Scalable Synthesis of 3-Hydroxyoxetane via Glycerol Derivatives for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries have long recognized the strategic value of oxetane rings as versatile molecular building blocks, particularly for enhancing the solubility and metabolic stability of drug candidates. Patent CN103554064A, published in early 2014, introduces a robust and scalable preparation method for 3-hydroxy oxetane compounds that addresses significant historical limitations in synthetic accessibility. Unlike earlier methodologies that relied on hazardous reagents or extreme conditions, this invention leverages substituted glycerol as a renewable and abundant starting material to construct the strained four-membered ring system. The disclosed process employs a logical sequence of protection, functionalization, and cyclization steps that operate under remarkably mild conditions, thereby minimizing thermal degradation and safety risks associated with high-energy intermediates. For R&D directors and process chemists, this patent represents a pivotal shift towards safer, more sustainable manufacturing protocols that do not compromise on the structural integrity or purity of the final active pharmaceutical ingredient intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxetane derivatives has been plagued by reliance on dangerous reagents and energetically demanding processes that complicate industrial adoption. For instance, British Patent no. 787406 describes a method utilizing triols reacted with carbonic acid derivatives, which frequently necessitates the use of phosgene, a highly toxic and regulated gas that imposes severe containment and disposal costs on manufacturing facilities. Alternative approaches, such as those found in Japanese Patent 10-7669, involve the thermal decomposition of cyclic carbonates at temperatures ranging from 160°C to 260°C, requiring specialized high-temperature reactors and consuming substantial energy resources. Furthermore, photochemical [2+2] cycloadditions like the Paterno-Buchi reaction demand inert atmospheres and high-pressure mercury lamps, creating bottlenecks in light penetration and throughput that are difficult to overcome in large-scale batch processing. These conventional pathways often result in complex impurity profiles due to side reactions promoted by harsh conditions, necessitating expensive and time-consuming purification steps that erode overall process economics.
The Novel Approach
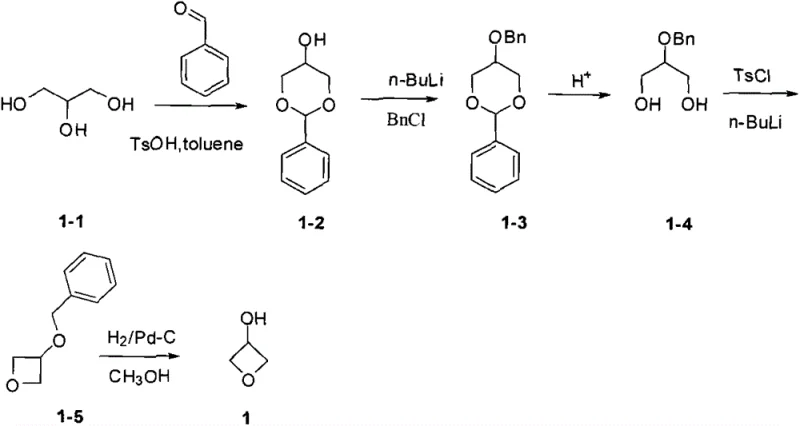
In stark contrast, the methodology outlined in CN103554064A circumvents these hazards by utilizing a stepwise organic synthesis strategy centered on readily available glycerol derivatives. The process initiates with a condensation reaction between substituted glycerol and aldehydes or ketones to form a stable acetal intermediate, followed by selective protection of the remaining hydroxyl group with a benzyl moiety. This strategic use of orthogonal protecting groups allows for the subsequent removal of the acetal under mild acidic conditions without disturbing the benzyl ether, setting the stage for a clean intramolecular cyclization. The final ring closure is achieved through a Williamson ether synthesis variant using tosyl chloride and a strong base, followed by a catalytic hydrogenation to reveal the target 3-hydroxy oxetane. This approach not only eliminates the need for toxic gases and extreme heat but also simplifies post-reaction workups, as each intermediate can be isolated or carried forward with high purity, significantly streamlining the path from raw material to commercial product.
Mechanistic Insights into Protective Group Strategy and Cyclization
The success of this synthetic route hinges on the precise orchestration of protective group chemistry to manage the reactivity of the polyol starting material. The initial formation of the cyclic acetal serves a dual purpose: it protects two of the hydroxyl groups while simultaneously directing the regioselectivity of the subsequent benzylation to the remaining primary or secondary alcohol. By employing n-butyllithium as a strong base in tetrahydrofuran, the synthesis ensures quantitative deprotonation of the specific hydroxyl site, allowing for efficient nucleophilic attack on benzyl chloride to install the robust benzyl protecting group. This benzyl group is strategically chosen for its stability against the acidic conditions used in the next step to hydrolyze the acetal, yet it remains susceptible to removal via catalytic hydrogenation at the very end of the sequence. This orthogonality is critical for preventing premature deprotection or cross-reactivity that could lead to oligomerization or the formation of intractable tars.
