Efficient Synthesis of Pyrimidine Furanone for Commercial Scale Pharmaceutical Manufacturing
Efficient Synthesis of Pyrimidine Furanone for Commercial Scale Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective pathways for critical intermediates. Patent CN115557901A introduces a groundbreaking methodology for the high-efficiency production of pyrimidine furanone, chemically known as (E)-2-[2-(6-chloropyrimidin-4-yloxy)phenyl]-3-methoxymethyl acrylate. This compound serves as a pivotal building block in the synthesis of novel antioxidants, pharmaceutical agents, and food additives, necessitating a supply chain capable of delivering high-purity materials consistently. The disclosed technology fundamentally reimagines the synthetic landscape by replacing multi-step, low-yield protocols with a streamlined four-step sequence that leverages accessible raw materials like phenol and chloroacetyl chloride. 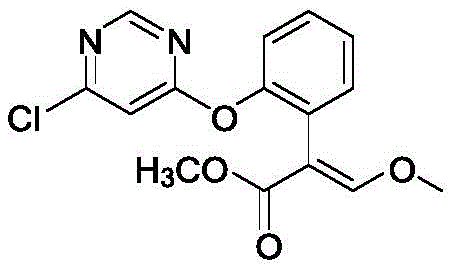 This strategic shift not only enhances the overall atom economy but also addresses critical pain points related to waste generation and operational complexity. For R&D directors and procurement managers alike, understanding the nuances of this patented route is essential for securing a reliable pharmaceutical intermediate supplier who can navigate the complexities of modern chemical manufacturing while maintaining stringent quality standards.
This strategic shift not only enhances the overall atom economy but also addresses critical pain points related to waste generation and operational complexity. For R&D directors and procurement managers alike, understanding the nuances of this patented route is essential for securing a reliable pharmaceutical intermediate supplier who can navigate the complexities of modern chemical manufacturing while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrimidine furanone has been plagued by inefficiencies that hinder large-scale commercial viability. As documented in prior art such as Patent EP0242081, traditional routes often rely on o-hydroxyphenylacetic acid as a starting material, necessitating a cumbersome series of protection and deprotection steps.  Specifically, the hydroxyl group must be protected with a benzyl group, followed by formylation and methylation, before the protecting group is removed to allow for the final coupling reaction. This linear progression results in a significantly elongated production timeline and accumulates substantial material losses at each stage, leading to a low total yield that drives up the cost per kilogram. Furthermore, alternative methods reported in patents like CN102311392A, which utilize one-pot or stepwise approaches with DABCO catalysts, often suffer from poor atom economy and the formation of difficult-to-separate closed-loop by-products. These side reactions not only compromise the purity of the final API intermediate but also create significant downstream processing burdens, making it challenging to achieve the consistent quality required for regulatory compliance in pharmaceutical manufacturing.
Specifically, the hydroxyl group must be protected with a benzyl group, followed by formylation and methylation, before the protecting group is removed to allow for the final coupling reaction. This linear progression results in a significantly elongated production timeline and accumulates substantial material losses at each stage, leading to a low total yield that drives up the cost per kilogram. Furthermore, alternative methods reported in patents like CN102311392A, which utilize one-pot or stepwise approaches with DABCO catalysts, often suffer from poor atom economy and the formation of difficult-to-separate closed-loop by-products. These side reactions not only compromise the purity of the final API intermediate but also create significant downstream processing burdens, making it challenging to achieve the consistent quality required for regulatory compliance in pharmaceutical manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN115557901A presents a radically simplified and economically superior pathway. The new strategy initiates with the direct esterification of phenol and chloroacetyl chloride to form phenyl chloroacetate, bypassing the need for expensive protected starting materials entirely. 