Advanced Catalytic Synthesis of Etocrilene UV Absorber Intermediate for Commercial Scale
Advanced Catalytic Synthesis of Etocrilene UV Absorber Intermediate for Commercial Scale
The global demand for high-performance ultraviolet (UV) absorbers continues to surge, driven by stringent regulations in the cosmetics and polymer industries regarding skin protection and material stability. At the heart of this market lies Etocrilene, chemically known as ethyl 2-cyano-3,3-diphenylacrylate and commercially referred to as Viosorb 910, which serves as a critical intermediate for the synthesis of Octocrilene, a ubiquitous UV-B filter. Patent CN103242197B introduces a groundbreaking preparation method that fundamentally redefines the manufacturing landscape for this valuable chemical entity. By leveraging a sophisticated organic amine and organic acid dual-catalyst system, this technology circumvents the historical limitations of ammonia generation and complex purification, delivering a product with exceptional purity profiles suitable for sensitive cosmetic applications. 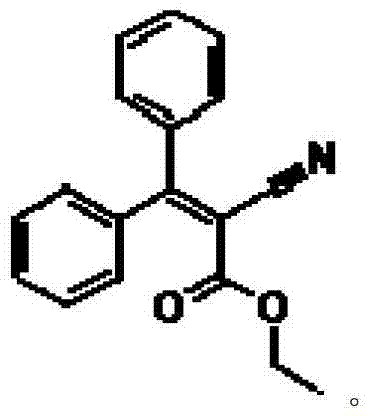
This proprietary methodology not only enhances the chemical integrity of the final intermediate but also aligns perfectly with modern green chemistry principles through extensive material recycling loops. For R&D directors and procurement specialists alike, understanding the nuances of this catalytic system is paramount for securing a reliable supply chain of high-purity UV absorber intermediates. The process utilizes readily available starting materials such as benzophenone and ethyl cyanoacetate, reacting them under controlled reflux conditions to achieve conversion rates that significantly outperform legacy amino acid-catalyzed routes. As we delve deeper into the technical specifics, it becomes evident that this patent represents a pivotal shift towards more sustainable and economically viable production strategies for fine chemical intermediates used in personal care formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Etocrilene and its downstream derivative Octocrilene has been plagued by significant technical hurdles that inflate production costs and compromise product quality. Traditional pathways, such as those disclosed in DE1954894, rely on the reaction of benzophenone imine with ethyl cyanoacetate, a route that inevitably generates ammonia gas as a stoichiometric byproduct. This ammonia evolution necessitates complex engineering controls, such as continuous nitrogen sweeping or vacuum distillation during the reaction, to prevent the formation of stubborn amide impurities that degrade the optical clarity and stability of the final UV absorber. Furthermore, alternative Knoevenagel condensation methods utilizing ammonium acetate and organic acids suffer from similar drawbacks, where the accumulation of ammonium salts creates severe separation challenges and generates substantial quantities of acidic wastewater that is difficult to treat effectively.
Moreover, prior art attempts to utilize amino acids like beta-alanine as catalysts, while avoiding ammonia gas, have resulted in unacceptably long reaction times exceeding 90 hours and mediocre yields ranging merely from 28% to 64%. These inefficiencies render such processes economically unfeasible for large-scale commercial manufacturing, as the high cost of amino acid catalysts combined with low throughput creates a prohibitive barrier to entry. The inability to efficiently recover and recycle these expensive catalysts further exacerbates the economic burden, while the resulting product often fails to meet the rigorous purity specifications required for high-end cosmetic applications, leading to potential batch rejections and supply chain disruptions for downstream formulators seeking consistent performance.
