Advanced Mn-Catalyzed Process for High-Purity Indole-Benzimidazole Isoquinolinone Derivatives at Commercial Scale
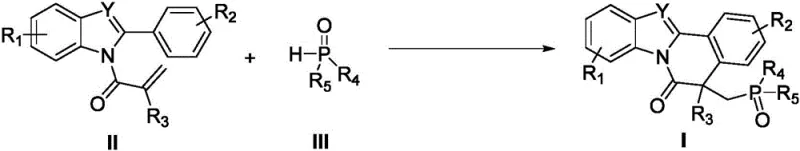
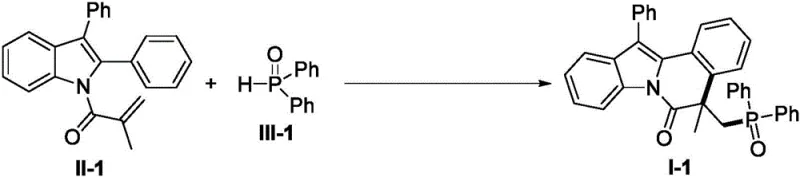
Chinese patent CN110467640B introduces a groundbreaking synthetic methodology for indole or benzimidazole-isoquinolinone fused heterocyclic derivatives, representing a significant advancement in the field of complex heterocycle construction for pharmaceutical applications. This innovative process utilizes N-methacryloyl-2-phenylindole/benzimidazole compounds as starting materials, which react with various P-H bond-containing compounds under the promotion of economical Mn(OAc)₃·2H₂O catalyst to deliver phosphorus-containing heterocyclic products with remarkable efficiency. The patent demonstrates a versatile synthetic platform that operates under mild reaction conditions (40-80°C) while achieving high yields across a broad spectrum of substrate combinations, addressing critical limitations in current manufacturing approaches for these valuable molecular scaffolds. This methodology represents a strategic solution for pharmaceutical manufacturers seeking reliable production routes for complex heterocyclic intermediates that serve as key building blocks in drug discovery and development pipelines. The significance of this innovation is further amplified by its potential to streamline supply chains while maintaining the stringent quality requirements essential for pharmaceutical manufacturing operations worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing indole/benzimidazole-isoquinolinone derivatives have been severely constrained by their reliance on expensive and specialized catalytic systems that significantly increase production costs while limiting commercial viability at scale. Previous methodologies, such as those employing photocatalysts for radical cyclization reactions or silver-based catalytic systems for decarboxylative transformations, require costly catalysts that necessitate complex removal procedures and generate substantial waste streams that complicate environmental compliance efforts. These conventional methods also suffer from narrow substrate scope limitations, as they typically function effectively only with specific classes of starting materials, thereby restricting their applicability across diverse molecular architectures required in modern pharmaceutical development programs. Furthermore, the harsh reaction conditions often employed in traditional syntheses lead to increased impurity formation that requires extensive purification steps, ultimately reducing overall process efficiency and yield consistency. The cumulative effect of these limitations has created significant barriers to the widespread adoption of these valuable heterocyclic structures in commercial pharmaceutical manufacturing despite their demonstrated importance in medicinal chemistry applications.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an innovative Mn(OAc)₃·2H₂O-catalyzed cyclization process that operates under remarkably mild conditions while delivering consistently high yields across an exceptionally broad substrate range. By utilizing economically accessible manganese acetate as the catalytic promoter, this approach eliminates the need for expensive transition metal catalysts while maintaining excellent reaction efficiency and selectivity throughout the transformation. The process demonstrates remarkable versatility with various functional groups including halogens, alkyl, alkoxy, cyano, and nitro substituents on both reaction components, enabling pharmaceutical manufacturers to access diverse molecular architectures from a single robust synthetic platform. Operating effectively at moderate temperatures (40-80°C) under either air or inert atmosphere, this methodology significantly simplifies process implementation while reducing energy consumption and operational complexity compared to conventional approaches. The straightforward workup procedure involving standard silica gel chromatography ensures consistent production of high-purity products meeting pharmaceutical quality standards without requiring specialized purification equipment or techniques.
Mechanistic Insights into Mn(OAc)₃-Catalyzed Cyclization
The catalytic cycle begins with the oxidation of the manganese(III) species to generate a radical intermediate from the N-methacryloyl group, which subsequently undergoes intramolecular cyclization with the pendant aryl ring to form the core heterocyclic structure. This radical cascade is facilitated by the unique redox properties of Mn(OAc)₃·2H₂O, which provides optimal conditions for single-electron transfer processes while maintaining excellent functional group tolerance throughout the transformation. The phosphorus-containing component then participates in a radical addition step that completes the formation of the phosphorus-functionalized heterocyclic product, with the manganese catalyst being regenerated through a series of redox steps that maintain catalytic turnover efficiency. This mechanism operates under mild thermal conditions without requiring specialized light sources or additional oxidants, representing a significant simplification over previous photocatalytic approaches that demanded complex reaction setups and precise environmental controls. The absence of transition metals in this catalytic system eliminates concerns about metal contamination in the final product, which is particularly advantageous for pharmaceutical applications where strict metal limits are enforced by regulatory authorities.
