Advanced Synthetic Route for 3-Aryl Pyrroles: Enabling Scalable Pharmaceutical Intermediate Production
Advanced Synthetic Route for 3-Aryl Pyrroles: Enabling Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access nitrogen-containing heterocycles, which serve as the backbone for countless bioactive molecules. A significant breakthrough in this domain is detailed in patent CN109369496B, which discloses a novel synthetic method for 3-aryl substituted pyrrole compounds. This technology represents a paradigm shift from traditional pyrrole construction, utilizing a strategic oxidative ring contraction of readily available N-substituted piperidines. By transforming saturated six-membered rings into aromatic five-membered pyrrole systems through a copper-catalyzed process, this method bypasses the limitations of classical heterocycle synthesis. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: the ability to generate high-value scaffolds from low-cost, commodity feedstocks using robust catalytic cycles that are amenable to industrial optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aryl substituted pyrroles has been fraught with challenges that hinder efficient commercial production. Traditional methods, such as the Paal-Knorr synthesis, often suffer from poor regioselectivity, making it difficult to install substituents at specific positions without generating complex mixtures of isomers that require costly separation. Furthermore, many existing routes rely on specialized pyrrole precursors that are not only expensive but also possess limited availability in the global supply chain. These precursors often require multi-step preparation themselves, adding layers of complexity, waste, and time to the overall manufacturing process. Additionally, conventional cross-coupling strategies to introduce aryl groups frequently demand harsh conditions or sensitive organometallic reagents that complicate safety protocols and increase the environmental footprint of the operation.
The Novel Approach
In stark contrast, the methodology outlined in CN109369496B introduces a streamlined two-step sequence that fundamentally alters the economic landscape of pyrrole synthesis. The core innovation lies in the direct conversion of N-substituted piperidines into iodopyrrole intermediates via a cascade of oxidative ring shrinkage, decarboxylation, dehydrogenation, and aromatization reactions. This transformation is catalyzed by inexpensive copper salts in the presence of oxygen, turning a simple saturation into a functionalized aromatic system in a single pot. 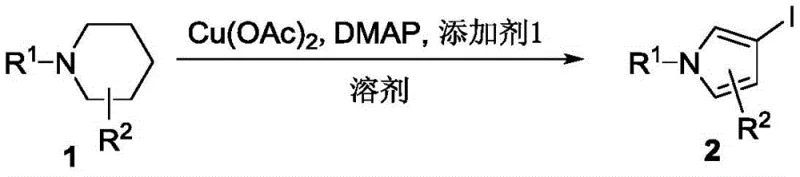 Following this, the resulting iodopyrrole undergoes a standard palladium-catalyzed cross-coupling with arylboronic acids to yield the final 3-aryl substituted product. This approach not only simplifies the operational workflow but also drastically expands the scope of accessible derivatives, allowing for the rapid generation of diverse libraries for drug discovery programs.
Following this, the resulting iodopyrrole undergoes a standard palladium-catalyzed cross-coupling with arylboronic acids to yield the final 3-aryl substituted product. This approach not only simplifies the operational workflow but also drastically expands the scope of accessible derivatives, allowing for the rapid generation of diverse libraries for drug discovery programs.
Mechanistic Insights into Copper-Catalyzed Oxidative Ring Contraction
The first stage of this synthetic pathway is a masterpiece of oxidative catalysis, where copper acetate acts as the primary driver for the structural reorganization of the piperidine ring. Under an oxygen atmosphere, the copper species facilitates the removal of hydrogen atoms and the cleavage of carbon-carbon bonds, effectively shrinking the six-membered ring into a five-membered pyrrole core. The presence of iodine and DMAP (4-dimethylaminopyridine) is critical, as they promote the beta-iodo substitution that installs the necessary handle for the subsequent coupling reaction. This tandem process of aromatization and functionalization eliminates the need for pre-functionalized pyrrole starting materials, which are often the cost bottleneck in traditional syntheses. The reaction proceeds smoothly in polar aprotic solvents like acetonitrile at moderate temperatures around 80°C, ensuring energy efficiency while maintaining high conversion rates across a wide range of substrates.
The second stage leverages the well-established Suzuki-Miyaura coupling mechanism to attach the aryl group with high precision.  Using a palladium salt such as Pd(OAc)2 alongside phosphine ligands and a base like potassium carbonate, the iodopyrrole intermediate reacts with arylboronic acids to form the carbon-carbon bond at the 3-position of the pyrrole ring. This step is highly tolerant of various functional groups on the aryl ring, including halogens, alkyls, and alkoxy groups, as demonstrated by the extensive substrate scope in the patent examples. The use of additives like pivalic acid and tetrabutylammonium bromide further enhances the catalytic turnover and stability, ensuring that the reaction reaches completion with minimal formation of homocoupling byproducts. This mechanistic robustness is key for R&D teams aiming to scale this chemistry for kilogram-level production of active pharmaceutical ingredients.