The Novel Approach
In stark contrast to these archaic methodologies, the novel approach detailed in Patent CN103242197B employs a highly efficient dual-catalyst system comprising a specific organic amine, such as dibutylamine or piperidine, paired with an organic acid like propionic acid. This synergistic catalytic combination facilitates the Knoevenagel condensation between benzophenone and ethyl cyanoacetate under mild reflux conditions without generating any gaseous ammonia byproducts. By eliminating the source of amide formation at the molecular level, the process inherently produces a cleaner crude reaction mixture, thereby drastically simplifying the downstream purification workflow and reducing the need for energy-intensive distillation steps solely dedicated to byproduct removal. The use of water-insoluble solvents, such as cyclohexane or n-heptane, enables a seamless phase separation strategy where reaction-generated water is continuously removed via azeotropic distillation, driving the equilibrium towards completion.
Furthermore, this innovative process design integrates a comprehensive material recovery system that captures and recycles unreacted starting materials, solvents, and even the catalyst components themselves. The reaction mixture is subjected to a strategic workup where the aqueous layer is distilled to recover water for reuse in washing steps, while the organic layer undergoes fractional distillation to reclaim the solvent and excess ethyl cyanoacetate for immediate reintroduction into the reactor. This closed-loop architecture not only minimizes raw material consumption but also significantly reduces the environmental footprint of the manufacturing process, addressing the growing regulatory pressure on chemical producers to adopt sustainable practices. The result is a robust, scalable synthesis route that delivers Etocrilene with a GC purity exceeding 99.0%, setting a new benchmark for quality in the UV absorber intermediate market.
Mechanistic Insights into Organic Amine-Acid Catalyzed Condensation
The core of this technological breakthrough lies in the precise mechanistic interplay between the secondary amine and the carboxylic acid within the reaction medium. Unlike primary amines or ammonia sources that can lead to unstable imine intermediates and subsequent degradation, the selected secondary amines (e.g., di-n-butylamine) form stable enamine intermediates with ethyl cyanoacetate that are highly nucleophilic yet resistant to hydrolysis under the reaction conditions. The concurrent presence of the organic acid, preferably propionic acid, serves to activate the carbonyl group of the benzophenone through hydrogen bonding, thereby lowering the activation energy for the nucleophilic attack. This dual-activation mechanism ensures a rapid and selective condensation reaction that proceeds to high conversion within 12 to 15 hours, a timeframe that is remarkably short compared to the multi-day durations required by amino acid-catalyzed alternatives.
Crucially, the absence of free ammonia in the reaction vessel prevents the nucleophilic attack on the ester moiety of the product or the starting material, which is the primary pathway for the formation of amide impurities in conventional processes. This mechanistic advantage translates directly into superior impurity control, as evidenced by the patent data showing single impurity levels below 0.1% and total impurities under 0.5%. 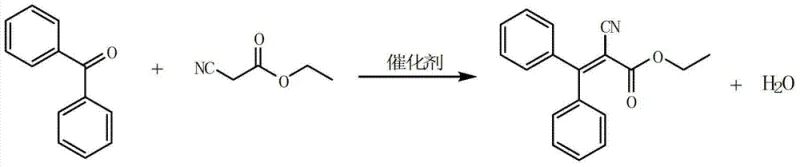
The purification strategy further leverages the solubility differences of the product in ethanol-water mixtures. By adding 75-100% ethanol to the concentrated residue and cooling the mixture to temperatures between 0°C and 20°C, the Etocrilene crystallizes out as high-quality off-white crystals, leaving residual impurities and trace catalysts in the mother liquor. This crystallization step is not merely a isolation technique but a critical refining stage that ensures the final product meets the stringent color and purity specifications demanded by the cosmetics industry, where even trace discoloration can render a batch unusable for premium sunscreen formulations.
How to Synthesize Etocrilene Efficiently
The synthesis of Etocrilene via this patented route involves a sequence of carefully orchestrated unit operations designed to maximize yield and purity while minimizing waste. The process begins with the charging of benzophenone, ethyl cyanoacetate, the catalyst system, and a water-immiscible solvent into a condensation reactor equipped with a water separator. The mixture is heated to reflux, typically between 85°C and 88°C when using cyclohexane, allowing for the continuous removal of water generated during the condensation. Once the water evolution ceases, indicating reaction completion, the mixture is cooled and subjected to a liquid-liquid extraction workup to separate the catalyst into the aqueous phase, followed by a series of distillation steps to recover solvents and unreacted materials for recycling. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and recycling protocols, are outlined in the guide below.