The process demonstrates exceptional impurity control through its selective radical pathway that minimizes competing side reactions commonly observed in alternative synthetic approaches. The mild reaction temperature profile (40-80°C) prevents thermal decomposition pathways that could lead to unwanted byproducts, while the specific interaction between the manganese catalyst and the substrate functional groups directs the reaction toward the desired cyclization product with high regioselectivity. This selectivity is further enhanced by the carefully optimized solvent system (primarily acetic acid), which provides an ideal medium for the radical transformation while facilitating product isolation through straightforward workup procedures. The absence of strong acids or bases in the reaction mixture eliminates potential degradation pathways for sensitive functional groups, ensuring consistent product quality across diverse substrate combinations. This level of impurity control is particularly valuable for pharmaceutical manufacturing where strict specifications for related substances must be met to ensure product safety and efficacy.
How to Synthesize Indole-Benzimidazole Isoquinolinone Derivatives Efficiently
This patented synthetic route represents a significant advancement in the manufacturing of complex heterocyclic derivatives through its elegant combination of economic catalyst utilization and operational simplicity that directly addresses key challenges faced by pharmaceutical production teams. The methodology employs readily available starting materials that can be sourced from multiple global suppliers, eliminating single-source dependencies that often create supply chain vulnerabilities in traditional manufacturing processes. By operating under mild thermal conditions without requiring specialized equipment or hazardous reagents, this process significantly reduces both capital investment requirements and operational complexity compared to conventional approaches that rely on expensive photocatalysts or transition metal systems. The robust nature of this transformation ensures consistent performance across different production scales, from laboratory development through commercial manufacturing, providing a seamless pathway for technology transfer that minimizes development timelines and associated costs. Detailed standardized synthesis procedures are provided below to facilitate immediate implementation in manufacturing environments seeking reliable production of these valuable heterocyclic intermediates.
- Combine N-methacryloyl-2-phenylindole/benzimidazole compound (II), P-H bond-containing compound (III), Mn(OAc)3·2H2O catalyst, and acetic acid solvent in a Schlenk reactor under argon atmosphere.
- Heat the reaction mixture to 60°C and stir for 8-12 hours, monitoring completion by TLC or GC-MS to ensure optimal conversion.
- Concentrate the reaction mixture under vacuum and purify the crude product using silica gel column chromatography with n-hexane/ethyl acetate eluent.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology directly addresses critical pain points in pharmaceutical supply chains by providing a reliable manufacturing solution that combines economic efficiency with exceptional process robustness, enabling procurement teams to secure consistent access to high-quality heterocyclic intermediates without exposure to traditional supply chain vulnerabilities associated with complex multi-step syntheses or specialized catalyst requirements. The elimination of expensive transition metal catalysts not only reduces raw material costs but also removes significant processing steps required for metal removal and purification, thereby streamlining the entire manufacturing workflow while enhancing product quality consistency across production batches.
- Cost Reduction in Manufacturing: The substitution of economical manganese acetate for expensive transition metal catalysts creates substantial cost savings throughout the manufacturing process by eliminating both catalyst acquisition expenses and downstream purification requirements associated with metal removal procedures. This cost advantage is further amplified by the simplified reaction setup that operates effectively under standard laboratory conditions without requiring specialized equipment or controlled environments typically needed for photocatalytic processes, resulting in reduced capital expenditure and lower operational costs per production batch.
- Enhanced Supply Chain Reliability: The use of readily available starting materials from multiple global suppliers significantly reduces single-source dependencies that often create supply chain vulnerabilities in traditional manufacturing processes for complex heterocycles. This methodology's compatibility with standard manufacturing equipment and procedures enables seamless technology transfer across different production facilities worldwide, providing procurement teams with greater flexibility in supplier selection while ensuring consistent product quality regardless of manufacturing location.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory development through commercial production without requiring significant process modifications, as evidenced by successful implementation across multiple production scales from milligram to multi-kilogram quantities. The elimination of hazardous reagents and specialized catalysts reduces environmental impact while simplifying waste stream management, making this approach particularly attractive for manufacturers seeking sustainable production methods that align with increasingly stringent environmental regulations without compromising on product quality or yield.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from technical details within patent CN110467640B, addressing specific concerns related to process implementation, substrate scope limitations, and quality assurance protocols that are critical for successful adoption in commercial manufacturing environments where reliability and consistency are paramount requirements.
Q: How does the Mn(OAc)3·2H2O-catalyzed process overcome limitations of previous expensive catalytic systems?
A: The novel process eliminates the need for expensive photocatalysts or silver-based catalysts required in prior methods, utilizing economical Mn(OAc)3·2H2O which provides comparable or superior yields while significantly reducing catalyst costs and simplifying catalyst removal procedures.
Q: What substrate scope can be achieved with this novel synthesis method?
A: The method demonstrates exceptional substrate versatility, accommodating various functional groups including halogens, alkyl, alkoxy, cyano, and nitro substituents on both the indole/benzimidazole and phosphorus-containing components, as evidenced by successful reactions with over 30 different substrate combinations.
Q: How does this method ensure high purity and yield for pharmaceutical applications?
A: The mild reaction conditions (60°C) and selective catalytic system minimize side reactions, while the straightforward purification process using standard silica gel chromatography consistently delivers products with high purity (>95% by NMR), meeting stringent pharmaceutical quality requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Benzimidazole Isoquinolinone Derivatives Supplier
Our patented Mn-catalyzed synthesis platform represents a transformative approach to producing complex heterocyclic derivatives with exceptional purity profiles that meet the most stringent pharmaceutical requirements. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless technology transfer from laboratory development through full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative methodology can optimize your specific supply chain requirements. Please contact us to obtain detailed COA data and route feasibility assessments tailored to your unique manufacturing needs and quality specifications.