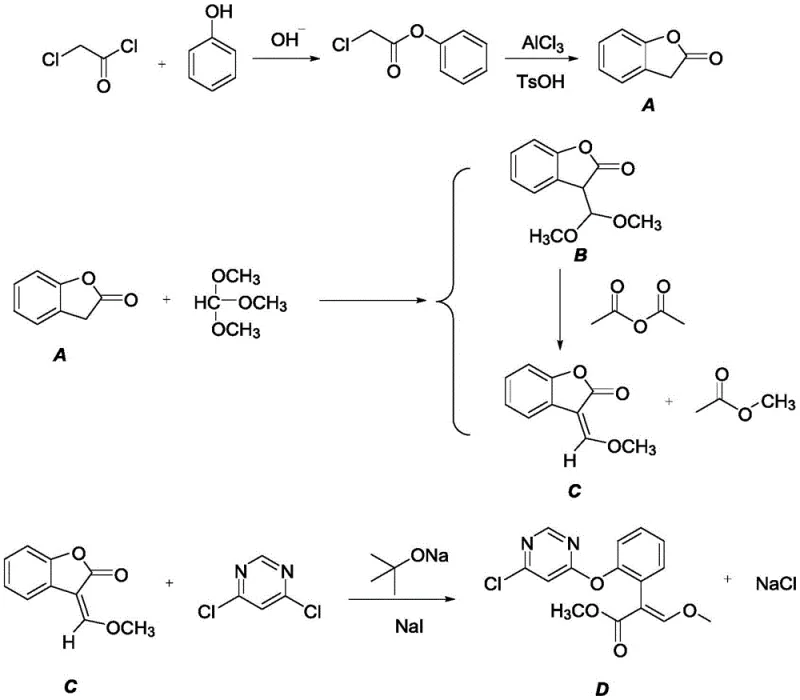 This intermediate then undergoes a highly efficient Friedel-Crafts cyclization to generate the benzofuranone core, a transformation that is accelerated by the use of specialized supported catalysts. Subsequent formylation with trimethyl orthoformate and acetic anhydride, followed by a final coupling with 4,6-dichloropyrimidine, completes the synthesis in just four distinct operations. This reduction in step count drastically minimizes the cumulative yield loss typically associated with multi-step syntheses. Moreover, the operational simplicity allows for easier scale-up, as the reaction conditions are optimized to avoid the extreme temperatures and pressures often required in older methodologies. For supply chain heads, this translates to a more resilient production schedule with reduced lead times for high-purity pharmaceutical intermediates, ensuring a steady flow of materials to meet market demand without the bottlenecks inherent in conventional processing.
This intermediate then undergoes a highly efficient Friedel-Crafts cyclization to generate the benzofuranone core, a transformation that is accelerated by the use of specialized supported catalysts. Subsequent formylation with trimethyl orthoformate and acetic anhydride, followed by a final coupling with 4,6-dichloropyrimidine, completes the synthesis in just four distinct operations. This reduction in step count drastically minimizes the cumulative yield loss typically associated with multi-step syntheses. Moreover, the operational simplicity allows for easier scale-up, as the reaction conditions are optimized to avoid the extreme temperatures and pressures often required in older methodologies. For supply chain heads, this translates to a more resilient production schedule with reduced lead times for high-purity pharmaceutical intermediates, ensuring a steady flow of materials to meet market demand without the bottlenecks inherent in conventional processing.
Mechanistic Insights into Supported Lewis Acid Catalyzed Cyclization
The cornerstone of this innovative synthesis lies in the second step: the intramolecular Friedel-Crafts acylation that constructs the benzofuranone ring system. Unlike traditional homogeneous Lewis acids which can be corrosive and difficult to remove, this patent employs a supported catalyst system, specifically aluminum trichloride loaded with p-toluenesulfonic acid. This heterogeneous catalytic approach offers profound mechanistic advantages by providing a high density of active acidic sites on a solid support, which facilitates the activation of the carbonyl group in the phenyl chloroacetate precursor. The activated carbonyl becomes highly electrophilic, promoting a rapid nucleophilic attack by the ortho-position of the aromatic ring. This cyclization is critical because it establishes the rigid heterocyclic scaffold necessary for the biological activity of the final derivative. The use of the supported catalyst not only accelerates the reaction kinetics, allowing completion within 5-8 hours at moderate temperatures of 40-100°C, but also simplifies the workup procedure significantly. By enabling simple filtration for catalyst recovery, the process eliminates the need for extensive aqueous washes to remove metal residues, thereby reducing wastewater treatment costs and environmental impact.
Furthermore, the final coupling reaction between the methoxymethylene benzofuranone intermediate and 4,6-dichloropyrimidine is meticulously controlled to ensure regioselectivity and minimize impurity formation. The reaction utilizes sodium tert-butoxide as a strong base to deprotonate the active methylene group, generating a nucleophilic enolate species. In the presence of a phase transfer catalyst or halide source like sodium iodide, this enolate selectively attacks the C-4 position of the dichloropyrimidine ring, displacing the chlorine atom via a nucleophilic aromatic substitution mechanism. The choice of solvent, such as methylcyclohexane or toluene, plays a vital role in solubilizing the organic intermediates while maintaining the stability of the reactive anionic species. This precise control over reaction parameters prevents the formation of bis-substituted by-products or hydrolysis of the sensitive ester moieties. For R&D teams, understanding these mechanistic details is crucial for troubleshooting potential scale-up issues and ensuring that the impurity profile remains within the strict limits required for clinical-grade materials, ultimately safeguarding the integrity of the downstream drug development pipeline.
