Advanced Organocatalytic Synthesis of 2-Trifluoromethylquinoline Intermediates for Commercial Scale-Up
Introduction to Next-Generation Fluorinated Heterocycle Synthesis
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to access fluorinated heterocyclic scaffolds, particularly the 2-trifluoromethylquinoline motif, which serves as a critical backbone for numerous bioactive agents. As detailed in Chinese Patent CN115466212A, a novel synthetic strategy has been developed that addresses the longstanding challenges associated with constructing these complex molecular architectures. This innovation is particularly relevant for the development of antimalarial drugs like mefloquine, serotonin receptor modulators, and potent AChE inhibitors, all of which rely on the unique physicochemical properties imparted by the trifluoromethyl group. 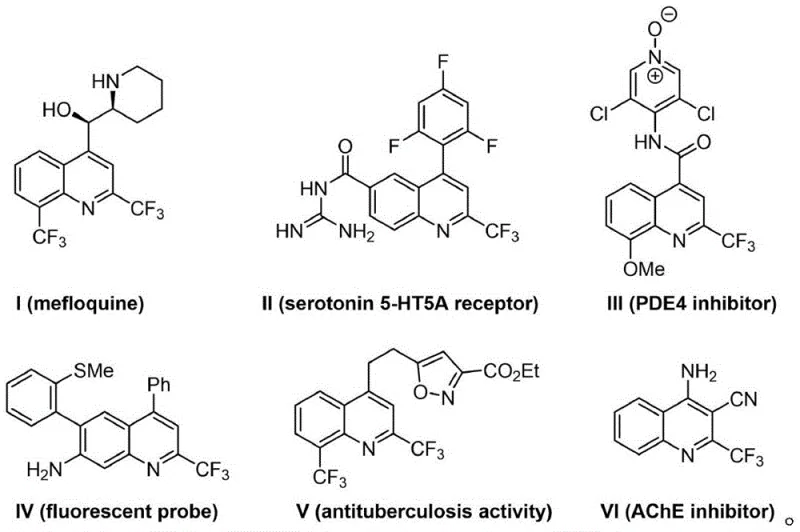 The patent highlights that introducing fluorine atoms or fluorine-containing groups into specific positions of organic molecules can significantly alter their acidity, lipophilicity, and metabolic stability, thereby enhancing their therapeutic efficacy. Consequently, the ability to synthesize these structures efficiently is of paramount importance for R&D teams aiming to accelerate drug discovery pipelines.
The patent highlights that introducing fluorine atoms or fluorine-containing groups into specific positions of organic molecules can significantly alter their acidity, lipophilicity, and metabolic stability, thereby enhancing their therapeutic efficacy. Consequently, the ability to synthesize these structures efficiently is of paramount importance for R&D teams aiming to accelerate drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-trifluoromethylquinoline skeletons has been fraught with significant technical hurdles that impede efficient manufacturing and scale-up. Traditional approaches often involve the direct introduction of a trifluoromethyl group into the C-2 position of a pre-formed quinoline ring, a process that typically requires harsh reaction conditions and complex catalytic systems. These conventional methods frequently rely on expensive fluorinating reagents and necessitate the removal of large protecting groups, resulting in poor atom economy and generating substantial chemical waste. Furthermore, when dealing with complex molecular structures containing sensitive functional groups, these aggressive conditions often lead to decomposition or failure to obtain the target compound, severely limiting the scope of accessible derivatives. The reliance on transition metal catalysts or Lewis acids such as indium and rubidium not only increases raw material costs but also introduces the risk of heavy metal contamination, which is a critical quality concern for pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN115466212A offers a transformative solution by utilizing a metal-free organocatalytic condensation strategy. This innovative route employs commercially available o-aminobenzaldehydes and ethyl trifluoroacetoacetates as building blocks, reacting them in the presence of simple secondary amine catalysts like pyrrolidine or piperidine. 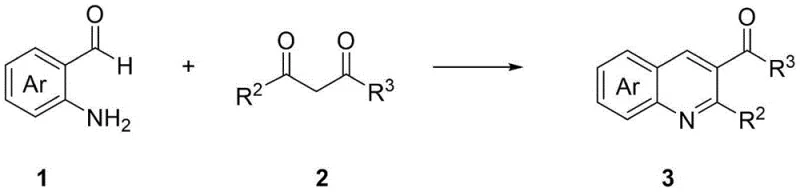 The reaction proceeds under remarkably mild conditions, typically in ethanol at temperatures ranging from 50 to 110 degrees Celsius, and generates water as the sole byproduct, exemplifying the principles of green chemistry. This approach eliminates the need for toxic metal catalysts and harsh reagents, thereby simplifying the downstream purification process and significantly improving the environmental profile of the synthesis. The high functional group tolerance observed in this system allows for the successful incorporation of diverse substituents, including halogens, nitro groups, and alkoxy groups, enabling the rapid generation of diverse libraries for biological screening.
The reaction proceeds under remarkably mild conditions, typically in ethanol at temperatures ranging from 50 to 110 degrees Celsius, and generates water as the sole byproduct, exemplifying the principles of green chemistry. This approach eliminates the need for toxic metal catalysts and harsh reagents, thereby simplifying the downstream purification process and significantly improving the environmental profile of the synthesis. The high functional group tolerance observed in this system allows for the successful incorporation of diverse substituents, including halogens, nitro groups, and alkoxy groups, enabling the rapid generation of diverse libraries for biological screening.
