Revolutionizing Tryptamine Derivative Production: A Deep Dive into Immobilized Catalytic Technology
The pharmaceutical industry is constantly seeking robust synthetic routes that balance high purity with operational efficiency, particularly for complex tryptamine derivatives. Patent CN102391170A introduces a transformative approach to the preparation of N,N-diallyl-5-methoxytryptamine hydrochloride, a critical intermediate with significant pharmacological potential. This technology shifts the paradigm from traditional homogeneous inorganic base catalysis to a sophisticated heterogeneous system utilizing an organic amine immobilized catalyst. By leveraging this advanced catalytic architecture, manufacturers can overcome longstanding bottlenecks related to reaction time, environmental impact, and post-reaction processing complexity. For R&D directors and procurement specialists alike, understanding this shift is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N-diallyl-5-methoxytryptamine hydrochloride has relied heavily on inorganic base catalysts, which present substantial drawbacks for large-scale manufacturing. Prior art indicates that these conventional processes often necessitate a catalyst loading up to three times the molar amount of the starting 5-methoxytryptamine, creating a massive burden on raw material costs and waste disposal. Furthermore, the reaction kinetics are sluggish, typically requiring extended heating periods of up to 16 hours to reach completion, which severely limits throughput in a commercial reactor. The post-reaction workup is equally problematic, involving intricate extraction and washing steps to remove residual inorganic salts, leading to significant solvent consumption and potential product loss. These inefficiencies not only inflate the cost reduction in pharmaceutical intermediates manufacturing but also introduce variability that complicates quality control protocols.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes an organic amine immobilized catalyst supported on silicon oxide, fundamentally altering the reaction landscape. This heterogeneous system operates effectively at room temperature, reducing the reaction window to a mere 6 to 8 hours while maintaining superior conversion rates. The physical state of the catalyst allows for a simplified workup procedure where the catalyst is removed via simple filtration rather than complex aqueous quenching. This innovation not only accelerates the production cycle but also aligns with green chemistry principles by minimizing solvent use and hazardous waste generation. For supply chain heads, this translates to a more predictable and streamlined manufacturing process that enhances overall supply chain reliability and reduces the risk of batch failures due to processing errors.
Mechanistic Insights into Organic Amine Immobilized Catalyzed Alkylation
The core of this technological advancement lies in the unique interaction between the substrate and the immobilized tertiary amine groups on the silica support. The reaction proceeds through a nucleophilic substitution mechanism where the amine nitrogen of the 5-methoxytryptamine attacks the allyl chloride. The immobilized catalyst acts as a proton scavenger and phase transfer facilitator, stabilizing the transition state without dissolving into the reaction medium. This heterogeneity ensures that the catalytic active sites remain accessible while preventing contamination of the final product with catalyst residues. Such precision is crucial for achieving the reported content of ≥98%, as it eliminates the need for aggressive purification steps that might degrade the sensitive tryptamine scaffold. Understanding this mechanism is key for any team evaluating the feasibility of commercial scale-up of complex pharmaceutical intermediates.
Beyond the primary transformation, the impurity control mechanism inherent in this solid-supported system offers distinct advantages over homogeneous alternatives. In traditional liquid-phase catalysis, side reactions such as over-alkylation or polymerization of the allyl group can occur more readily due to uncontrolled local concentrations of base. The immobilized nature of the catalyst creates a microenvironment that moderates reactivity, thereby suppressing the formation of closely related impurities. Additionally, the ability to activate and recycle the catalyst using sodium carbonate solution ensures that the catalytic activity remains consistent across multiple batches. This consistency is paramount for maintaining stringent purity specifications required by regulatory bodies, ensuring that every batch of high-purity pharmaceutical intermediates meets the rigorous standards expected by global partners.
How to Synthesize N,N-Diallyl-5-Methoxytryptamine Hydrochloride Efficiently
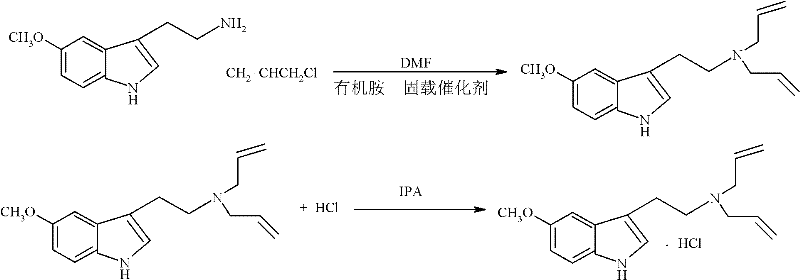
Implementing this synthesis route requires careful attention to the specific ratios and conditions outlined in the patent to maximize yield and purity. The process begins with dissolving the 5-methoxytryptamine starting material in DMF, followed by the addition of the specialized organic amine immobilized catalyst and allyl chloride. The reaction is allowed to proceed under mild conditions, after which the catalyst is filtered off for regeneration. The filtrate is then concentrated, and the residue is treated with isopropanol and hydrochloric acid to precipitate the final hydrochloride salt. While the general concept is straightforward, the precise execution of these steps is critical for success. Detailed standardized synthesis steps see the guide below for exact parameters regarding mass volume ratios and activation procedures.
