Scalable Manufacturing of High-Purity Eriodictyol via Mild One-Pot Oxidative Demethylation
Introduction to Advanced Eriodictyol Manufacturing Technologies
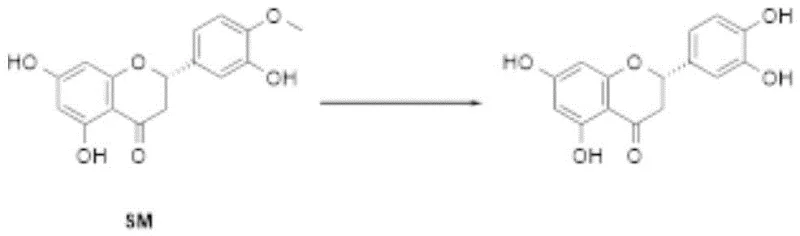
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for high-value flavonoid intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN116239558A, which discloses a novel preparation method for Eriodictyol, a bioactive flavonoid known for its antioxidant and anti-inflammatory properties. This patent introduces a streamlined one-pot oxidative-reductive strategy that converts Hesperetin directly into Eriodictyol, bypassing the severe limitations associated with traditional demethylation techniques. By utilizing 2-iodoxybenzoic acid (IBX) as a selective oxidant followed by a mild reduction step, the process achieves high purity without the need for complex chromatographic purification or hazardous reagents. For R&D directors and procurement specialists, this methodology represents a paradigm shift towards safer, more cost-effective manufacturing of complex pharmaceutical intermediates, offering a reliable supply chain solution for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eriodictyol has been plagued by significant operational hazards and purification bottlenecks that hinder large-scale production. One prevalent prior art method involves the catalytic hydrogenation of Luteolin using palladium on carbon, which unfortunately yields a product with non-absolute configuration, necessitating additional, costly chiral resolution steps to isolate the biologically active enantiomer. Another common approach utilizes anhydrous aluminum chloride in pyridine at elevated temperatures to effect demethylation; however, this reagent is extremely hygroscopic and corrosive, posing severe safety risks during handling. Furthermore, the quenching of aluminum salts generates intense exothermic reactions and leads to stubborn emulsification during the aqueous workup, drastically complicating the extraction process and generating substantial volumes of corrosive liquid waste. These factors collectively render conventional methods economically inefficient and environmentally unsustainable for modern commercial scale-up.
The Novel Approach
In stark contrast, the innovative protocol described in the patent employs a mild, two-stage one-pot sequence that elegantly circumvents these industrial pain points. The process initiates with the selective oxidation of Hesperetin using IBX in a solvent like tetrahydrofuran, forming a reactive intermediate solution without disturbing the sensitive chiral centers of the molecule. Subsequently, a water-soluble reducing agent, such as sodium dithionite, is introduced directly into the reaction mixture to complete the transformation to Eriodictyol. This tandem operation eliminates the need for isolating unstable intermediates and avoids the use of heavy metal catalysts or corrosive Lewis acids entirely. The result is a simplified workflow where the final product can be isolated through straightforward crystallization or slurry techniques, significantly reducing solvent consumption and processing time while ensuring the structural integrity of the final flavonoid scaffold.
Mechanistic Insights into IBX-Mediated Oxidative Demethylation
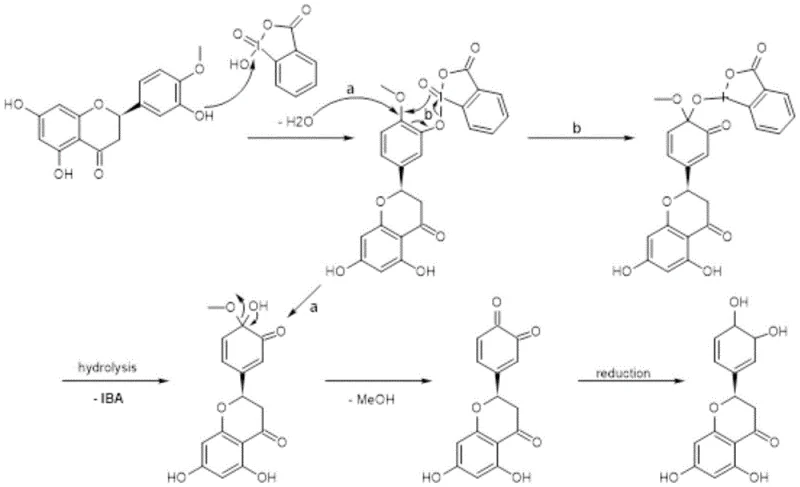
The core chemical innovation lies in the precise mechanistic pathway facilitated by the hypervalent iodine reagent, 2-iodoxybenzoic acid. Unlike harsh acidic conditions that might promote non-selective degradation or racemization, IBX acts as a specific oxidant that targets the electron-rich aromatic system of the Hesperetin B-ring. The reaction likely proceeds through the formation of an o-quinone methide or similar oxidized intermediate species, which activates the methoxy group for subsequent nucleophilic attack or hydrolysis upon reduction. This selectivity is crucial for maintaining the fidelity of the A-ring resorcinol moiety and the chiral C-2 position, ensuring that the biological activity associated with the specific stereochemistry is preserved throughout the synthesis. The mildness of the oxidation step allows the reaction to proceed at room temperature, minimizing thermal stress on the substrate and preventing the formation of polymeric byproducts often seen in high-temperature demethylation protocols.