Mechanistic Insights into Organocatalytic Cyclization
The core of this synthetic breakthrough lies in the efficient activation of the carbonyl components by the organocatalyst, facilitating a cascade of condensation and cyclization events. The secondary amine catalyst likely forms an enamine or iminium intermediate with the trifluoroacetoacetate derivative, increasing its nucleophilicity or electrophilicity to promote the initial Knoevenagel-type condensation with the aldehyde group of the o-aminobenzaldehyde. This step is crucial for establishing the carbon-carbon bond that forms the basis of the quinoline ring system. Subsequent intramolecular cyclization involving the amino group and the activated carbonyl functionality leads to the formation of the dihydroquinoline intermediate, which then undergoes spontaneous oxidation or dehydration to yield the aromatic 2-trifluoromethylquinoline product. The use of ethanol as a solvent not only provides a polar medium conducive to these ionic mechanisms but also participates in hydrogen bonding networks that may stabilize transition states, further lowering the activation energy required for the transformation.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions commonly associated with metal-catalyzed processes. Since the reaction does not involve the cleavage and formation of C-Rf bonds under extreme conditions, the integrity of the trifluoromethyl group is preserved, preventing the formation of defluorinated byproducts that are difficult to separate. The simplicity of the reaction mixture, consisting primarily of the starting materials, catalyst, and solvent, ensures that the impurity profile is clean and predictable, which is essential for meeting stringent regulatory specifications for pharmaceutical intermediates. Moreover, the fact that water is the only byproduct means that there are no complex organic waste streams to manage, reducing the burden on wastewater treatment facilities and aligning with modern sustainability goals in chemical manufacturing.
How to Synthesize 2-Trifluoromethylquinoline Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The patent outlines a straightforward procedure where the reactants are mixed in a standard reactor equipped with stirring, followed by the addition of the catalyst and solvent to form a homogeneous solution. The reaction is then heated to the optimal temperature, typically around 90 degrees Celsius, and maintained for a period of 1 to 10 hours depending on the specific substrate reactivity. Upon completion, the workup involves simple filtration to remove any insoluble materials, followed by solvent removal under reduced pressure, leaving a crude residue that can be purified via standard silica gel column chromatography.
- Mix commercially available o-aminobenzaldehyde and ethyl trifluoroacetoacetate with an organocatalyst like pyrrolidine in ethanol.
- Heat the reaction mixture to temperatures between 50-110°C, preferably 90°C, and stir for 1 to 10 hours under air atmosphere.
- Filter the mixture, remove solvent under reduced pressure, and purify the residue via silica gel column chromatography to obtain the target quinoline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic protocol presents a compelling value proposition driven by operational simplicity and cost efficiency. The elimination of expensive transition metal catalysts and specialized fluorinating reagents directly translates to a reduction in raw material expenditures, while the use of commodity chemicals like ethanol and pyrrolidine ensures a stable and reliable supply chain. The mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility. Furthermore, the simplified purification process reduces the consumption of chromatography media and solvents during workup, leading to significant savings in operational expenses and waste disposal fees.
- Cost Reduction in Manufacturing: The removal of metal catalysts from the process flow eliminates the need for costly and time-consuming heavy metal scavenging steps, which are often required to meet strict residual metal limits in active pharmaceutical ingredients. By avoiding these additional purification stages, manufacturers can achieve substantial cost savings in both materials and labor, while also shortening the overall production cycle time. The high atom economy of the reaction ensures that a greater proportion of the starting mass is converted into the desired product, minimizing waste and maximizing the return on investment for raw material purchases.
- Enhanced Supply Chain Reliability: The reliance on commercially available and widely produced starting materials such as o-aminobenzaldehydes and ethyl trifluoroacetoacetates mitigates the risk of supply disruptions associated with specialized or proprietary reagents. This accessibility allows for flexible sourcing strategies and the ability to qualify multiple suppliers, thereby strengthening the resilience of the supply chain against market volatility. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different batches or vendors.
- Scalability and Environmental Compliance: The use of ethanol, a green and renewable solvent, aligns with increasingly stringent environmental regulations and corporate sustainability mandates, facilitating easier permitting and compliance reporting for large-scale production. The absence of toxic metal waste simplifies the handling and disposal of process effluents, reducing the environmental liability and associated costs for the manufacturing site. This green chemistry profile not only enhances the company's reputation but also future-proofs the manufacturing process against evolving regulatory landscapes regarding chemical safety and environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthetic route, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-making. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production workflows.
Q: What are the advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive and environmentally hazardous metal catalysts like indium or rubidium, simplifying purification and reducing heavy metal contamination risks in the final API.
Q: What is the typical yield and purity profile for these 2-trifluoromethylquinoline derivatives?
A: The patent reports yields up to 96% with high functional group tolerance, producing water as the only byproduct, which significantly enhances the atom economy and ease of isolation.
Q: Can this synthesis be scaled for industrial production of agrochemical intermediates?
A: Yes, the use of common solvents like ethanol and mild reaction conditions makes this process highly suitable for commercial scale-up, offering a sustainable alternative for manufacturing complex fluorinated heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethylquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing high-quality fluorinated intermediates for the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch adheres to the highest industry standards for pharmaceutical intermediates.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis. Whether you require specific COA data for regulatory filings or detailed route feasibility assessments for process optimization, our experts are ready to provide the technical depth and commercial flexibility necessary to drive your projects forward successfully.
