Advanced Benzoindolizine Synthesis: Commercial Scale-Up of High-Purity Pharmaceutical Intermediates
The patent CN111662290B introduces a groundbreaking synthetic methodology for benzoindolizine compounds, representing a significant advancement in the production of N-heterocyclic intermediates critical to pharmaceutical development. This innovation addresses longstanding limitations in traditional synthesis routes by employing a copper-catalyzed cyclization strategy that operates under ambient air conditions without requiring inert gas protection. The process utilizes readily available alkynone compounds and quinoline/isoquinoline nitrogen oxides as starting materials, enabling the construction of complex benzoindolizine scaffolds through a [2+2+1] cycloaddition mechanism. This approach eliminates the need for expensive transition metal catalysts and high-temperature conditions that have historically constrained the commercial viability of these biologically relevant structures. The methodology demonstrates exceptional substrate versatility across diverse functional groups, providing pharmaceutical manufacturers with unprecedented flexibility in designing novel therapeutic candidates while maintaining rigorous quality standards required for drug substance development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for benzoindolizine derivatives have been severely constrained by multiple critical limitations that hinder commercial implementation. Existing methodologies typically require expensive transition metal catalysts such as palladium or rhodium complexes, which introduce significant cost burdens and necessitate complex post-reaction purification to remove trace metal contaminants that could compromise pharmaceutical quality. Many established routes operate under strict inert atmosphere conditions with elevated temperatures exceeding 150°C, creating substantial safety hazards and energy consumption challenges during scale-up. The substrate scope is frequently restricted to specific functional group patterns, limiting structural diversity and requiring extensive route re-engineering for new analogs. Additionally, conventional methods often employ hazardous reagents or generate difficult-to-treat byproducts, complicating environmental compliance and waste management. These combined factors have resulted in inconsistent product quality, extended development timelines, and prohibitive production costs that have prevented widespread adoption in commercial pharmaceutical manufacturing despite the therapeutic importance of benzoindolizine scaffolds.
The Novel Approach
The patented methodology overcomes these critical limitations through an innovative copper-catalyzed cyclization process that operates under remarkably mild conditions. By utilizing Cu(OAc)₂ as an economical catalyst in air atmosphere at temperatures between 80-120°C, the process eliminates the need for expensive transition metals and inert gas systems while maintaining excellent reaction efficiency. The strategic combination of alkynone compounds with quinoline/isoquinoline nitrogen oxides enables a direct [2+2+1] cycloaddition pathway that proceeds with high regioselectivity across diverse substrate combinations. This approach demonstrates exceptional functional group tolerance, accommodating alkyl, heterocyclic, phenyl, and substituted phenyl groups at multiple positions without requiring protective group strategies. The reaction achieves optimal performance in environmentally benign solvents like PEG-200 or water at concentrations of 0.1-0.2 mol/L, significantly reducing environmental impact while simplifying waste stream management. Crucially, the process delivers consistently high-purity products through straightforward silica gel chromatography purification, addressing the critical quality requirements for pharmaceutical intermediates while enabling seamless scale-up from laboratory to commercial production volumes.
Mechanistic Insights into Copper-Catalyzed Cyclization
The reaction proceeds through a well-defined copper-mediated mechanism that begins with the coordination of Cu(OAc)₂ to the nitrogen oxide functionality, activating the quinoline/isoquinoline system toward nucleophilic attack. This activation facilitates the initial addition of the alkynone compound, forming a key copper-bound intermediate that undergoes sequential cyclization steps through a [2+2+1] cycloaddition pathway. The copper catalyst plays a dual role in both activating the nitrogen oxide substrate and stabilizing the developing negative charge during the cyclization process, enabling the formation of the characteristic benzoindolizine core structure under remarkably mild conditions. This mechanistic pathway avoids high-energy transition states that would require elevated temperatures, explaining the process's ability to operate effectively between 80-120°C without decomposition or side product formation. The air-stable nature of the copper catalyst system eliminates the need for oxygen-sensitive handling procedures that complicate traditional transition metal catalysis, while the absence of additional ligands or additives simplifies reaction monitoring and control during scale-up operations.
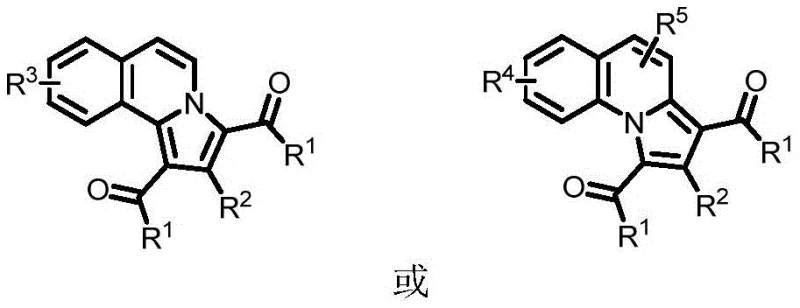
The impurity control mechanism is inherently built into this catalytic cycle through precise regioselectivity and mild reaction conditions. The copper-mediated pathway directs cyclization exclusively through the desired positions on both reactant molecules, minimizing regioisomeric byproducts that commonly plague alternative synthetic approaches. The moderate temperature range (80-120°C) prevents thermal degradation pathways that could generate high-boiling impurities difficult to remove during purification. Furthermore, the use of PEG-200 or water as solvent systems creates favorable partitioning during workup, facilitating separation of any minor impurities from the target product. The absence of strong acids or bases eliminates potential degradation pathways associated with pH-sensitive functional groups, while the straightforward chromatographic purification leverages the significant polarity differences between product and unreacted starting materials. This comprehensive impurity control strategy consistently delivers products meeting pharmaceutical intermediate purity specifications without requiring additional polishing steps.