Following the oxidation phase, the addition of sodium dithionite serves a dual purpose: it reduces the oxidized intermediate back to the phenolic state while simultaneously cleaving the methyl ether functionality to reveal the catechol group characteristic of Eriodictyol. The mechanism involves the transfer of electrons from the sulfur species to the iodine-bound intermediate, facilitating the release of the methyl group as a soluble byproduct and regenerating the aromatic system. This reductive workup is performed in an aqueous-organic biphasic system that naturally separates the inorganic iodine byproducts from the organic product, thereby simplifying the purification landscape. The entire sequence demonstrates a sophisticated understanding of redox chemistry applied to natural product modification, offering a clean alternative to stoichiometric metal salt reagents.
How to Synthesize Eriodictyol Efficiently
Implementing this synthesis requires careful control of stoichiometry and temperature to maximize yield and purity. The patent outlines a standardized procedure where Hesperetin is first dissolved in tetrahydrofuran under an inert atmosphere to prevent premature oxidation by atmospheric oxygen. The oxidant is added in a specific molar excess to drive the reaction to completion over a 24-hour period, after which the mixture is cooled to control the exotherm of the subsequent reduction. The reducing agent is prepared as an aqueous solution and added portion-wise to manage gas evolution and heat, followed by a warming period to ensure full conversion. While the specific operational parameters are detailed below, it is critical to note that the simplicity of the workup—involving solvent removal, extraction, and slurry purification—makes this route particularly amenable to technology transfer.
- Dissolve Hesperetin in THF and react with 2-iodoxybenzoic acid (IBX) under nitrogen protection at room temperature for 24 hours to form the intermediate solution.
- Cool the reaction mixture in an ice bath and add an aqueous solution of sodium dithionite in portions to reduce the intermediate.
- Warm to room temperature, remove solvent, extract with ethyl acetate, wash, dry, and purify via slurry in methanol/ethyl acetate to obtain pure Eriodictyol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of palladium catalysts removes the need for expensive metal scavenging steps and rigorous testing for residual heavy metals, which are critical quality attributes for pharmaceutical intermediates. Furthermore, replacing anhydrous aluminum chloride with stable, solid reagents like IBX and sodium dithionite significantly enhances workplace safety and reduces the regulatory burden associated with handling corrosive substances. The one-pot nature of the reaction consolidates multiple unit operations into a single vessel, thereby decreasing equipment occupancy time and lowering utility costs related to heating and cooling cycles. These efficiencies translate directly into a more resilient supply chain capable of meeting fluctuating market demands without the bottlenecks typical of multi-step, hazard-prone syntheses.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the dependency on precious metal catalysts and avoiding the complex waste treatment required for aluminum salts. By utilizing commercially available and inexpensive reagents, the raw material cost profile is optimized, and the simplified workup reduces labor and solvent expenses significantly.
- Enhanced Supply Chain Reliability: Sourcing stable, non-hazardous reagents ensures consistent production schedules free from the delays often caused by the strict transportation regulations governing corrosive acids or pyrophoric materials. The robustness of the reaction conditions allows for flexible manufacturing windows, ensuring that delivery timelines for high-purity pharmaceutical intermediates are met reliably.
- Scalability and Environmental Compliance: The absence of heavy metal waste and corrosive effluents aligns the process with stringent environmental regulations, facilitating easier permitting for scale-up. The straightforward isolation procedure minimizes solvent usage and waste generation, supporting green chemistry initiatives and reducing the overall environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability and purity profiles of this synthesis method. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the advantages of the IBX/Sodium Dithionite method over traditional AlCl3 demethylation?
A: The novel method avoids the use of highly corrosive and moisture-sensitive anhydrous aluminum chloride, eliminating dangerous exothermic quenching steps and severe emulsification during extraction, thereby simplifying post-treatment and reducing hazardous waste.
Q: Does this synthesis method preserve the stereochemistry of the starting material?
A: Yes, unlike palladium-catalyzed hydrogenation methods which may require subsequent chiral resolution, this oxidative-reductive pathway proceeds under mild conditions that maintain the absolute configuration of the chiral center present in Hesperetin.
Q: What is the preferred solvent system for scaling up this reaction?
A: Tetrahydrofuran (THF) is identified as the optimal solvent due to its ability to dissolve reactants effectively and its low boiling point, which facilitates easy removal via rotary evaporation during the workup phase.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eriodictyol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the one-pot oxidative demethylation route for Eriodictyol and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Eriodictyol meets the highest standards required for downstream drug formulation and clinical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and value in your pharmaceutical intermediate sourcing strategy.
