Advanced Electro-Oxidative Cyclization for Scalable Dibenzosuberone Pharmaceutical Intermediates Production
The construction of the 6-7-6 polycyclic skeleton represents one of the most formidable challenges in modern organic synthesis, particularly due to the entropic penalties associated with forming seven-membered rings. This structural motif is the pharmacophore core of colchicine and its potent analogues, such as NSC 51046 and ZD 6126, which exhibit profound anti-tumor activity by inhibiting tubulin polymerization. 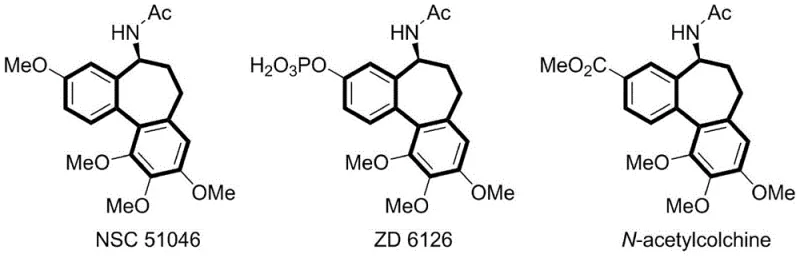 . Traditionally, accessing these complex architectures has required multi-step sequences involving harsh conditions or expensive noble metal catalysts. However, recent advancements in electro-organic synthesis have opened new avenues for greener manufacturing. Specifically, Patent CN113957463A discloses a groundbreaking method for synthesizing dibenzosuberone derivatives via radical serial cyclization under electro-oxidation conditions. This technology leverages the precise control of electron transfer to generate reactive radical species in situ, offering a sustainable alternative for producing high-purity pharmaceutical intermediates essential for next-generation oncology therapeutics.
. Traditionally, accessing these complex architectures has required multi-step sequences involving harsh conditions or expensive noble metal catalysts. However, recent advancements in electro-organic synthesis have opened new avenues for greener manufacturing. Specifically, Patent CN113957463A discloses a groundbreaking method for synthesizing dibenzosuberone derivatives via radical serial cyclization under electro-oxidation conditions. This technology leverages the precise control of electron transfer to generate reactive radical species in situ, offering a sustainable alternative for producing high-purity pharmaceutical intermediates essential for next-generation oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dibenzosuberone frameworks has relied heavily on transition-metal-catalyzed cross-coupling reactions or intramolecular Friedel-Crafts acylations. These conventional pathways often necessitate the use of stoichiometric amounts of Lewis acids or precious metal catalysts like palladium and rhodium, which not only drive up raw material costs but also introduce significant downstream processing burdens. The removal of trace heavy metals to meet stringent regulatory limits for Active Pharmaceutical Ingredients (APIs) requires specialized scavenging resins and extensive purification protocols, drastically increasing the overall production timeline and environmental footprint. Furthermore, thermal cyclization methods frequently suffer from poor regioselectivity and the formation of polymeric byproducts, leading to inconsistent yields that complicate supply chain planning for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the electro-oxidative strategy outlined in the patent utilizes electricity as a traceless reagent to drive the formation of the seven-membered ring. By employing an o-propargyl propionyl biphenyl compound and a sodium benzenesulfinate salt as starting materials, the reaction proceeds through a radical mechanism that avoids the need for external chemical oxidants. 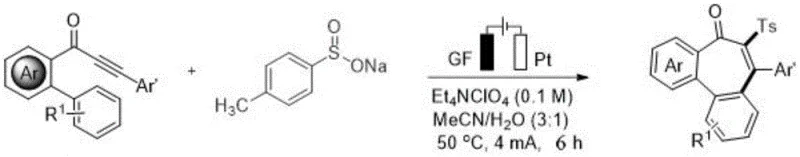 . The process operates under mild constant current conditions in a mixed solvent system of acetonitrile and water, significantly reducing the reliance on volatile organic compounds. This approach not only simplifies the reaction setup by eliminating sensitive catalyst handling but also enhances the atom economy of the transformation, providing a robust platform for cost reduction in API manufacturing where efficiency and purity are paramount.
