Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Triazole Synthesis
The 1,2,4-triazole scaffold represents a cornerstone structural motif in modern medicinal chemistry, underpinning the efficacy of numerous blockbuster drugs and functional materials. As illustrated in the diverse array of bioactive molecules below, ranging from iron chelators like deferasirox to antifungal agents like fluconazole and aromatase inhibitors like letrozole, the incorporation of this nitrogen-rich heterocycle is critical for biological activity. Furthermore, the strategic introduction of a trifluoromethyl group onto this scaffold offers profound advantages, including enhanced metabolic stability, improved lipophilicity, and increased electronegativity, which are essential parameters for optimizing drug pharmacokinetics.
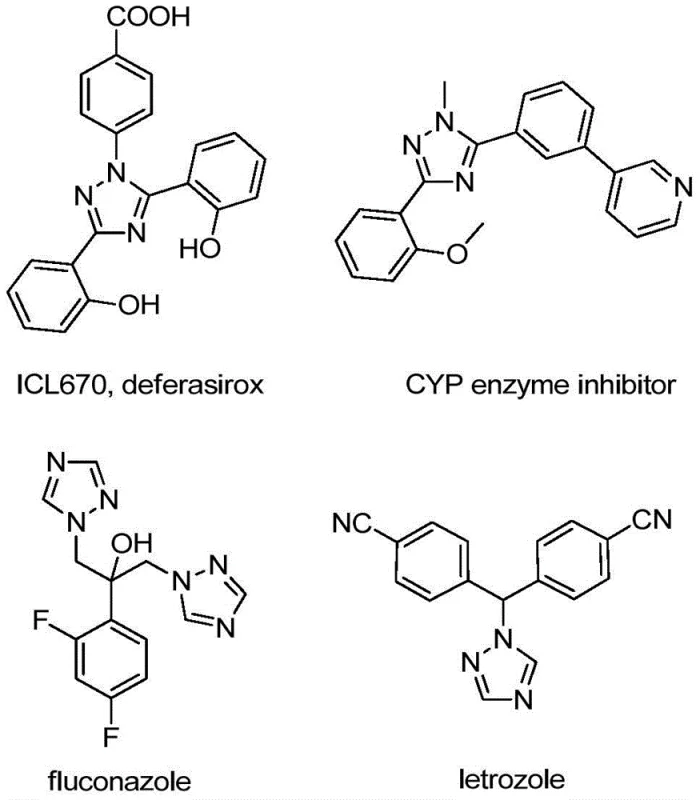
In response to the growing demand for efficient access to these valuable building blocks, patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses the longstanding challenges associated with traditional synthetic routes by employing a non-metallic iodine-promoted strategy. By utilizing inexpensive and readily available hydrazones and trifluoroethylimidoyl chlorides as starting materials, this novel approach eliminates the need for complex pre-functionalization or hazardous reagents. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology offers a robust pathway to high-purity intermediates with a significantly simplified operational profile.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has been fraught with synthetic inefficiencies and safety concerns. Literature reports generally categorize existing methods into two cumbersome streams: the direct trifluoromethylation of pre-synthesized heterocycles or the coupling of trifluoromethyl synthons with suitable substrates. The former often relies on specialized and costly trifluoromethylating reagents that drive up the cost of goods sold (COGS) and complicate waste management. The latter frequently employs unstable or dangerous species such as trifluorodiazoethane, which poses significant safety risks during scale-up due to its explosive potential. Additionally, many traditional protocols necessitate the use of expensive transition metal catalysts, which introduce the risk of heavy metal contamination in the final API, requiring rigorous and costly purification steps to meet regulatory standards.
The Novel Approach
The methodology outlined in CN110467579B represents a paradigm shift towards greener and more economical synthesis. As depicted in the general reaction scheme below, the process utilizes a simple condensation between a trifluoroethylimidoyl chloride and a hydrazone, promoted by elemental iodine and sodium acetate. This route bypasses the need for pre-formed heterocycles or dangerous diazo compounds entirely. The reaction proceeds smoothly in common organic solvents like dichloroethane at moderate temperatures, typically around 80°C. Crucially, the process does not require stringent anhydrous or anaerobic conditions, allowing for execution in standard glassware without specialized inert atmosphere equipment. This operational simplicity translates directly into reduced capital expenditure for manufacturing facilities and lower barrier to entry for commercial production.
