Advanced Synthesis of Naproxen Nitroxy Derivatives for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for non-steroidal anti-inflammatory drug (NSAID) derivatives that offer enhanced therapeutic profiles without compromising manufacturing efficiency. Patent CN1678560A introduces a groundbreaking process for the preparation of nitroxy derivatives of Naproxen, a compound known for its potential in nitric oxide donation and reduced gastrointestinal toxicity. This technology addresses critical stability issues found in prior art by utilizing Naproxen salts instead of unstable acyl chlorides, ensuring the preservation of stereochemical integrity throughout the synthesis. The method demonstrates exceptional control over impurity profiles, achieving enantiomeric excess levels that match the starting material, which is a significant advancement for high-purity API intermediate production. By shifting the paradigm from acid chloride chemistry to nucleophilic substitution on stable alkyl derivatives, this process offers a reliable pathway for manufacturers aiming to produce complex esterified NSAIDs with consistent quality. The implications for supply chain stability are profound, as the starting materials are commercially available and less prone to degradation during storage and transport.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Naproxen nitroxy esters often rely on the formation of 2-arylpropionic acid halides, specifically acyl chlorides, as key intermediates. These acid chlorides are notoriously chemically unstable and susceptible to racemization, which poses a severe risk to the enantiomeric purity of the final product. Furthermore, the corresponding nitrooxy alkyl alcohols required for esterification are often unstable in their pure form, making isolation and purification difficult and costly. The formation of multiple by-products during the reaction necessitates complex downstream processing, which increases waste generation and reduces overall yield. These factors collectively contribute to higher production costs and inconsistent batch quality, creating significant bottlenecks for procurement teams managing tight margins. The reliance on such sensitive intermediates also complicates logistics, as specialized storage conditions are required to prevent decomposition before the reaction even begins.
The Novel Approach
The innovative method disclosed in the patent circumvents these challenges by employing Naproxen salts, such as sodium or potassium salts, which are chemically and enantiomerically stable. Instead of reacting with alcohols, the process utilizes nitrooxy alkyl derivatives equipped with suitable leaving groups, such as halogens or sulfonates, which are significantly more stable than their alcohol counterparts. This strategic shift allows for a cleaner reaction profile with minimal by-product formation, simplifying the purification process and enhancing overall efficiency. The structural versatility of this approach is evident in the wide range of acceptable linkers and substituents, allowing for the customization of the final molecule for specific pharmacological needs. 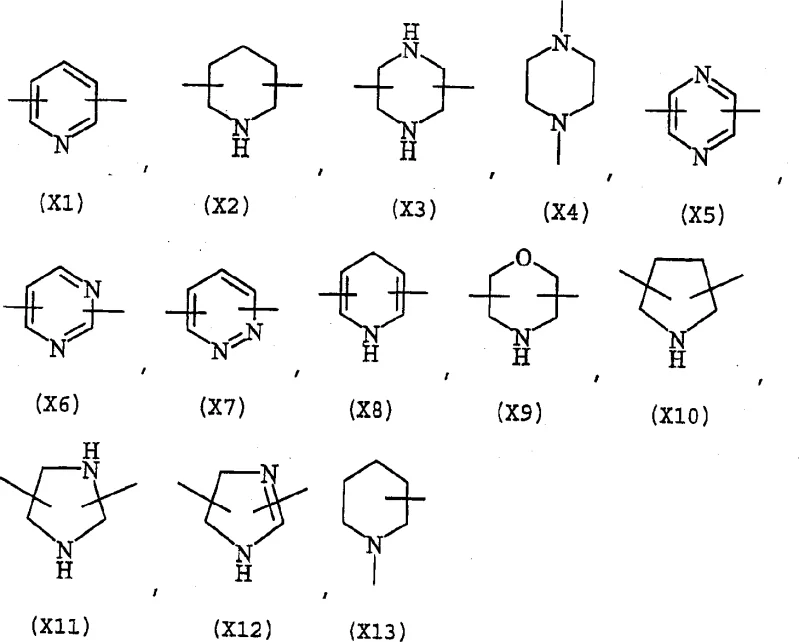 This flexibility enables manufacturers to tailor the physicochemical properties of the derivative without altering the core synthetic strategy. Consequently, this novel approach not only improves product quality but also streamlines the manufacturing workflow, making it an attractive option for large-scale commercial production.
