Advanced Synthesis of 2-Amino-4,6-Lutidine: A Breakthrough in Pharmaceutical Intermediate Manufacturing
Introduction to Patent CN111303047B
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for critical heterocyclic building blocks, and the recent disclosure in patent CN111303047B represents a significant leap forward in the synthesis of 2-amino-4,6-lutidine. This vital intermediate serves as a cornerstone for the development of neonicotinoid insecticides such as imidacloprid and acetamiprid, as well as various pyridine-based pharmaceutical derivatives. The patented methodology addresses long-standing challenges in the field by replacing hazardous, high-pressure protocols with a streamlined, two-step sequence that leverages inexpensive commodity chemicals. By utilizing 3-aminocrotonenitrile as the primary starting material, the process achieves a remarkable total yield exceeding 70% and a gas chromatography (GC) purity of greater than 99%, setting a new benchmark for quality and efficiency in fine chemical manufacturing.
For R&D directors and procurement strategists, the implications of this technology are profound, offering a reliable alternative to legacy routes that often suffer from poor selectivity and dangerous operating conditions. The innovation lies not just in the chemical transformation itself, but in the holistic optimization of the workflow, from the initial cyclization in acetic acid to the final recrystallization with isopropyl ether. This report delves deep into the mechanistic underpinnings and commercial viability of this novel approach, providing a comprehensive analysis for stakeholders looking to secure a stable supply of high-purity 2-amino-4,6-lutidine. As market demand for these downstream applications continues to rise year by year, adopting such an efficient synthetic route becomes a strategic imperative for maintaining competitiveness in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-amino-4,6-lutidine has been plagued by significant technical and safety hurdles that hinder large-scale adoption and cost efficiency. Traditional synthetic routes typically rely on the functionalization of 2,6-lutidine, a process that necessitates aggressive oxidation using hydrogen peroxide followed by nitration with mixed acids and subsequent reduction using palladium on carbon (Pd/C) catalysts under hydrogen pressure. These multi-step sequences are inherently risky due to the handling of explosive intermediates and the requirement for high-pressure hydrogenation equipment, which drastically increases capital expenditure and operational complexity. Alternatively, another reported method involves the reaction of dehydroacetic acid with ammonia water under high pressure to generate 2,6-dimethyl-4-hydroxypyridine, followed by further amination. This pathway is equally problematic, characterized by poor reaction specificity, uncontrollable side reactions, and low conversion rates that result in difficult purification scenarios and substantial waste generation. The harsh experimental conditions associated with these legacy methods pose severe safety risks and make consistent quality control nearly impossible, rendering them unsuitable for modern, green industrial production standards.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in CN111303047B introduces a remarkably elegant and efficient strategy centered around the cyclization of 3-aminocrotonenitrile. This method bypasses the need for high-pressure hydrogenation and hazardous nitration entirely, instead utilizing a controlled thermal cyclization in acetic acid followed by acid-catalyzed hydrolysis. The reaction begins with the batch addition of 3-aminocrotonenitrile into acetic acid at moderate temperatures, initiating a ring-closing reaction that forms the key intermediate, 6-amino-2,4-dimethyl-3-pyridine nitrile. This intermediate is then subjected to hydrolysis in concentrated sulfuric acid, effectively converting the nitrile group and finalizing the pyridine structure without the formation of complex byproduct mixtures. The simplicity of this route allows for precise control over reaction parameters, minimizing side reactions and maximizing the yield of the desired product. Furthermore, the purification protocol is straightforward, involving standard unit operations like filtration, extraction, and distillation, which significantly lowers the barrier for industrial scale-up and ensures a consistent supply of high-quality material.
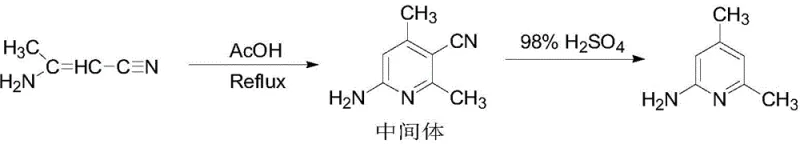
Mechanistic Insights into Acetic Acid-Mediated Cyclization and Hydrolysis
The core of this synthetic breakthrough lies in the precise manipulation of reaction conditions to drive the cyclization of 3-aminocrotonenitrile into the pyridine ring system with high fidelity. In the first stage, the reaction is conducted in glacial acetic acid, which serves both as a solvent and a mild acid catalyst to facilitate the nucleophilic attack and subsequent ring closure. The process requires careful temperature management, heating the mixture to 120-130°C under reflux to ensure complete conversion while preventing thermal degradation of the sensitive amino-nitrile species. The batch addition of the starting material is critical here; by adding 3-aminocrotonenitrile in four portions over a period of time, the local concentration of reactants is kept in check, which suppresses polymerization and other oligomerization side reactions that could otherwise compromise the purity of the intermediate. This controlled environment fosters the formation of 6-amino-2,4-dimethyl-3-pyridine nitrile with exceptional selectivity, laying a clean foundation for the subsequent transformation.
Following the isolation of the intermediate, the second critical phase involves hydrolysis and structural refinement using 98% concentrated sulfuric acid at elevated temperatures ranging from 160-180°C. This step is designed to hydrolyze the nitrile functionality and stabilize the aromatic pyridine core, a transformation that demands robust acidic conditions to proceed to completion. The use of concentrated sulfuric acid not only drives the hydrolysis equilibrium forward but also helps in solubilizing the intermediate, ensuring homogeneous reaction conditions that are vital for uniform product quality. After the curing period, the reaction mixture is carefully quenched with water and neutralized to a pH of 8-9 using sodium hydroxide, a step that precipitates the crude product while leaving soluble impurities in the aqueous phase. The subsequent extraction with toluene and washing with saturated sodium chloride further refine the organic layer, removing residual acids and inorganic salts. Finally, the crude material undergoes reduced pressure solid distillation and recrystallization with isopropyl ether, a dual-purification strategy that effectively strips away trace isomers and colored impurities, resulting in a white crystalline product with a GC purity exceeding 99%.
