Advanced Synthesis of Lorcaserin Intermediate via Safe Reduction and Condensation
Introduction to Patent CN106631823B and Technological Breakthroughs
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency, safety, and environmental sustainability. Patent CN106631823B introduces a transformative preparation method for Lorcaserin Intermediate I, a critical precursor in the synthesis of appetite suppressants targeting the 5-HT2C receptor. This innovation addresses long-standing challenges in the manufacturing of anti-obesity drug intermediates by replacing hazardous reagents like thionyl chloride and borane with a safer sodium borohydride and boron trifluoride diethyl etherate system. By shifting the starting material strategy to inexpensive p-chlorobenzonitrile, the process significantly lowers the barrier to entry for large-scale production while maintaining high purity standards essential for regulatory compliance.
For R&D directors and process chemists, the significance of this patent lies in its elegant simplification of the reaction sequence. Traditional routes often suffer from complex workups involving column chromatography or the generation of toxic metal waste. In contrast, this novel approach utilizes standard organic solvents like tetrahydrofuran and acetonitrile, facilitating easier solvent recovery and recycling. The method achieves high yields through controlled temperature gradients, specifically maintaining reduction temperatures between 0-5°C during reagent addition and 25-55°C during reaction progression. This precise thermal control minimizes side reactions, ensuring a cleaner impurity profile that simplifies downstream purification and enhances the overall economic viability of the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing chlorophenylethylamine derivatives have been plagued by significant operational and environmental drawbacks. As illustrated in earlier patent documents such as CN1037090434A and CN103965057, conventional routes frequently rely on metal chlorides like aluminum trichloride or ferric chloride. These reagents not only generate vast quantities of wastewater containing heavy metal ions but also complicate the recycling process, posing severe environmental compliance risks for modern chemical plants. Furthermore, alternative pathways reported in patents like WO2009111004A1 utilize expensive starting materials such as p-chlorophenethylamine directly or require costly condensing agents like 2,5,6-trifluorophenylboronic acid.
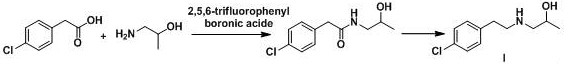
The reliance on hazardous reagents extends beyond metals; many established protocols employ thionyl chloride or hydrobromic acid for halogenation steps. These substances are highly corrosive and produce toxic gases, necessitating specialized scrubbing equipment and rigorous safety protocols that inflate capital expenditure. Additionally, the use of Raney nickel in hydrogenation steps introduces flammability risks and requires strict storage conditions. Perhaps most critically for procurement teams, several legacy methods yield products that can only be purified via column chromatography. This technique is notoriously difficult to scale industrially, leading to low throughput, high solvent consumption, and inconsistent batch-to-batch quality, making these routes unsuitable for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN106631823B represents a paradigm shift towards greener and more cost-effective manufacturing. By initiating the synthesis from p-chlorobenzonitrile, a commodity chemical, the process bypasses the need for expensive pre-functionalized amines. The core innovation involves a mild reduction step using sodium borohydride activated by boron trifluoride diethyl etherate. This combination effectively reduces the nitrile group to a primary amine without the extreme hazards associated with borane gas or high-pressure hydrogenation. The subsequent condensation with 1-chloro-2-propanol proceeds smoothly in acetonitrile under alkaline conditions, avoiding the use of aggressive halogenating agents entirely.
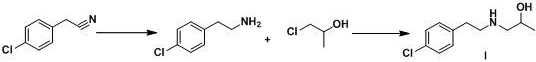
This novel approach drastically simplifies the purification workflow. Instead of labor-intensive chromatography, the final product is isolated through straightforward recrystallization using ethyl acetate and n-heptane mixtures. This change alone translates to substantial reductions in processing time and solvent waste. The reaction conditions are optimized for scalability, with molar ratios carefully tuned to 1:1.5-2.0 for the reduction components and 1:1:1.5-2.0 for the condensation step. Such precision ensures high conversion rates while minimizing excess reagent costs. For supply chain managers, this route offers a reliable lorcaserin intermediate supplier pathway that mitigates the risks associated with volatile raw material markets and stringent environmental regulations.
Mechanistic Insights into NaBH4/BF3-Catalyzed Reduction and Condensation
The chemical elegance of this process centers on the activation of sodium borohydride by Lewis acids. In the first stage, boron trifluoride diethyl etherate coordinates with the hydride source to generate a more potent reducing species capable of attacking the nitrile triple bond. This reaction is highly exothermic, necessitating the strict temperature control observed in the patent, where reagents are added dropwise at 0-5°C to prevent runaway reactions. Once the addition is complete, the mixture is warmed to 25-55°C to drive the reduction to completion, converting p-chlorobenzonitrile into p-chlorophenylethylamine with exceptional selectivity. The mechanism avoids the formation of secondary amine byproducts often seen in less controlled reductions, thereby preserving the integrity of the primary amine functionality required for the next step.
