Scalable Electro-Oxidative Synthesis of Fluoroalkyl Sulfonamide Polycyclic Compounds for Pharma
Scalable Electro-Oxidative Synthesis of Fluoroalkyl Sulfonamide Polycyclic Compounds for Pharma
Introduction: A Green Breakthrough in Sulfonamide Chemistry
The pharmaceutical industry continuously seeks robust methodologies to construct complex heterocyclic scaffolds, particularly those containing sulfur and fluorine motifs which are pivotal for metabolic stability and bioavailability. As illustrated by the prevalence of sulfonamide drugs such as Celecoxib and Piroxicam shown in 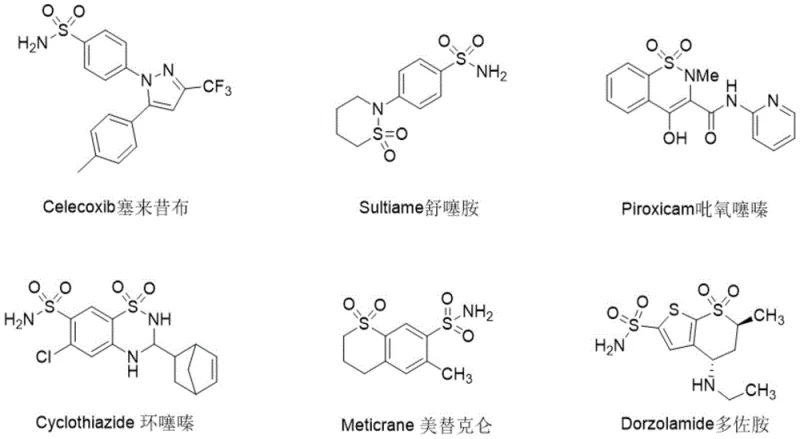 , the arylsulfonamide group remains a cornerstone of modern medicinal chemistry. Addressing the critical need for sustainable synthesis, patent CN114934287A discloses a novel electro-oxidative method for synthesizing fluoroalkyl-substituted sulfonamide polycyclic compounds. This technology represents a paradigm shift from traditional stoichiometric oxidant-dependent routes to a cleaner, electricity-driven radical cascade process. By leveraging anodic oxidation to generate fluoroalkyl radicals in situ, this approach bypasses the need for hazardous chemical oxidants and expensive transition metal catalysts, offering a compelling value proposition for manufacturers of high-purity pharmaceutical intermediates seeking to reduce their environmental footprint while maintaining rigorous quality standards.
, the arylsulfonamide group remains a cornerstone of modern medicinal chemistry. Addressing the critical need for sustainable synthesis, patent CN114934287A discloses a novel electro-oxidative method for synthesizing fluoroalkyl-substituted sulfonamide polycyclic compounds. This technology represents a paradigm shift from traditional stoichiometric oxidant-dependent routes to a cleaner, electricity-driven radical cascade process. By leveraging anodic oxidation to generate fluoroalkyl radicals in situ, this approach bypasses the need for hazardous chemical oxidants and expensive transition metal catalysts, offering a compelling value proposition for manufacturers of high-purity pharmaceutical intermediates seeking to reduce their environmental footprint while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing fluoroalkylated sulfonamide frameworks often rely heavily on transition metal catalysis, such as copper or palladium systems, coupled with stoichiometric amounts of strong chemical oxidants. These conventional methods present significant challenges for large-scale manufacturing, primarily due to the stringent regulatory limits on heavy metal residues in active pharmaceutical ingredients (APIs). The removal of trace metals requires additional purification steps, such as scavenging resins or repeated recrystallizations, which drastically increase production costs and reduce overall process efficiency. Furthermore, the use of harsh chemical oxidants generates substantial amounts of toxic waste, complicating waste management and increasing the environmental burden of the manufacturing facility. Thermal conditions required for these reactions are often vigorous, leading to potential safety hazards and limited functional group tolerance, which restricts the structural diversity accessible to process chemists during lead optimization phases.
The Novel Approach
In stark contrast, the electro-oxidative strategy detailed in the patent utilizes electrons as the primary reagent to drive the transformation, effectively replacing chemical oxidants with electrical energy. As depicted in the general reaction scheme in 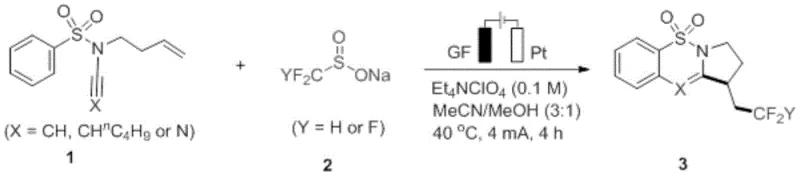 , the process involves the direct anodic oxidation of sodium fluoroalkyl sulfinates to generate reactive fluoroalkyl radicals under mild conditions. This metal-free protocol operates at moderate temperatures, typically around 40°C, and utilizes readily available graphite felt and platinum electrodes. The absence of transition metals not only simplifies the workup procedure by eliminating metal scavenging steps but also ensures a cleaner impurity profile, which is critical for regulatory compliance. This methodology enables the efficient construction of complex 6-6-5 fused ring systems through a tandem radical cyclization sequence, providing a versatile platform for accessing diverse chemical space that was previously difficult or costly to explore using traditional thermal methods.