How to Synthesize 2-Amino-4,6-Lutidine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety at scale. The process is divided into distinct operational phases, beginning with the preparation of the reaction vessel and the controlled addition of reagents to manage exotherms and mixing efficiency. Operators must monitor the temperature profiles closely during the reflux and curing stages to prevent runaway reactions, while the workup procedures demand precise pH control to maximize recovery yields. The following guide outlines the standardized operational framework derived from the patent examples, serving as a blueprint for process engineers aiming to replicate this high-efficiency route in a commercial setting. For the complete, step-by-step technical instructions including specific flow rates and equipment specifications, please refer to the detailed protocol below.
- Cyclize 3-aminocrotonenitrile in acetic acid at 120-130°C to form the intermediate 6-amino-2,4-dimethyl-3-pyridine nitrile.
- Hydrolyze the intermediate in 98% concentrated sulfuric acid at 160-180°C, followed by neutralization and extraction.
- Purify the crude product via reduced pressure distillation and recrystallization with isopropyl ether to achieve >99% GC purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers transformative benefits for procurement managers and supply chain leaders tasked with optimizing costs and ensuring continuity. The primary driver of cost reduction is the substitution of expensive, specialized reagents with ubiquitous industrial commodities; acetic acid, concentrated sulfuric acid, and sodium hydroxide are among the most widely produced chemicals globally, ensuring stable pricing and ready availability regardless of market fluctuations. By eliminating the need for precious metal catalysts like palladium and avoiding the logistical complexities associated with high-pressure hydrogen gas, the process drastically simplifies the supply chain infrastructure required for production. This shift not only lowers the direct material costs but also reduces the overhead associated with catalyst recovery, recycling, and disposal, contributing to a leaner and more agile manufacturing model that can respond quickly to market demand.
- Cost Reduction in Manufacturing: The economic advantage of this route is further amplified by the significant improvement in overall yield and the simplification of the purification train. Achieving a total yield of over 70% with a purity greater than 99% means that less raw material is wasted per kilogram of finished product, directly enhancing the cost-efficiency of the operation. Moreover, the avoidance of high-pressure equipment reduces capital depreciation costs and maintenance expenses, while the use of standard solvents like toluene and isopropyl ether allows for efficient solvent recovery and reuse systems. These factors combine to create a substantially lower cost of goods sold (COGS), enabling manufacturers to offer competitive pricing without sacrificing margin, a critical factor in the highly price-sensitive fine chemical and intermediate markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on non-proprietary, easily sourced raw materials that are not subject to the same geopolitical or logistical bottlenecks as specialized catalysts or high-pressure gases. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuums, ensures that production can be maintained consistently across different manufacturing sites with standard glass-lined or stainless steel reactors. This flexibility reduces the risk of production stoppages due to equipment failure or specialized part shortages, guaranteeing a steady flow of 2-amino-4,6-lutidine to downstream customers. Furthermore, the simplified workflow shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to urgent procurement requests from pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of hazardous high-pressure steps and the use of well-understood unit operations. The environmental footprint is also favorably impacted, as the process generates fewer toxic byproducts compared to nitration-based routes, and the aqueous waste streams are easier to treat due to the lack of heavy metal contaminants. The ability to operate within standard environmental compliance frameworks without needing specialized permits for high-risk operations accelerates the timeline for regulatory approval and plant commissioning. This scalability ensures that suppliers can rapidly ramp up production capacity to meet surging demand for agrochemical intermediates, securing long-term supply contracts and fostering trust with global partners who prioritize sustainable and compliant manufacturing practices.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis method, we have compiled a set of frequently asked questions based on the specific data and beneficial effects outlined in patent CN111303047B. These answers provide clarity on the operational nuances, purity profiles, and scalability potential of the process, helping stakeholders make informed decisions about integrating this technology into their supply chains. Understanding these details is crucial for evaluating the feasibility of adoption and anticipating the performance of the final product in downstream applications. The following section covers the most critical aspects of the synthesis, from raw material selection to final product specifications.
Q: What are the key advantages of the 3-aminocrotonenitrile route over traditional methods?
A: Unlike traditional routes requiring high-pressure hydrogenation or hazardous nitration, this method utilizes mild cyclization and hydrolysis, significantly improving safety and reaction controllability while achieving yields over 70%.
Q: How is high purity (>99%) achieved in this synthesis?
A: High purity is secured through a rigorous purification sequence involving pH-controlled precipitation, toluene extraction, reduced pressure solid distillation, and final recrystallization using isopropyl ether.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses cheap, readily available industrial raw materials like acetic acid and sulfuric acid, and avoids extreme high-pressure conditions, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4,6-Lutidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates like 2-amino-4,6-lutidine in the development of next-generation pharmaceuticals and agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials that meet the most stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying every batch against exacting standards, including GC purity analysis and NMR confirmation, guaranteeing that the product delivered matches the high-performance profile described in the patent literature. We are committed to bridging the gap between innovative academic research and industrial reality, translating complex synthetic routes into robust, GMP-compliant manufacturing processes.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments to validate the fit for your supply chain. Whether you are developing new neonicotinoid insecticides or exploring novel pyridine-based therapeutics, our expertise in process optimization and scale-up ensures that you have a dependable partner ready to support your growth. Contact us today to discuss how we can assist in securing your supply of high-purity 2-amino-4,6-lutidine and driving your projects forward with confidence.
