacturing infrastructures seeking to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of phenylglycine has relied heavily on the sodium cyanide method, a process fraught with significant operational and environmental challenges. Traditional protocols typically necessitate the use of large quantities of excessive ammonia water to drive the reaction forward, resulting in the generation of substantial volumes of wastewater and solid residues. The treatment of this waste stream is not only technically difficult but also incurs considerable costs related to environmental compliance and disposal. Furthermore, the harsh reaction conditions often required in legacy processes can lead to the formation of unwanted by-products, complicating the purification stages and ultimately reducing the overall yield of the desired API intermediate. These inefficiencies create bottlenecks in the supply chain, making it difficult for manufacturers to guarantee consistent quality and timely delivery in a cost-effective manner.
Beyond the immediate chemical inefficiencies, the reliance on non-recoverable catalysts or stoichiometric reagents in older methods exacerbates the economic burden on production facilities. The inability to easily separate catalysts from the reaction mixture often leads to product contamination, necessitating rigorous and expensive purification steps to meet stringent pharmaceutical purity standards. Additionally, the high energy consumption associated with maintaining elevated temperatures and pressures in conventional reactors contributes to a larger carbon footprint, which is increasingly scrutinized by regulatory bodies and corporate sustainability mandates. These cumulative factors highlight the urgent need for a paradigm shift towards greener, more atom-economical synthetic routes that can deliver high-purity phenylglycine without compromising on economic viability or environmental responsibility.
The Novel Approach
In stark contrast to these legacy issues, the novel approach described in the patent utilizes a superparamagnetic nanoparticle loaded ionic liquid catalyst to revolutionize the synthesis landscape. This advanced catalytic system enables the reaction to proceed efficiently at mild temperatures of 50-60°C, significantly lowering energy requirements while maintaining high reaction rates. The core innovation lies in the magnetic properties of the catalyst support, which allows for instantaneous and complete separation of the catalyst from the reaction mixture simply by applying an external magnetic field. This eliminates the need for complex filtration or centrifugation steps, thereby streamlining the workflow and reducing the potential for product loss during isolation. The result is a cleaner reaction profile with fewer impurities, directly translating to higher yields and reduced downstream processing costs.
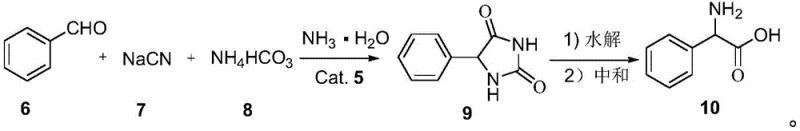
Furthermore, the versatility of this novel approach is demonstrated by its compatibility with a wide range of solvents, including methanol, ethanol, and toluene, providing flexibility for process optimization. The reaction stoichiometry is carefully balanced, utilizing a molar ratio of benzaldehyde to sodium cyanide to solid ammonium salt to ammonia water of approximately 1:1-2:1-5:1-5, ensuring optimal resource utilization. As illustrated in the reaction scheme, the process smoothly converts benzaldehyde derivatives into phenylhydantoin intermediates, which are then hydrolyzed to the final amino acid product. This robust methodology not only enhances the purity of the final output, achieving content levels up to 99%, but also establishes a scalable framework that is ideally suited for the commercial scale-up of complex amino acid derivatives required by the global pharmaceutical market.
Mechanistic Insights into Superparamagnetic Nanoparticle Catalysis
The efficacy of this synthesis route is fundamentally rooted in the unique structural and chemical properties of the superparamagnetic nanoparticle loaded ionic liquid catalyst. The catalyst is constructed upon a core of Fe3O4/SiO2 magnetic nanoparticles, which provides the essential superparamagnetic behavior allowing for rapid magnetic separation. This core is functionalized with an ionic liquid moiety, specifically an imidazolium-based structure, which acts as the active catalytic site. The synthesis of this sophisticated catalyst involves a multi-step grafting process where 3-chloropropyltriethoxysilane reacts with imidazole to form an intermediate, which is then anchored onto the silica-coated magnetic particles. Finally, the surface is modified with 3-chloro glycerol to complete the ionic liquid functionality, creating a highly active and stable heterogeneous catalyst system.
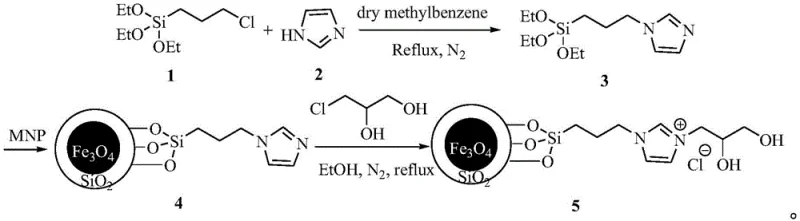
From a mechanistic perspective, the high specific surface area of the nanoparticles ensures that a maximum number of active sites are available for the reactants, facilitating the nucleophilic attack of cyanide on the benzaldehyde carbonyl group. The ionic liquid environment likely stabilizes the transition states and intermediates, such as the aminonitrile species, promoting the cyclization to phenylhydantoin with high selectivity. Moreover, the heterogeneous nature of the catalyst prevents the leaching of active species into the solution, which is a common issue with homogeneous catalysts that can contaminate the final product. The ability to recover the catalyst simply by magnetic decantation means that the active sites remain intact and accessible for subsequent cycles, as evidenced by the patent data showing consistent activity over multiple reuses. This mechanistic robustness is crucial for maintaining batch-to-batch consistency, a key requirement for any reliable phenylglycine supplier aiming to serve regulated pharmaceutical markets.