Following the reduction, the nucleophilic substitution mechanism dominates the second stage. The freshly synthesized p-chlorophenylethylamine acts as a nucleophile, attacking the electrophilic carbon of 1-chloro-2-propanol. The presence of a base, such as potassium carbonate or triethylamine, serves to neutralize the hydrochloric acid byproduct, driving the equilibrium forward. The choice of acetonitrile as a solvent is critical here, as it provides excellent solubility for both organic reactants and inorganic bases while remaining stable at the reaction temperature of 55-60°C. This mechanistic understanding allows for precise impurity control; by maintaining the pH and temperature within the specified narrow windows, the formation of dialkylated byproducts or elimination products is suppressed, ensuring the high-purity lorcaserin intermediates required for downstream API synthesis.
How to Synthesize Lorcaserin Intermediate I Efficiently
Implementing this synthesis route requires careful attention to thermal management and stoichiometry to maximize yield and safety. The process is divided into two distinct operational phases: the reduction of the nitrile and the subsequent alkylation. Operators must ensure that the addition of boron trifluoride diethyl etherate is strictly controlled to manage gas evolution and heat generation. Detailed standardized operating procedures regarding quenching, pH adjustment, and solvent swapping are essential for reproducible results. The following guide outlines the critical parameters derived from the patent examples to assist technical teams in replicating this efficient protocol.
- Reduce p-chlorobenzonitrile using sodium borohydride and boron trifluoride diethyl etherate in THF at 0-55°C to obtain p-chlorophenylethylamine.
- Condense the resulting amine with 1-chloro-2-propanol in acetonitrile under alkaline conditions at 55-60°C, followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages beyond mere technical feasibility. The shift away from specialized, high-cost reagents to commodity chemicals fundamentally alters the cost structure of the intermediate. By eliminating the dependency on imported or scarce materials like specific boronic acids or pre-made amines, manufacturers can insulate themselves from supply shocks and price volatility. Furthermore, the simplification of the purification process from chromatography to crystallization dramatically increases throughput capacity, allowing facilities to meet large-volume demands without proportional increases in labor or equipment costs.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by raw material selection and waste minimization. Utilizing p-chlorobenzonitrile instead of expensive p-chlorophenylethylamine significantly lowers the input cost per kilogram of product. Additionally, the avoidance of metal chlorides eliminates the expensive wastewater treatment processes required to remove heavy metals, leading to substantial cost savings in environmental compliance. The high yields reported in the patent examples, exceeding 80% for both steps, further enhance the overall material efficiency, reducing the cost of goods sold and improving margin potential for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Sodium borohydride, boron trifluoride complexes, and simple alkyl halides are produced globally in massive quantities, ensuring consistent availability even during market disruptions. The removal of hazardous gases like borane and corrosive liquids like thionyl chloride simplifies logistics and storage requirements, reducing the regulatory burden on transportation and warehousing. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and more stable production schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is inherently safer and more scalable. The absence of pyrophoric catalysts like Raney nickel and toxic gases removes major safety barriers to scaling up reactor sizes. The process generates minimal hazardous waste, aligning with green chemistry principles and facilitating easier permitting for new production lines. The ability to recycle solvents like tetrahydrofuran and acetonitrile further reduces the environmental footprint. This alignment with sustainability goals not only future-proofs the manufacturing asset but also appeals to increasingly eco-conscious stakeholders in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in CN106631823B, offering clarity on safety, purity, and operational feasibility. Understanding these nuances is vital for technical teams evaluating the transition from legacy processes to this optimized workflow.
Q: Why is the NaBH4/BF3 reduction system preferred over traditional metal chlorides?
A: Traditional methods using aluminum or ferric chlorides generate significant metal-containing wastewater that is difficult to treat. The NaBH4/BF3 system avoids heavy metal contamination, simplifying waste management and enhancing environmental compliance.
Q: How does this method reduce production costs compared to prior art?
A: This route utilizes cheap, readily available starting materials like p-chlorobenzonitrile instead of expensive p-chlorophenylethylamine or specialized boronic acids. Additionally, it eliminates the need for costly column chromatography by using simple recrystallization.
Q: What are the safety advantages of avoiding thionyl chloride and borane?
A: Thionyl chloride and borane are highly toxic, corrosive, and potentially explosive. Replacing them with safer reagents reduces occupational health risks, lowers storage hazards, and ensures a more stable supply chain for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lorcaserin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust intermediate supply chains in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Lorcaserin Intermediate I meets the highest international standards. Our commitment to safety and environmental stewardship mirrors the values embedded in this advanced synthetic route, making us an ideal partner for long-term collaboration.
We invite pharmaceutical companies and contract research organizations to leverage our expertise for their anti-obesity drug programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain with high-quality intermediates that drive efficiency and reliability in your drug development pipeline.