The core transformation of the entire process is the intramolecular cyclization step, which constructs the strained oxetane ring from a 1,3-diol precursor. After the acetal is removed to regenerate the diol functionality, one hydroxyl group is activated as a leaving group, typically a tosylate, using p-toluenesulfonyl chloride in the presence of a base. The addition of a second equivalent of base then generates an alkoxide at the adjacent position, which performs a nucleophilic attack on the carbon bearing the tosylate group. This intramolecular SN2 reaction displaces the sulfonate and closes the four-membered ring, a process that is entropically favored despite the ring strain involved. The careful control of stoichiometry and temperature during this phase is essential to suppress intermolecular etherification, ensuring that the reaction proceeds exclusively towards the desired monomeric oxetane structure rather than forming polymeric byproducts.
How to Synthesize 3-Hydroxyoxetane Efficiently
Executing this synthesis requires strict adherence to the sequential addition of reagents and precise temperature control to maintain high yields across all five steps. The process begins with the acid-catalyzed condensation in toluene with water removal, followed by low-temperature lithiation and benzylation to secure the protecting group. Subsequent acidic hydrolysis must be monitored closely to ensure complete acetal removal without degrading the benzyl ether, setting up the critical cyclization event.
- Condense substituted glycerol with benzaldehyde using p-toluenesulfonic acid catalyst in toluene to form the acetal protected intermediate.
- Protect the remaining hydroxyl group with a benzyl group using n-butyllithium and benzyl chloride in THF.
- Remove the acetal protecting group under acidic conditions to reveal the 1,3-diol motif required for cyclization.
- Perform intramolecular Williamson ether synthesis using tosyl chloride and base to close the oxetane ring.
- Execute catalytic hydrogenation using Pd-C to remove the benzyl protecting group and yield the final 3-hydroxyoxetane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this glycerol-based synthesis offers profound advantages in terms of raw material security and operational expenditure. By shifting away from specialized, high-cost reagents like phosgene or cyclic carbonates, manufacturers can source the majority of their inputs from the commodity chemical market, where glycerol and benzaldehyde are produced in massive volumes with stable pricing. This decoupling from niche, hazard-classified chemicals reduces the regulatory burden and insurance costs associated with storage and transport, directly contributing to a lower total cost of ownership for the intermediate. Furthermore, the mild reaction conditions eliminate the need for exotic metallurgy in reactor construction, allowing production to occur in standard glass-lined or stainless steel vessels found in most multipurpose chemical plants.
- Cost Reduction in Manufacturing: The elimination of toxic phosgene and high-temperature decomposition steps removes the necessity for expensive scrubbing systems and specialized high-pressure reactors, leading to significant capital expenditure savings. Additionally, the high yields reported in each step of the sequence minimize raw material waste, ensuring that a greater proportion of the input mass is converted into saleable product rather than discarded as hazardous waste. The use of common solvents like toluene and methanol further simplifies solvent recovery and recycling loops, reducing the ongoing operational costs associated with consumable materials and waste disposal fees.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the primary feedstock, glycerol, is a ubiquitous byproduct of the biodiesel industry with a robust and diversified global supply network. This abundance mitigates the risk of supply disruptions that often plague dependencies on single-source specialty reagents or controlled substances. The stability of the intermediates also allows for flexible production scheduling, where batches can be paused at stable protected stages without significant degradation, providing supply chain planners with greater agility to respond to fluctuating downstream demand from pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as distillation, extraction, and crystallization that are well-understood by plant engineers. The absence of heavy metal catalysts in the main synthetic line, reserving palladium only for the final hydrogenation step where it can be efficiently recovered, aligns with increasingly stringent environmental regulations regarding metal residues in APIs. This green chemistry profile facilitates faster regulatory approval and reduces the environmental footprint of the manufacturing site, supporting corporate sustainability goals without compromising production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route in a GMP environment. These answers are derived directly from the experimental data and process descriptions provided in the patent literature to ensure accuracy and relevance for technical decision-makers.
Q: Why is the glycerol-based route superior to traditional phosgene methods?
A: Traditional methods often rely on toxic phosgene or high-temperature thermal decomposition, posing significant safety and environmental risks. The glycerol-based route described in CN103554064A utilizes mild acid and base catalysis at ambient or reflux temperatures, eliminating the need for hazardous gas handling and specialized high-pressure equipment.
Q: What represents the key yield bottleneck in this synthesis?
A: While individual steps show high efficiency (often exceeding 85-90%), the cumulative yield of a five-step linear sequence requires rigorous process control. The critical step is the intramolecular cyclization, where precise stoichiometry of the base and activating agent is required to prevent polymerization or elimination side reactions.
Q: Is this process suitable for multi-ton commercial manufacturing?
A: Yes, the patent explicitly states suitability for industrialized production due to simple post-treatment and convenient detection. The use of common solvents like toluene, THF, and methanol, along with standard unit operations like distillation and crystallization, facilitates straightforward scale-up from pilot to commercial plant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxyoxetane Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of novel therapeutics depends on the consistent availability of high-quality intermediates like 3-hydroxyoxetane. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in preclinical development or late-stage manufacturing. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that you can rely on us not just as a vendor, but as a strategic partner dedicated to the success of your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies translate into tangible value for your organization. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven process capabilities.