- Charge benzophenone, ethyl cyanoacetate, a specific organic amine-acid catalyst pair, and a water-insoluble solvent into a reactor, then reflux with water separation for 12-15 hours.
- Cool the reaction mixture, wash with water to separate layers, and distill both the aqueous and organic layers to recover water, solvent, and unreacted starting materials for recycling.
- Subject the distillation residue to vacuum distillation, followed by crystallization using 75-100% ethanol at low temperatures to isolate high-purity Etocrilene crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers profound strategic advantages that extend far beyond simple yield improvements. The elimination of expensive and difficult-to-recycle amino acid catalysts, replaced by commodity chemicals like dibutylamine and propionic acid, results in a drastic reduction in raw material costs. Furthermore, the ability to recycle the solvent and unreacted benzophenone creates a circular economy within the production facility, insulating the manufacturing process from volatility in raw material pricing and ensuring a consistent cost structure that allows for competitive pricing in the global market. This economic resilience is critical for maintaining margins in the highly competitive fine chemical sector.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by enabling the near-quantitative recovery and reuse of key inputs. Unlike traditional methods where catalysts are consumed or lost in waste streams, this system allows for the distillation and recovery of the organic amine-acid catalyst pair from the aqueous wash layer, which can then be replenished with minor fresh additions and returned to the reactor. Additionally, the recovery of excess ethyl cyanoacetate and benzophenone from the mother liquor and distillation residues means that the effective consumption of these raw materials approaches stoichiometric limits, virtually eliminating waste-related costs and maximizing the atom economy of the entire synthesis campaign.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade starting materials such as benzophenone and ethyl cyanoacetate ensures a robust and resilient supply chain that is not dependent on niche or specialized reagents. The simplicity of the operation, which avoids complex pressure controls or hazardous gas handling associated with ammonia generation, reduces the risk of unplanned downtime due to equipment failure or safety incidents. This operational stability guarantees consistent delivery schedules for downstream customers, mitigating the risk of production stoppages in their own sunscreen or polymer additive manufacturing lines due to intermediate shortages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is exceptionally well-suited for expansion from pilot to multi-ton commercial scale. The generation of wastewater is minimized to less than 0.3 tons per ton of product through the recycling of reaction water, significantly lowering effluent treatment costs and easing regulatory compliance burdens. The use of standard distillation and crystallization equipment means that the technology can be scaled up using existing infrastructure without the need for specialized reactors, facilitating rapid capacity expansion to meet surging market demand for high-performance UV absorbers in the Asia-Pacific and North American regions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for procurement teams assessing the quality credentials of potential suppliers.
Q: What represents the primary advantage of the organic amine-acid catalyst system over traditional ammonium acetate methods?
A: The primary advantage is the elimination of ammonia gas byproducts, which prevents the formation of difficult-to-remove amide impurities and simplifies the purification process significantly.
Q: How does the patented process address environmental concerns regarding wastewater generation?
A: The process utilizes a closed-loop water recycling system where reaction water is separated, distilled, and reused, reducing wastewater generation to less than 0.3 tons per ton of product.
Q: What level of purity can be achieved using the ethanol crystallization step described in the patent?
A: The crystallization step yields off-white crystals with a GC purity of greater than 99.0%, with single impurities controlled below 0.1% and total impurities below 0.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etocrilene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates like Etocrilene play in the formulation of next-generation sun protection and polymer stabilization products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay, guaranteeing that every batch performs consistently in your final applications.
We invite you to collaborate with us to leverage this advanced catalytic technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and high-quality supply of essential UV absorber intermediates.