- Dissolve 5-methoxytryptamine in DMF and add the organic amine immobilized catalyst along with allyl chloride.
- Stir the mixture at room temperature for 6 to 8 hours to complete the substitution reaction.
- Filter the reaction liquid to recover the catalyst, evaporate the filtrate, dissolve in IPA, and treat with hydrochloric acid to form the salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers compelling economic and operational benefits that extend beyond simple yield improvements. The shift to a recyclable immobilized catalyst system directly addresses the rising costs associated with raw materials and waste management in fine chemical production. By eliminating the need for excessive inorganic bases and simplifying the isolation process, manufacturers can achieve substantial cost savings without compromising on product integrity. Furthermore, the reduction in reaction time from 16 hours to under 8 hours effectively doubles the capacity of existing reactor infrastructure, allowing for faster turnaround times on orders. These factors combined create a more resilient supply chain capable of meeting fluctuating market demands with greater agility and efficiency.
- Cost Reduction in Manufacturing: The implementation of a reusable organic amine immobilized catalyst significantly lowers the variable costs associated with each production batch. Unlike traditional methods that consume stoichiometric or excess amounts of inorganic bases which end up as waste, this catalyst can be recovered, activated, and reintroduced into the process multiple times. This循环利用 (recycling) capability drastically reduces the expenditure on catalytic materials and minimizes the costs linked to the disposal of hazardous chemical waste. Consequently, the overall cost of goods sold is optimized, providing a competitive edge in pricing strategies for high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The simplified workup procedure, characterized by a straightforward filtration step instead of complex extractions, reduces the operational complexity and the potential for human error during manufacturing. This robustness ensures higher batch success rates and more consistent delivery schedules, which is critical for maintaining continuity in the supply of key drug substances. Additionally, the use of common solvents like DMF and IPA, coupled with mild reaction conditions, reduces dependency on specialized equipment or extreme utility requirements. This accessibility enhances the reliability of the production network, ensuring that lead times for high-purity pharmaceutical intermediates remain stable even during periods of high demand.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with modern sustainability goals by reducing the E-factor (mass of waste per mass of product). The ability to recycle the catalyst and the reduction in solvent usage for workup contribute to a smaller environmental footprint, facilitating easier compliance with increasingly strict environmental regulations. Moreover, the process is inherently scalable; the heterogeneous nature of the reaction translates well from laboratory glassware to large-scale industrial reactors without significant re-engineering. This scalability ensures that production volumes can be ramped up from 100 kgs to 100 MT annual commercial production seamlessly, supporting long-term growth strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of N,N-diallyl-5-methoxytryptamine hydrochloride. These insights are derived directly from the technical specifications and beneficial effects described in the underlying patent literature. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains. We encourage partners to review these points to fully appreciate the value proposition of this advanced manufacturing route.
Q: How does the immobilized catalyst improve the workup process compared to traditional inorganic bases?
A: Traditional methods often require complex aqueous workups to remove soluble inorganic salts. The immobilized catalyst allows for simple filtration, significantly reducing solvent usage and waste generation while enabling catalyst recycling.
Q: What are the purity and yield specifications achievable with this patented method?
A: According to patent CN102391170A, this method consistently achieves a chemical content of ≥98% and a reaction yield of ≥88%, ensuring high quality for downstream pharmaceutical applications.
Q: Is the catalyst reusable, and how does this impact production costs?
A: Yes, the organic amine immobilized catalyst can be activated with sodium carbonate solution and reused. This recyclability drastically reduces raw material consumption and lowers the overall cost of goods sold (COGS).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Diallyl-5-Methoxytryptamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to ensure the highest quality and efficiency in chemical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of patented technologies like the immobilized catalyst system are fully realized at an industrial level. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch meets the ≥98% content standard required for pharmaceutical applications. Our dedication to technical excellence makes us a trusted partner for companies seeking to optimize their supply chains with superior intermediates.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us help you secure a stable, high-quality supply of this essential intermediate while driving down costs and improving operational efficiency.