How to Synthesize Phenylglycine Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the reaction mixture in a three-neck flask equipped with magnetic stirring, where benzaldehyde, sodium cyanide, solid ammonium salt, and ammonia water are combined in a selected solvent. The choice of solvent can influence the reaction kinetics, with polar protic solvents like methanol often yielding superior results. Once the reagents are mixed, the superparamagnetic catalyst is introduced, and the system is heated to the optimal range of 50-60°C. Monitoring the reaction progress via HPLC is recommended to determine the exact endpoint, typically occurring within 8-14 hours, ensuring complete conversion of the starting materials before proceeding to the workup phase.
- Prepare the reaction mixture by combining benzaldehyde, sodium cyanide, solid ammonium salt, and ammonia water in a suitable solvent such as methanol or ethanol.
- Add the superparamagnetic nanoparticle loaded ionic liquid catalyst to the mixture and stir at 50-60°C under normal pressure for 8-14 hours.
- Recover the catalyst using an external magnetic field, then hydrolyze, decolor, and neutralize the reaction liquid to obtain high-purity phenylglycine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnetic nanocatalyst technology offers profound strategic advantages that extend beyond mere chemical efficiency. The primary value proposition lies in the significant cost reduction in pharmaceutical intermediates manufacturing achieved through process intensification. By eliminating the need for excessive ammonia and reducing the volume of wastewater generated, facilities can drastically lower their expenditure on waste treatment and environmental compliance. Furthermore, the simplicity of the catalyst recovery process removes the necessity for expensive filtration equipment and reduces labor hours associated with downstream processing. These operational efficiencies translate directly into a more competitive cost structure, allowing manufacturers to offer high-purity phenylglycine at more attractive price points without sacrificing margin.
- Cost Reduction in Manufacturing: The elimination of complex separation steps and the reduction in waste disposal costs contribute to a leaner manufacturing model. Since the catalyst can be reused multiple times without significant loss of activity, the recurring cost of catalyst consumption is minimized. This contrasts sharply with traditional methods where reagents are consumed stoichiometrically or catalysts are lost during filtration. The qualitative reduction in solvent usage and energy consumption due to mild reaction conditions further compounds these savings, creating a financially robust production process that is resilient to fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent production output, which is critical for maintaining supply continuity for downstream antibiotic manufacturers. The ease of catalyst recovery means that production downtime between batches is significantly reduced, enhancing overall throughput. Additionally, the use of readily available starting materials like benzaldehyde and sodium cyanide ensures that the supply chain is not dependent on exotic or scarce reagents. This stability is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond swiftly to market demands and inventory requirements.
- Scalability and Environmental Compliance: The technology is inherently scalable, moving seamlessly from laboratory benchtop to commercial production scales. The magnetic separation mechanism works equally well in large reactors as it does in small flasks, avoiding the engineering challenges often associated with scaling up filtration processes. From an environmental standpoint, the drastic reduction in hazardous waste aligns with global sustainability goals and stricter regulatory frameworks. This proactive approach to green chemistry not only mitigates regulatory risk but also enhances the corporate image of the manufacturer as a responsible and forward-thinking partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is vital for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the operational parameters, catalyst lifecycle, and product quality assurance associated with this patented technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of the magnetic nanocatalyst over traditional methods?
A: The magnetic nanocatalyst allows for simple recovery via an external magnetic field, eliminating complex filtration steps. It operates under mild conditions (50-60°C) and significantly reduces wastewater generation compared to traditional sodium cyanide processes requiring excessive ammonia.
Q: Can the catalyst be reused without loss of activity?
A: Yes, the patent data indicates that after the reaction, the catalyst can be recovered, washed with ethanol, and dried. It can be repeatedly used multiple times with no obvious reduction in catalytic activity, ensuring consistent yield and content.
Q: What is the typical yield and purity achievable with this method?
A: Experimental examples in the patent demonstrate yields ranging from 87% to 93% with product content (purity) consistently reaching 97% to 99% as detected by HPLC, depending on the specific solvent and substrate used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylglycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN111269134B for the production of critical pharmaceutical intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that the consistency of phenylglycine is paramount for the efficacy of the final antibiotic products, and our state-of-the-art facilities are designed to deliver exactly that level of reliability.
We invite global partners to collaborate with us to leverage these cutting-edge synthetic routes for their specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your total cost of ownership. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a supplier, but a strategic ally dedicated to advancing your pharmaceutical development goals through superior chemistry and supply chain excellence.
