Advanced Pyrazole Phosphine Ligands for Efficient Pharmaceutical Intermediate Manufacturing
Advanced Pyrazole Phosphine Ligands for Efficient Pharmaceutical Intermediate Manufacturing
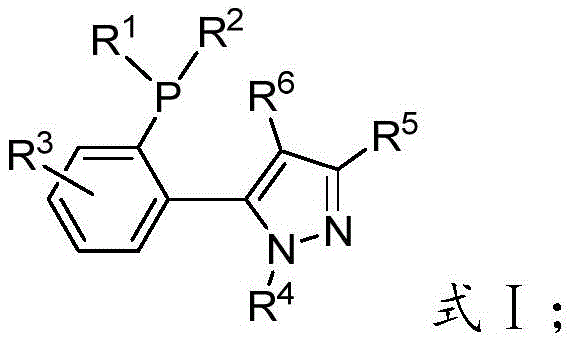
The landscape of transition metal-catalyzed cross-coupling reactions is continuously evolving, driven by the demand for more efficient, sustainable, and cost-effective methodologies in the synthesis of complex organic molecules. A significant breakthrough in this domain is detailed in patent CN114907404A, which introduces a novel class of 5-(2-(disubstituted phosphino)phenyl)-1-alkyl-1H-pyrazole phosphine ligands. These ligands represent a strategic advancement for R&D directors and process chemists seeking robust catalytic systems capable of handling challenging substrates under mild conditions. The core innovation lies in the unique pyrazole-phosphine hybrid skeleton, which offers exceptional tunability through variations in the substituents on the phosphorus atom and the pyrazole ring. This structural flexibility allows for precise modulation of steric and electronic properties, optimizing the ligand for specific catalytic transformations. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such advanced ligand systems is crucial for developing scalable processes that meet stringent purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the formation of carbon-carbon bonds via cross-coupling reactions such as Suzuki, Hiyama, Negishi, Kumada, and Stille couplings has relied heavily on the availability of specific electrophilic partners. Alkenyl halides have long served as common substrates for constructing olefinic compounds found in valuable natural products and drugs. However, the synthesis of polysubstituted alkenyl halides often necessitates harsh reaction conditions and multi-step sequences, limiting their widespread availability and increasing the overall cost of manufacturing. Furthermore, conventional phosphine ligands, while effective in many scenarios, often struggle to activate less reactive electrophiles like enol esters without requiring excessive catalyst loading or elevated temperatures. This limitation poses significant challenges for supply chain heads aiming to streamline production workflows and reduce lead times for high-purity pharmaceutical intermediates. The inability of standard ligand systems to efficiently couple enol carboxylates, particularly those with low activity, has remained a persistent bottleneck in the industry.
The Novel Approach
The novel approach presented in the patent addresses these deficiencies by leveraging enol ester electrophiles, such as enol pivalates, which are readily synthesized from corresponding carbonyl compounds and offer a diverse range of substituents. The key to unlocking the reactivity of these challenging substrates lies in the newly developed 5-(2-(disubstituted phosphino)phenyl)-1-alkyl-1H-pyrazole ligands. Unlike traditional ligands, this new class forms structurally stable complexes with transition metals like palladium, significantly enhancing catalytic activity even under mild reaction conditions. The ability to operate at room temperature or with minimal heating not only improves energy efficiency but also preserves sensitive functional groups such as esters, ketones, and methoxy groups that might otherwise degrade. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, this translates to a more robust process with fewer side reactions and higher selectivity, ultimately leading to substantial cost savings through improved yield and reduced waste generation.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling with Pyrazole Ligands
The superior performance of these ligands can be attributed to their unique ability to stabilize the active palladium species throughout the catalytic cycle. The pyrazole moiety, combined with the bulky phosphine group, creates a specific coordination environment that facilitates oxidative addition and reductive elimination steps critical for cross-coupling. By adjusting the substituents R1 through R6, chemists can fine-tune the cone angle and electron density at the metal center, optimizing the catalyst for specific substrate combinations. This level of control is essential for R&D teams working on complex molecule synthesis where minor impurities can derail an entire batch. The ligands demonstrate remarkable stability against air and moisture, a property that simplifies handling and storage requirements compared to highly sensitive phosphines. This stability ensures consistent performance across different batches, providing the reliability needed for commercial scale-up of complex pharmaceutical intermediates.
Furthermore, the mechanism allows for the activation of notoriously difficult enol pivalate substrates in Kumada and Negishi coupling reactions. The catalytic system effectively lowers the activation energy barrier, enabling reactions to proceed with palladium loadings as low as 0.5 mol%. This efficiency is pivotal for minimizing residual heavy metal content in the final API, a critical quality attribute for regulatory compliance. The compatibility with various functional groups means that protecting group strategies can often be simplified, shortening the overall synthetic route. For supply chain stakeholders, this mechanistic robustness implies a lower risk of batch failure and a more predictable production schedule, ensuring continuous supply of critical materials.