. The process operates under mild constant current conditions in a mixed solvent system of acetonitrile and water, significantly reducing the reliance on volatile organic compounds. This approach not only simplifies the reaction setup by eliminating sensitive catalyst handling but also enhances the atom economy of the transformation, providing a robust platform for cost reduction in API manufacturing where efficiency and purity are paramount.
Mechanistic Insights into Electro-Oxidative Radical Cyclization
The elegance of this synthesis lies in its precise mechanistic pathway, which orchestrates the formation of multiple bonds in a single operational step. The process initiates at the anode, where benzene sulfinate anions undergo single-electron oxidation to generate highly reactive benzene sulfonyl radicals. These electrophilic radicals selectively attack the alpha-position of the carbonyl group on the alkynone substrate, forming a stabilized alkenyl radical intermediate. 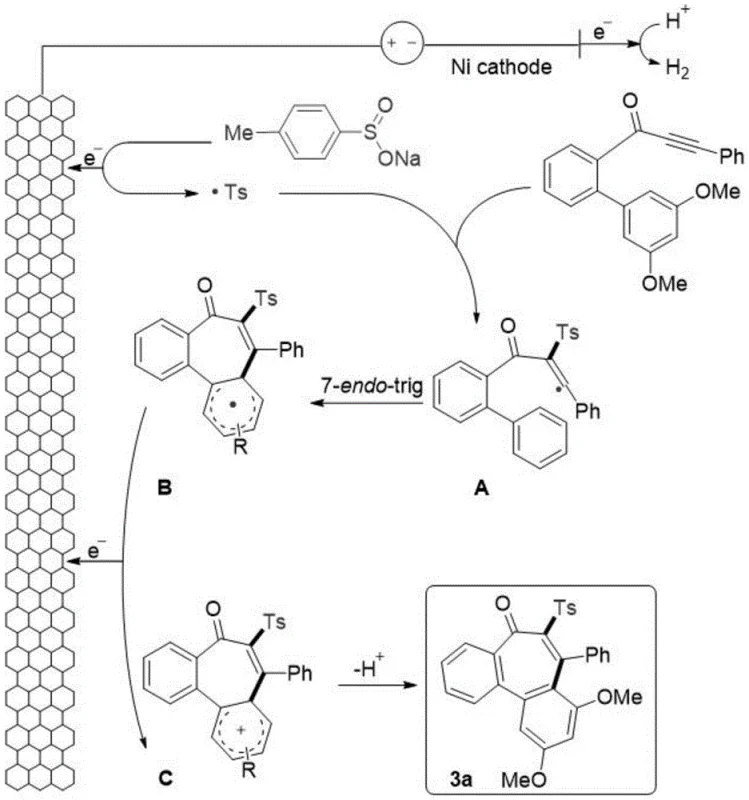 . Subsequently, this alkenyl radical undergoes a critical 7-endo-trig cyclization, adding to the pendant aromatic ring to construct the central seven-membered ring of the dibenzosuberone core. The resulting cyclohexadienyl radical is then oxidized at the anode to form a cationic species, which finally undergoes deprotonation to restore aromaticity and yield the final ketone product. This cascade sequence effectively bypasses the high energy barriers typically associated with medium-ring formation.
. Subsequently, this alkenyl radical undergoes a critical 7-endo-trig cyclization, adding to the pendant aromatic ring to construct the central seven-membered ring of the dibenzosuberone core. The resulting cyclohexadienyl radical is then oxidized at the anode to form a cationic species, which finally undergoes deprotonation to restore aromaticity and yield the final ketone product. This cascade sequence effectively bypasses the high energy barriers typically associated with medium-ring formation.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. Because the reaction is driven by electrode potential rather than bulk chemical reactivity, side reactions such as over-oxidation or non-specific nucleophilic attacks are minimized. The use of sodium p-toluenesulfinate serves as a efficient radical source that decomposes cleanly, leaving behind minimal organic residues that could complicate purification. The tolerance for various substituents on the aromatic rings, including halogens and esters, suggests that the radical intermediates are sufficiently stable to prevent premature decomposition, ensuring a cleaner impurity profile that aligns with the rigorous quality standards required for reliable pharmaceutical intermediate supplier partnerships.