, the process involves the direct anodic oxidation of sodium fluoroalkyl sulfinates to generate reactive fluoroalkyl radicals under mild conditions. This metal-free protocol operates at moderate temperatures, typically around 40°C, and utilizes readily available graphite felt and platinum electrodes. The absence of transition metals not only simplifies the workup procedure by eliminating metal scavenging steps but also ensures a cleaner impurity profile, which is critical for regulatory compliance. This methodology enables the efficient construction of complex 6-6-5 fused ring systems through a tandem radical cyclization sequence, providing a versatile platform for accessing diverse chemical space that was previously difficult or costly to explore using traditional thermal methods.
Mechanistic Insights into Electro-Oxidative Radical Cyclization
The core innovation of this technology lies in its elegant radical cascade mechanism, which is initiated by the anodic oxidation of the fluoroalkyl sulfinate anion. As detailed in the mechanistic pathway shown in 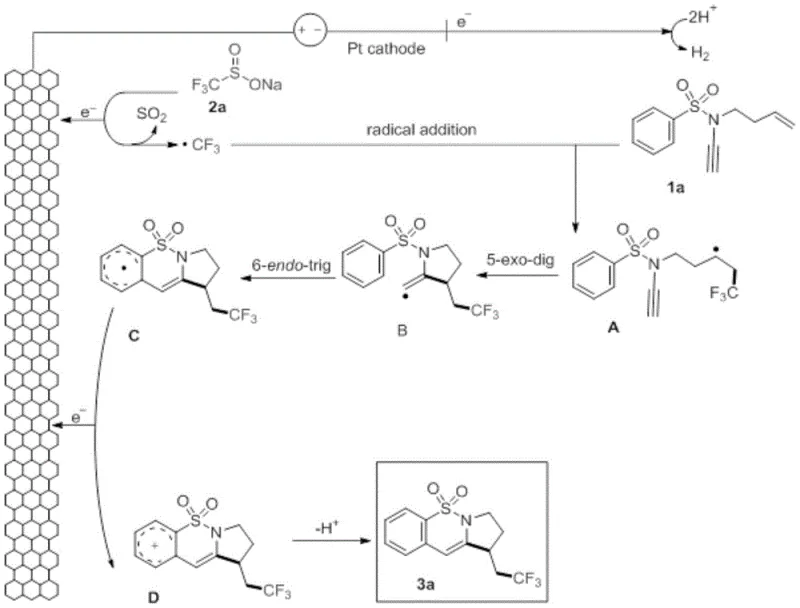 , the process begins with the loss of an electron at the anode surface to generate a trifluoromethyl or difluoromethyl radical species. This highly reactive radical selectively attacks the terminal alkene moiety of the N,N-disubstituted sulfonamide substrate, forming a new carbon-carbon bond and generating a secondary alkyl radical intermediate. This intermediate subsequently undergoes a rapid 5-exo-dig cyclization onto the pendant alkyne or cyano group, constructing the five-membered ring component of the final scaffold. The resulting benzylic alkenyl radical then adds to the aromatic ring, followed by a second oxidation event at the anode to form an aryl cation, which finally undergoes deprotonation to restore aromaticity and yield the stable 6-6-5 fused polycyclic product. This intricate sequence demonstrates precise control over radical reactivity, ensuring high regioselectivity and minimizing side reactions that typically plague free-radical chemistry.
, the process begins with the loss of an electron at the anode surface to generate a trifluoromethyl or difluoromethyl radical species. This highly reactive radical selectively attacks the terminal alkene moiety of the N,N-disubstituted sulfonamide substrate, forming a new carbon-carbon bond and generating a secondary alkyl radical intermediate. This intermediate subsequently undergoes a rapid 5-exo-dig cyclization onto the pendant alkyne or cyano group, constructing the five-membered ring component of the final scaffold. The resulting benzylic alkenyl radical then adds to the aromatic ring, followed by a second oxidation event at the anode to form an aryl cation, which finally undergoes deprotonation to restore aromaticity and yield the stable 6-6-5 fused polycyclic product. This intricate sequence demonstrates precise control over radical reactivity, ensuring high regioselectivity and minimizing side reactions that typically plague free-radical chemistry.
From an impurity control perspective, this mechanism offers distinct advantages over thermal radical initiators like AIBN or peroxides, which often decompose unpredictably and generate nitrogenous or oxygenated byproducts. The electrochemical generation of radicals is tunable via current density, allowing operators to match the rate of radical formation with the consumption rate, thereby maintaining a low steady-state concentration of reactive intermediates. This "slow-release" kinetic profile suppresses bimolecular radical termination pathways, such as dimerization, which are common sources of difficult-to-remove impurities. Furthermore, the use of a divided or undivided cell configuration with specific electrolytes like tetraethylammonium perchlorate ensures efficient charge transfer while minimizing competing oxidation of the organic substrate at the anode. For R&D directors, understanding this mechanism is crucial for troubleshooting and optimizing the process for new substrates, as the electronic properties of the aromatic ring and the steric bulk of the N-substituents can be fine-tuned to enhance the efficiency of the cyclization steps without altering the fundamental green chemistry principles of the route.