This flexibility enables manufacturers to tailor the physicochemical properties of the derivative without altering the core synthetic strategy. Consequently, this novel approach not only improves product quality but also streamlines the manufacturing workflow, making it an attractive option for large-scale commercial production.
Mechanistic Insights into Salt-Based Nucleophilic Substitution
The core of this synthesis lies in a nucleophilic substitution reaction where the carboxylate anion of the Naproxen salt attacks the electrophilic carbon of the nitrooxy alkyl derivative. This mechanism avoids the harsh conditions typically associated with acid chloride formation, thereby protecting the chiral center at the alpha-position of the propionic acid moiety. The use of polar aprotic solvents such as dimethylformamide (DMF) or acetonitrile facilitates the dissolution of the salt and enhances the nucleophilicity of the carboxylate ion. Reaction temperatures can be carefully controlled between 0°C and 100°C to optimize the rate of substitution while minimizing side reactions. The presence of a good leaving group on the alkyl chain ensures that the reaction proceeds to completion with high selectivity, resulting in the desired ester bond formation without compromising the nitrooxy functionality. This mechanistic pathway is robust and reproducible, providing a solid foundation for process validation and regulatory compliance in pharmaceutical manufacturing.
Impurity control is inherently built into this mechanism due to the stability of the starting materials and the specificity of the substitution reaction. Unlike acid chloride routes where racemization can occur via enolization, the salt-based method maintains the stereochemical configuration of the Naproxen backbone. The absence of reactive acid chlorides also eliminates the risk of forming symmetric anhydrides or other chloride-related impurities. Post-reaction workup typically involves simple aqueous extraction and chromatography, which effectively removes inorganic salts and unreacted starting materials. 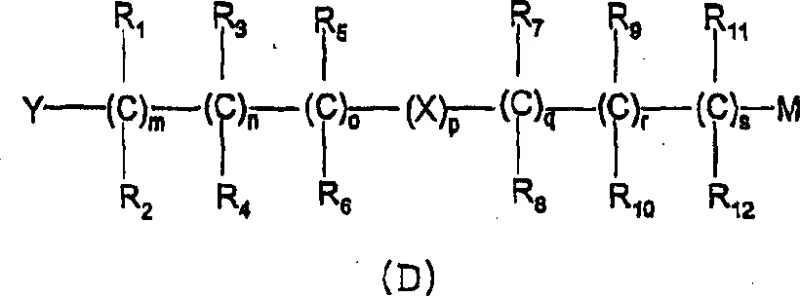 The ability to achieve enantiomeric excess greater than 99% directly from the reaction mixture underscores the precision of this chemical transformation. For R&D directors, this level of purity control reduces the burden on analytical testing and ensures that the final API meets stringent pharmacopeial standards without extensive reprocessing.
The ability to achieve enantiomeric excess greater than 99% directly from the reaction mixture underscores the precision of this chemical transformation. For R&D directors, this level of purity control reduces the burden on analytical testing and ensures that the final API meets stringent pharmacopeial standards without extensive reprocessing.
How to Synthesize Naproxen Nitroxy Ester Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometry to maximize yield and purity. The process begins with the preparation of the Naproxen salt in situ or using a pre-formed commercial salt, followed by the addition of the nitrooxy alkyl derivative under controlled temperature conditions. Detailed standard operating procedures regarding mixing rates, addition sequences, and quenching methods are essential for consistent results. The patent provides specific examples using potassium bicarbonate and various leaving groups to demonstrate the versatility of the method across different scales.