Using a palladium salt such as Pd(OAc)2 alongside phosphine ligands and a base like potassium carbonate, the iodopyrrole intermediate reacts with arylboronic acids to form the carbon-carbon bond at the 3-position of the pyrrole ring. This step is highly tolerant of various functional groups on the aryl ring, including halogens, alkyls, and alkoxy groups, as demonstrated by the extensive substrate scope in the patent examples. The use of additives like pivalic acid and tetrabutylammonium bromide further enhances the catalytic turnover and stability, ensuring that the reaction reaches completion with minimal formation of homocoupling byproducts. This mechanistic robustness is key for R&D teams aiming to scale this chemistry for kilogram-level production of active pharmaceutical ingredients.
How to Synthesize 3-Aryl Substituted Pyrrole Efficiently
Implementing this synthetic strategy requires careful attention to reaction parameters to maximize yield and purity. The process begins with the oxidative conversion of the piperidine precursor, where the stoichiometry of copper acetate and iodine must be optimized to drive the ring contraction to completion without over-oxidation. Following isolation of the iodopyrrole intermediate, the cross-coupling step demands an inert atmosphere and precise temperature control to activate the palladium catalyst effectively. Detailed standardized operating procedures regarding reagent addition sequences, quenching protocols, and purification methods are essential for reproducibility. For a comprehensive guide on executing this synthesis with specific molar ratios and workup details, please refer to the technical protocol below.
- Oxidative Ring Contraction: React N-substituted piperidine with copper acetate, iodine, and DMAP in acetonitrile under oxygen atmosphere at 80°C to form the iodopyrrole intermediate.
- Palladium-Catalyzed Coupling: Couple the resulting iodopyrrole with arylboronic acid using a palladium catalyst, ligand, and base in DMF or toluene at 90°C.
- Workup and Purification: Quench the reaction, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel chromatography to obtain the final 3-aryl pyrrole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for supply chain resilience and cost management. By shifting the starting material base from scarce, high-cost pyrroles to abundant, low-cost piperidines, manufacturers can achieve substantial reductions in raw material expenditure. The reliance on commodity chemicals like copper acetate and molecular oxygen instead of precious metal catalysts or exotic oxidants further drives down the cost of goods sold. Moreover, the operational simplicity of the two-step sequence reduces the number of unit operations required, minimizing labor costs and equipment occupancy time. This efficiency translates directly into improved margins and a more competitive pricing structure for the final pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive specialized starting materials is the primary driver for cost savings in this process. Traditional pyrrole synthesis often necessitates custom synthesis of building blocks, which incurs high premiums due to low volume and complex handling. By utilizing N-substituted piperidines, which are produced on a massive scale for other industries, the input cost is drastically lowered. Additionally, the use of copper as a primary catalyst in the first step replaces more expensive noble metals, and the mild reaction conditions reduce energy consumption associated with heating and cooling cycles. These factors combine to create a leaner, more economical manufacturing profile that is highly attractive for high-volume production.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the dependency on single-source suppliers for niche intermediates. This new method mitigates that risk by relying on globally available feedstocks. N-substituted piperidines and arylboronic acids are stocked by numerous chemical vendors worldwide, ensuring that production schedules are not disrupted by raw material shortages. The robustness of the catalytic system also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain. For procurement managers, this translates to reduced lead times and greater flexibility in sourcing strategies, allowing for better inventory management and responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden bottlenecks related to safety and waste generation. This methodology addresses these concerns by employing green chemistry principles, such as the use of oxygen as a terminal oxidant, which generates water as the only byproduct of the oxidation step. The reaction conditions are mild enough to be handled in standard glass-lined reactors without requiring specialized high-pressure or cryogenic equipment. Furthermore, the high atom economy of the coupling step minimizes waste generation, simplifying effluent treatment and reducing the environmental compliance burden. These attributes make the process inherently scalable, allowing for seamless transition from pilot batches to multi-ton commercial production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical application and scope of this technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Whether you are concerned about substrate compatibility or process safety, the information below clarifies the capabilities and limitations of this innovative route.
Q: What are the primary advantages of using N-substituted piperidines as starting materials?
A: N-substituted piperidines are commercially abundant, inexpensive commodity chemicals compared to specialized pyrrole building blocks. This route leverages their availability to access complex heterocycles through oxidative ring contraction, significantly lowering raw material costs.
Q: Does this method require harsh reaction conditions or exotic reagents?
A: No, the process operates under relatively mild conditions (40-100°C) using standard solvents like acetonitrile and DMF. The catalysts employed, such as copper acetate and palladium acetate, are widely available industrial reagents, facilitating easy scale-up.
Q: Is this synthetic route suitable for large-scale manufacturing of API intermediates?
A: Yes, the methodology demonstrates broad substrate tolerance and utilizes robust catalytic systems. The elimination of complex multi-step sequences typical of traditional pyrrole synthesis enhances operational simplicity, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl Substituted Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in maintaining a competitive edge in the pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN109369496B and is fully prepared to implement this copper-catalyzed oxidative ring contraction strategy for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is smooth and successful. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-aryl substituted pyrrole delivered meets the highest industry standards for API intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in catalytic processes and process intensification, we can help you secure a stable supply of high-quality intermediates. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term growth objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