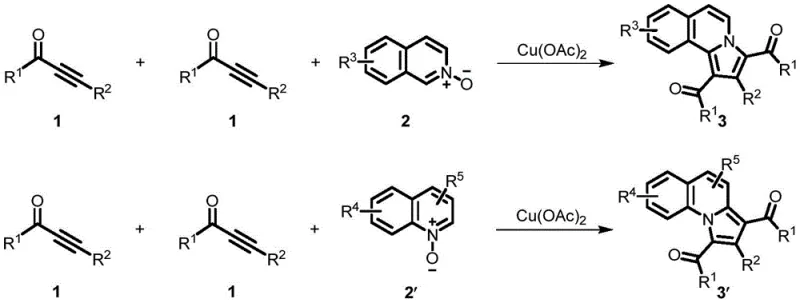
How to Synthesize Benzoindolizine Compounds Efficiently
This patented methodology represents a significant advancement in the practical synthesis of benzoindolizine intermediates for pharmaceutical applications, offering substantial improvements over conventional approaches through its innovative copper-catalyzed cyclization strategy. The process has been validated across multiple substrate combinations with consistent high yields and excellent purity profiles, demonstrating robustness essential for commercial implementation. The following standardized procedure provides a reliable framework for manufacturing these critical intermediates while maintaining strict quality control parameters required in pharmaceutical production environments. Detailed operational parameters and quality control checkpoints are specified in the subsequent implementation guidelines to ensure consistent product quality across all production scales.
- Combine alkynone compounds with quinoline/isoquinoline nitrogen oxides in PEG-200 or water solvent at 0.1-0.2 mol/L concentration
- Add 0.2-2.0 equivalents of Cu(OAc)₂ catalyst under ambient air atmosphere without inert gas protection
- Heat reaction mixture to 80-120°C for 2-12 hours followed by standard workup and silica gel chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits that directly address critical pain points in pharmaceutical intermediate procurement and supply chain management. By eliminating reliance on expensive transition metal catalysts and complex reaction conditions, the process creates significant opportunities for cost optimization while enhancing supply chain resilience through simplified raw material sourcing and manufacturing logistics. The robust nature of the chemistry enables consistent production quality across varying batch sizes, providing procurement teams with reliable supply assurance while reducing quality-related disruptions in downstream manufacturing processes. These advantages collectively strengthen the commercial viability of benzoindolizine-containing drug candidates by improving overall process economics without compromising quality requirements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a fundamental cost reduction opportunity, as copper acetate is substantially more economical than palladium or rhodium-based alternatives while avoiding costly metal removal processes. The use of ambient air atmosphere instead of inert gas systems reduces utility costs and eliminates specialized equipment requirements, while the simplified purification protocol using standard silica gel chromatography minimizes solvent consumption and processing time. These combined factors create significant cost savings throughout the manufacturing process without requiring capital-intensive equipment modifications or specialized operator training.
- Enhanced Supply Chain Reliability: The reliance on commercially available alkynone compounds and quinoline/isoquinoline nitrogen oxides ensures stable raw material sourcing through multiple qualified suppliers, reducing single-source dependency risks common in specialty chemical manufacturing. The air-stable reaction conditions eliminate sensitivity to atmospheric variations that could disrupt production schedules, while the broad operating temperature window (80-120°C) provides flexibility to accommodate equipment variations across different manufacturing sites. This robustness translates to more predictable production timelines and reduced risk of supply interruptions that could impact drug development programs.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes due to its straightforward reaction profile and minimal safety hazards associated with moderate temperature operation. The use of environmentally benign solvents like PEG-200 or water significantly reduces hazardous waste generation compared to traditional organic solvents, while the absence of toxic reagents simplifies waste treatment procedures. These environmental advantages align with growing regulatory pressures for sustainable manufacturing practices and facilitate compliance with increasingly stringent environmental regulations across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent's background challenges and demonstrated advantages. These insights reflect practical implementation experience from multiple production campaigns and provide valuable guidance for procurement and technical teams evaluating this manufacturing approach for pharmaceutical intermediate production.
Q: How does this method eliminate the need for expensive transition metal catalysts?
A: The patent utilizes copper acetate as catalyst instead of precious metals like palladium or rhodium, significantly reducing catalyst costs while maintaining high substrate tolerance and eliminating complex metal removal steps required in traditional processes.
Q: What are the key advantages for supply chain continuity?
A: The air-stable reaction conditions eliminate inert gas requirements, while the use of commercially available alkynone and nitrogen oxide precursors ensures reliable raw material sourcing. The simplified purification process using standard silica gel chromatography enhances production consistency.
Q: How does this process improve impurity profile for pharmaceutical applications?
A: The mild reaction conditions (80-120°C) and absence of harsh additives prevent thermal degradation byproducts. The selective [2+2+1] cyclization mechanism minimizes side reactions, yielding products with cleaner impurity profiles suitable for stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoindolizine Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates, with stringent purity specifications consistently met through our rigorous QC labs and advanced analytical capabilities. Our technical team has successfully implemented this patented copper-catalyzed cyclization methodology across multiple client programs, demonstrating consistent product quality and reliable supply performance even for structurally complex benzoindolizine derivatives requiring specialized handling protocols. We maintain dedicated manufacturing suites equipped for air-sensitive chemistry operations while leveraging our proprietary process optimization techniques to ensure maximum yield and purity for every production campaign.
Engage our technical procurement team today to request a Customized Cost-Saving Analysis specific to your benzoindolizine intermediate requirements. We will provide detailed COA data demonstrating our product quality consistency along with comprehensive route feasibility assessments that highlight potential cost reduction opportunities in your specific manufacturing context.