- Prepare Naproxen salt (e.g., sodium salt) in a polar aprotic solvent like DMF.
- React the salt with a nitrooxy alkyl derivative containing a suitable leaving group (e.g., bromide or tosylate).
- Purify the resulting ester via chromatography to achieve high enantiomeric purity (>99% e.e.).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial benefits by eliminating the need for expensive and hazardous acid chloride reagents. The use of stable salts and alkyl derivatives reduces raw material costs and minimizes the risks associated with handling reactive intermediates. Supply chain reliability is enhanced because the starting materials are commercially available and have longer shelf lives compared to traditional reagents. This stability allows for better inventory management and reduces the frequency of urgent procurement actions driven by material degradation. Additionally, the simplified workup process reduces solvent consumption and waste disposal costs, contributing to a more sustainable and cost-effective manufacturing operation. These factors collectively lower the total cost of ownership for the production of Naproxen derivatives, making it a financially sound choice for procurement managers.
- Cost Reduction in Manufacturing: The elimination of acid chloride formation steps removes the need for thionyl chloride or oxalyl chloride, which are costly and generate significant hazardous waste. By using stable salts, the process avoids the losses associated with racemization and by-product formation, leading to higher effective yields. The simplified purification process further reduces operational expenses related to chromatography media and solvent recovery. These cumulative efficiencies translate into significant cost savings without the need for complex equipment upgrades or specialized containment systems.
- Enhanced Supply Chain Reliability: The stability of Naproxen salts and nitrooxy alkyl derivatives ensures that raw materials can be sourced globally without stringent cold chain requirements. This robustness mitigates the risk of supply disruptions caused by material degradation during transit or storage. Procurement teams can negotiate better terms with suppliers due to the commoditized nature of the stable starting materials. Furthermore, the consistency of the reaction output reduces the variability in production schedules, allowing for more accurate demand forecasting and inventory planning.
- Scalability and Environmental Compliance: The reaction conditions are mild and compatible with standard stainless steel reactors, facilitating easy scale-up from pilot to commercial production. The absence of corrosive acid chlorides reduces equipment maintenance costs and extends the lifespan of manufacturing assets. Waste streams are less hazardous, simplifying compliance with environmental regulations and reducing disposal fees. This alignment with green chemistry principles enhances the corporate sustainability profile of the manufacturing facility while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this route for their production lines.
Q: Why is the salt-based method superior to acyl chloride routes for Naproxen derivatives?
A: The salt-based method avoids the use of chemically unstable acyl chlorides which are prone to racemization. Using stable Naproxen salts ensures the enantiomeric excess is retained throughout the synthesis, resulting in a higher purity final product.
Q: What are the stability advantages of the nitrooxy alkyl derivatives used in this process?
A: Unlike nitrooxy alkyl alcohols which are often unstable and difficult to isolate, the nitrooxy alkyl derivatives with leaving groups (such as bromides or tosylates) are chemically stable. This stability simplifies storage and handling, reducing supply chain risks.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes commercially available starting materials and standard solvents like DMF or acetonitrile. The reaction conditions are mild (0°C to 100°C), and the workup involves conventional extraction and chromatography, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naproxen Nitroxy Ester Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of salt-based esterification and can ensure that your Naproxen derivatives are produced with stringent purity specifications. We operate rigorous QC labs that employ advanced chiral HPLC and spectroscopic methods to verify enantiomeric excess and impurity profiles. Our commitment to quality ensures that every batch meets the high standards required by global pharmaceutical regulators. By leveraging our infrastructure, clients can accelerate their time-to-market while maintaining full control over product quality and supply continuity.
We invite you to discuss how this advanced synthesis route can optimize your supply chain and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your project. We are dedicated to forming long-term partnerships that drive innovation and efficiency in the pharmaceutical intermediate sector. Let us help you navigate the complexities of chemical synthesis with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