How to Synthesize Fluoroalkyl Sulfonamide Derivatives Efficiently
Implementing this electrochemical synthesis requires careful attention to reaction parameters to maximize yield and reproducibility. The patent provides specific experimental protocols, such as Example 1, where N-(but-3-en-1-yl)-N-ethynylbenzenesulfonamide reacts with sodium trifluoromethanesulfinate to yield the target tricyclic compound. As shown in the specific reaction example in 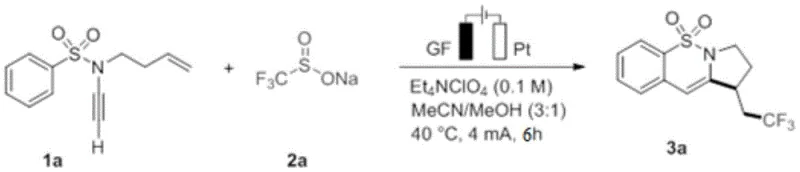 , the process utilizes a constant current of 4 mA in a MeCN/MeOH solvent system. The detailed standardized synthesis steps for replicating this high-value transformation are outlined below, providing a clear roadmap for process chemists to adapt this methodology for their specific target molecules.
, the process utilizes a constant current of 4 mA in a MeCN/MeOH solvent system. The detailed standardized synthesis steps for replicating this high-value transformation are outlined below, providing a clear roadmap for process chemists to adapt this methodology for their specific target molecules.
- Prepare the reaction mixture by combining N,N-disubstituted sulfonamide derivatives and sodium fluoroalkyl sulfinate compounds in a mixed solvent of acetonitrile and methanol with tetraethylammonium perchlorate electrolyte.
- Conduct the electrification reaction using a graphite felt anode and platinum cathode under constant current conditions (preferably 4 mA) at 40°C for approximately 6 hours.
- Purify the resulting crude mixture via silica gel column chromatography using n-hexane and ethyl acetate to isolate the target 6-6-5 skeleton fluoroalkyl sulfonamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electro-oxidative technology translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by eliminating the need for precious metal catalysts like palladium or rhodium, manufacturers are no longer exposed to the volatile pricing and geopolitical supply risks associated with these critical minerals. Additionally, the removal of heavy metals from the process flow significantly reduces the complexity and cost of wastewater treatment and solid waste disposal, aligning with increasingly stringent global environmental regulations. This streamlined process flow enhances overall equipment effectiveness (OEE) by reducing batch cycle times associated with extensive purification and metal scavenging operations, thereby increasing plant throughput without requiring capital investment in new reactors.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and stoichiometric chemical oxidants directly lowers the bill of materials (BOM) cost for each batch. Furthermore, the simplified downstream processing, which avoids complex metal removal steps, reduces the consumption of auxiliary materials such as scavenging resins and specialized filtration media. This cumulative effect leads to substantial cost savings in the manufacturing of complex pharmaceutical intermediates, making the final API more price-competitive in the global market while improving margin resilience against raw material inflation.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary "reagent" decouples production capacity from the availability of specialized chemical reagents that may face supply shortages. The starting materials, such as sodium fluoroalkyl sulfinates and simple sulfonamide derivatives, are commodity chemicals with robust, multi-vendor supply bases. This diversification of input materials mitigates the risk of production stoppages due to single-source supplier failures, ensuring consistent delivery schedules for downstream customers and strengthening the reliability of the pharmaceutical supply chain.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, moving from milligram-scale optimization to kilogram-scale production by increasing electrode surface area or using flow chemistry configurations. The mild reaction conditions (40°C, atmospheric pressure) reduce energy consumption for heating and cooling compared to high-temperature thermal processes. Moreover, the absence of toxic heavy metal waste simplifies regulatory compliance and environmental permitting, facilitating faster site approvals and reducing the long-term liability associated with hazardous waste storage and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electro-oxidative synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for stakeholders evaluating this technology for potential integration into their manufacturing portfolios.
Q: Does this electrochemical method require transition metal catalysts?
A: No, the method described in patent CN114934287A operates without transition metal catalysts, utilizing anodic oxidation to generate radicals directly, which significantly simplifies downstream purification and eliminates heavy metal residue concerns.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal results are achieved using a constant current of 4 mA at 40°C in a MeCN/MeOH (3:1) solvent system with Et4NClO4 as the electrolyte, typically yielding the target polycyclic compounds in moderate to good yields depending on substituents.
Q: Can this process be scaled for industrial API intermediate production?
A: Yes, the use of undivided cells with robust electrode materials like graphite felt and platinum, combined with mild temperatures and common solvents, suggests high potential for commercial scale-up compared to sensitive photochemical or high-pressure thermal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl Sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electro-organic synthesis in modernizing pharmaceutical manufacturing. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this electro-oxidative route can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of fluoroalkyl sulfonamide intermediate meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in electrochemical synthesis can accelerate your development timeline and optimize your production costs.