How to Synthesize Pyrimidine Furanone Efficiently
The practical implementation of this synthesis route requires careful attention to reaction conditions and reagent stoichiometry to maximize the reported yields of over 80%. The process begins with the preparation of phenyl chloroacetate, where phenol is reacted with chloroacetyl chloride in the presence of a base such as potassium tert-butoxide at temperatures ranging from 0°C to 150°C. Following isolation, the crude ester is subjected to the key cyclization step using the supported aluminum trichloride catalyst in a solvent like dichloromethane. The resulting benzofuranone is then formylated using trimethyl orthoformate and acetic anhydride, a step that can be performed under solvent-free conditions to further enhance green chemistry metrics. Finally, the coupling with 4,6-dichloropyrimidine is executed in a non-polar solvent with sodium tert-butoxide and a catalytic amount of sodium iodide.
- Prepare phenyl chloroacetate via esterification of phenol and chloroacetyl chloride using a base like potassium tert-butoxide.
- Perform Friedel-Crafts cyclization using a supported aluminum trichloride catalyst to generate the benzofuranone intermediate.
- React the benzofuranone with trimethyl orthoformate in the presence of acetic anhydride to form the methoxymethylene intermediate.
- Couple the intermediate with 4,6-dichloropyrimidine using sodium tert-butoxide and a catalyst like sodium iodide to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method offers tangible economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing commodity chemicals like phenol and chloroacetyl chloride instead of specialized protected intermediates, the cost of goods sold is significantly reduced. This shift mitigates the risk of supply disruptions associated with niche starting materials and leverages the stable pricing of bulk petrochemical derivatives. Additionally, the elimination of protection and deprotection steps removes entire unit operations from the manufacturing train, which directly correlates to lower energy consumption and reduced labor hours per batch. The ability to recover and reuse the supported catalyst further contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing the consumption of expensive Lewis acids and reducing the volume of hazardous waste requiring disposal. These factors combine to create a more competitive cost structure, allowing suppliers to offer better pricing without compromising on margin.
- Cost Reduction in Manufacturing: The streamlined four-step sequence eliminates the need for expensive protecting groups and the associated reagents for their installation and removal, which traditionally account for a significant portion of raw material costs. By avoiding these steps, the process reduces the total mass of reagents required per kilogram of product, leading to substantial cost savings. Furthermore, the high efficiency of the supported catalyst system minimizes catalyst loading and enables recycling, which lowers the operational expenditure related to consumable materials. The reduction in processing steps also means less solvent usage and lower utility costs for heating and cooling, contributing to a leaner and more cost-effective production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: Relying on widely available commodity feedstocks such as phenol ensures a robust and resilient supply chain that is less susceptible to the volatility of specialized chemical markets. The simplified process flow reduces the number of potential failure points in the manufacturing line, thereby increasing the reliability of delivery schedules. With fewer intermediate isolation and purification steps, the overall cycle time from raw material intake to finished goods is shortened, allowing for faster response to customer demand fluctuations. This agility is critical for maintaining continuous supply to downstream pharmaceutical manufacturers, preventing production stoppages and ensuring that critical drug development timelines are met without delay.
- Scalability and Environmental Compliance: The use of heterogeneous catalysts and the avoidance of harsh deprotection conditions make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduced generation of saline wastewater and organic waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and associated disposal fees. The high atom economy of the route ensures that a greater proportion of input materials end up in the final product, minimizing the environmental footprint of the manufacturing process. This sustainability profile is increasingly important for multinational corporations seeking to partner with suppliers who demonstrate a commitment to green chemistry and responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of pyrimidine furanone using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: What is the expected yield of the new pyrimidine furanone synthesis method?
A: According to patent CN115557901A, the technical solution achieves a product yield of 80% or more, with specific experimental examples demonstrating yields up to 93.7%.
Q: How does the new catalyst system improve the process compared to conventional methods?
A: The use of supported p-toluenesulfonic acid aluminum trichloride significantly accelerates the reaction speed and facilitates catalyst recovery, unlike traditional homogeneous catalysts that are difficult to separate.
Q: What are the main advantages regarding impurity control in this route?
A: By avoiding complex protection and deprotection steps found in prior art, this route minimizes side reactions and closed-loop by-products, resulting in higher purity and easier separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of global pharmaceutical and agrochemical projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering pyrimidine furanone with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific reaction conditions required by this patented route, including the management of supported catalysts and moisture-sensitive reagents, guaranteeing a product that meets your exacting requirements for downstream synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced production capabilities can support your long-term strategic goals. Let us be your partner in driving innovation and efficiency in your chemical manufacturing operations.