How to Synthesize 5-(2-(Disubstituted phosphino)phenyl)-1-alkyl-1H-pyrazole Efficiently
The synthesis of these high-performance ligands follows a logical and scalable two-step sequence that begins with commercially available starting materials. The process involves the initial formation of a bromo-intermediate followed by phosphination, utilizing standard organolithium chemistry that is well-understood in industrial settings. The simplicity of the route, combined with the use of accessible reagents like 1-alkylpyrazoles and 1,2-dibromobenzene, makes it an attractive candidate for large-scale production. Detailed operational parameters regarding temperature control and stoichiometry are critical to maximizing yield and purity. The following guide outlines the standardized synthetic steps derived from the patent data, providing a clear roadmap for process implementation.
- React 1-alkylpyrazole with n-butyllithium or tert-butyllithium in THF at low temperature, followed by addition of 1,2-dibromobenzene to form the 5-(2-bromophenyl)-1-alkyl-1H-pyrazole intermediate.
- Treat the bromo-intermediate with n-butyllithium or tert-butyllithium at -78°C to generate the aryl lithium species.
- Quench the lithiated species with disubstituted chlorophosphine (e.g., dicyclohexylphosphine chloride) to yield the final phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel ligand technology offers distinct commercial advantages that extend beyond mere technical performance, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. For procurement managers, the primary benefit lies in the drastic simplification of the supply chain for catalytic materials. Since the ligands are air-stable and easy to synthesize from common precursors, the reliance on exotic or highly specialized reagents is minimized. This accessibility ensures a steady supply of catalysts, reducing the risk of production delays caused by material shortages. Moreover, the ability to achieve high conversion rates with minimal catalyst loading means that the consumption of expensive palladium salts is significantly reduced. This reduction in precious metal usage directly correlates to lower raw material costs per kilogram of product, enhancing the overall economic viability of the manufacturing process without compromising on quality.
- Cost Reduction in Manufacturing: The implementation of these ligands facilitates a significant decrease in operational expenses primarily through the reduction of catalyst loading. By enabling effective catalysis at levels as low as 0.5 mol%, the process minimizes the expenditure on costly palladium sources. Additionally, the mild reaction conditions often eliminate the need for energy-intensive heating or cooling protocols, further lowering utility costs. The high selectivity of the system reduces the formation of by-products, which simplifies downstream purification processes and decreases solvent consumption. These cumulative effects result in a leaner, more cost-efficient manufacturing workflow that maximizes resource utilization.
- Enhanced Supply Chain Reliability: The robustness of the ligand synthesis and its stability during storage contribute to a more resilient supply chain. Unlike sensitive catalysts that require strict inert atmosphere handling and cold chain logistics, these pyrazole phosphine ligands can be managed with standard safety protocols. This ease of handling reduces logistical complexities and potential points of failure in the supply network. Furthermore, the use of readily available starting materials for ligand production ensures that upstream supply disruptions are less likely to impact final product delivery. This reliability is crucial for maintaining consistent inventory levels and meeting tight delivery schedules demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with green chemistry principles. The high atom economy and reduced waste generation associated with high-yield couplings minimize the environmental footprint of the operation. The ability to run reactions at room temperature or moderate heat reduces the energy burden on the facility. Scalability is enhanced by the straightforward workup procedures, which typically involve standard extraction and chromatography techniques familiar to plant operators. This ease of scale-up ensures that transitions from pilot plant to commercial production are smooth, allowing for rapid response to market demands while adhering to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the suitability of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent. These answers address common concerns regarding ligand stability, substrate scope, and operational parameters. Understanding these technical specifics is vital for integrating new catalytic systems into existing workflows and ensuring successful process validation. The following insights are derived directly from the documented performance of the ligands in various cross-coupling scenarios.
Q: What are the stability characteristics of these pyrazole phosphine ligands?
A: According to patent CN114907404A, these ligands exhibit significant stability towards both air and moisture, making them easier to store and handle compared to many traditional phosphine ligands.
Q: Can these ligands be used for challenging enol ester substrates?
A: Yes, the ligands are specifically designed to activate difficult substrates like enol pivalates in Kumada and Negishi cross-coupling reactions, achieving high separation yields up to 95%.
Q: What is the typical palladium loading required for these reactions?
A: The catalytic system allows for significantly reduced palladium usage, with effective catalytic amounts as low as 0.5 mol%, which helps reduce heavy metal contamination in the final product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole Phosphine Ligand Supplier
As the demand for advanced catalytic solutions grows, partnering with an experienced CDMO becomes essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM stands ready to support your development efforts with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical-grade intermediates. We understand the critical nature of catalyst performance in your synthesis and are committed to delivering high-quality ligands that ensure consistent batch-to-batch reproducibility. Our team of experts is prepared to collaborate closely with your R&D department to optimize reaction conditions and troubleshoot any process challenges that may arise during scale-up.
We invite you to explore the potential of these novel pyrazole phosphine ligands for your next project. Whether you are looking to improve the efficiency of an existing route or develop a new process for a challenging target, our technical procurement team is available to provide a Customized Cost-Saving Analysis. Please contact us to request specific COA data and route feasibility assessments tailored to your unique requirements. Let us help you achieve your production goals with greater efficiency and reliability.