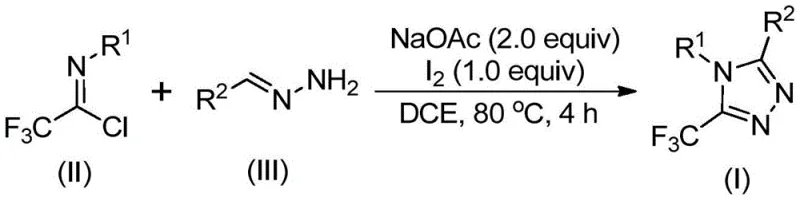
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for ensuring consistent quality and impurity control during manufacturing. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the imidoyl chloride, generating a trifluoroacetamidine intermediate. This step is facilitated by sodium acetate, which acts as a mild base to scavenge the generated hydrochloric acid, driving the equilibrium forward without causing decomposition of sensitive functional groups. The subsequent isomerization sets the stage for the critical ring-closing event, distinguishing this pathway from linear oligomerization side reactions.
Following the initial condensation, the addition of elemental iodine triggers a base-promoted oxidative iodination, forming a key iodinated intermediate. This species then undergoes an intramolecular electrophilic substitution reaction, effectively closing the five-membered triazole ring. The final step involves aromatization to yield the stable 5-trifluoromethyl-1,2,4-triazole product. This specific mechanistic sequence is highly advantageous for impurity control; by avoiding radical pathways often associated with metal catalysis, the process minimizes the formation of complex byproduct mixtures. The result is a cleaner crude reaction profile, which significantly eases the burden on downstream purification units and ensures the delivery of high-purity pharmaceutical intermediates.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for maximum efficiency and reproducibility in a pilot or production plant setting. The protocol involves charging a reactor with sodium acetate, the specific trifluoroethylimidoyl chloride derivative, and the chosen hydrazone substrate in an aprotic solvent. The mixture is heated to facilitate the initial coupling, followed by the controlled addition of iodine to drive the oxidative cyclization to completion. Detailed standardized operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for maintaining batch-to-batch consistency. Please refer to the specific technical guidelines below for the precise step-by-step execution.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation and cyclization.
- Add elemental iodine to the system and continue reacting for 1 to 2 hours to promote oxidative aromatization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The elimination of precious metal catalysts removes a major cost driver and supply chain bottleneck, as the volatility of metal prices no longer impacts the stability of raw material costs. Furthermore, the avoidance of hazardous reagents like trifluorodiazoethane reduces the regulatory burden and insurance costs associated with handling explosive materials. The robustness of the reaction conditions allows for flexible scheduling and reduced downtime, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive catalytic systems with commodity chemicals like elemental iodine and sodium acetate. By removing the requirement for transition metals, manufacturers eliminate the costly and time-consuming steps associated with metal scavenging and residual metal testing, which are mandatory for API compliance. This streamlining of the downstream processing workflow results in substantial cost savings per kilogram of produced intermediate, improving the overall margin profile for the final drug product.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically hydrazones and trifluoroethylimidoyl chlorides, are derived from widely available aldehydes and amines. This broad substrate availability ensures that the supply chain is not dependent on single-source suppliers for exotic reagents. Additionally, the tolerance of the reaction to ambient moisture and oxygen means that production is less susceptible to delays caused by equipment failure in inert gas systems, thereby securing a more consistent and reliable delivery schedule for downstream customers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns perfectly with modern green chemistry principles. The absence of heavy metals simplifies waste treatment and disposal, reducing the environmental footprint of the manufacturing site. The mild reaction temperatures and the use of standard organic solvents facilitate easy scale-up from laboratory grams to multi-ton commercial production without the need for specialized high-pressure or cryogenic reactors, ensuring that the process remains safe and compliant with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the process parameters and scope. These insights are derived directly from the experimental data and embodiments provided in the patent literature, ensuring accuracy and relevance for process development scientists. Understanding these nuances is critical for successful technology transfer and scale-up.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine as a promoter instead of toxic or expensive transition metal catalysts, significantly simplifying downstream purification and reducing heavy metal residue risks.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction typically proceeds in an aprotic organic solvent like dichloroethane at moderate temperatures around 80°C. It does not require strict anhydrous or anaerobic conditions, making it operationally simple.
Q: Can this method be scaled for industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded to the gram level and beyond. The use of cheap, commercially available starting materials and mild conditions supports large-scale manufacturing feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate adheres to the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your timeline and reduce your overall production costs.