How to Synthesize Dibenzosuberone Derivatives Efficiently
The practical implementation of this electro-oxidative protocol is straightforward and amenable to standard laboratory equipment, facilitating rapid process development. The method utilizes a simple undivided cell configuration with a graphite felt anode and a platinum or nickel cathode, materials that are both durable and cost-effective for industrial adaptation. The reaction conditions are optimized to balance reaction rate and selectivity, typically operating at a constant current of 4 mA and a temperature of 50°C for a duration of 6 hours. For detailed standardized synthesis steps and specific parameter adjustments for different substrates, please refer to the guide below.
- Prepare the electrolytic cell with an o-propargyl propionyl biphenyl compound and sodium benzenesulfinate in a MeCN/H2O solvent system containing Et4NClO4 electrolyte.
- Apply a constant current of 4 mA using a graphite felt anode and platinum cathode at a controlled temperature of 50°C for 6 hours.
- Purify the resulting crude mixture via silica gel column chromatography using n-hexane/EtOAc to isolate the target dibenzosuberone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift towards electro-organic synthesis represents a strategic opportunity to de-risk the supply of critical oncology intermediates. By replacing expensive noble metal catalysts with electricity and inexpensive sulfinate salts, the direct material costs associated with the synthesis are significantly reduced. Furthermore, the elimination of heavy metals removes the need for complex and costly purification steps dedicated to metal scavenging, streamlining the manufacturing workflow and reducing the overall lead time for high-purity pharmaceutical intermediates. This simplification of the process flow enhances supply chain reliability by reducing the number of unit operations and potential failure points.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with electrochemical oxidation fundamentally alters the cost structure of the synthesis. Without the need for palladium or rhodium complexes, the raw material expenditure is drastically lowered, and the associated costs of catalyst recovery or disposal are entirely eliminated. Additionally, the use of water as a co-solvent reduces the volume of organic solvents required, leading to substantial cost savings in solvent procurement and waste treatment, making the process economically superior to traditional thermal methods.
- Enhanced Supply Chain Reliability: The reagents required for this transformation, such as sodium benzenesulfinate and tetraethylammonium perchlorate, are commodity chemicals with stable global supply chains, unlike specialized ligands or catalysts that may face availability bottlenecks. The robustness of the electrochemical setup ensures consistent batch-to-batch reproducibility, which is critical for maintaining continuous production schedules. This reliability allows for better inventory planning and reduces the risk of stockouts for key API precursors.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable through the increase of electrode surface area or the use of flow chemistry reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates without the safety risks associated with large-scale exothermic oxidations. The generation of hydrogen gas at the cathode is the primary byproduct, which can be safely managed, resulting in a much greener E-factor compared to stoichiometric oxidant methods. This alignment with green chemistry principles simplifies environmental permitting and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electro-oxidative synthesis of dibenzosuberones. These insights are derived directly from the experimental data and scope defined in the patent literature, providing clarity on the feasibility and advantages of adopting this technology for your specific manufacturing needs.
Q: What are the key advantages of this electro-oxidative method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts such as palladium or copper, thereby removing the costly and complex heavy metal removal steps typically required in API manufacturing. Additionally, it utilizes electricity as a clean reagent, generating fewer hazardous waste byproducts compared to stoichiometric chemical oxidants.
Q: What is the substrate scope for the aryl group in the starting material?
A: The process demonstrates broad compatibility with various aromatic systems, including alkyl, aryl, halogen, or ester-substituted benzene rings, as well as heteroaromatic rings like furan, pyrrole, and thiophene, allowing for the synthesis of diverse dibenzosuberone analogues.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of constant current electrolysis with readily available electrodes (graphite felt and platinum/nickel) and benign solvent systems (MeCN/H2O) indicates high potential for scalability, addressing the supply chain continuity needs for complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzosuberone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electro-organic synthesis in the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this radical cyclization can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the exacting standards required for global drug development pipelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecule, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical anti-cancer intermediates.
